FACT SHEET FOR HEALTH CARE PROVIDERS EMERGENCY USE AUTHORIZATION (EUA) OF REGEN-COV® (casirivimab and imdevimab)
casirivimab with imdevimab by
Drug Labeling and Warnings
casirivimab with imdevimab by is a Prescription medication manufactured, distributed, or labeled by Regeneron Pharmaceuticals, Inc., Catalent Indiana, LLC, Integrated commercial solutions, LLC, Regeneron Pharmaceuticals, Inc, EMERGENT BIOSOLUTIONS. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CASIRIVIMAB- casirivimab injection, solution, concentrate
IMDEVIMAB- imdevimab injection, solution, concentrate
REGEN-COV- casirivimab and imdevimab injection, solution, concentrate
REGEN-COV- casirivimab and imdevimab
CASIRIVIMAB WITH IMDEVIMAB- casirivimab and imdevimab
Regeneron Pharmaceuticals, Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
FACT SHEET FOR HEALTH CARE PROVIDERS
EMERGENCY USE AUTHORIZATION (EUA) OF REGEN-COV® (casirivimab and imdevimab)
AUTHORIZED USE
TREATMENT
The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, for the treatment of mild to moderate coronavirus disease 2019 (COVID-19) in adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death.
Limitations of Authorized Use
- REGEN-COV is not authorized for treatment of mild to moderate COVID-19 in geographic regions where infection is likely to have been caused by a non-susceptible SARS-CoV-2 variant based on available information such as variant susceptibility to this drug and regional variant frequency.
- - FDA's determination and any updates will be available at: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#coviddrugs.1
- REGEN-COV (casirivimab and imdevimab) is not authorized for use in patients:
- - who are hospitalized due to COVID-19, OR
- - who require oxygen therapy due to COVID-19, OR
- - who require an increase in baseline oxygen flow rate due to COVID-19 in those on chronic oxygen therapy due to underlying non-COVID-19 related comorbidity.
- Monoclonal antibodies, such as REGEN-COV, may be associated with worse clinical outcomes when administered to hospitalized patients with COVID-19 requiring high flow oxygen or mechanical ventilation.
POST-EXPOSURE PROPHYLAXIS
The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) for post-exposure prophylaxis of COVID-19 in individuals who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- not fully vaccinated2 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications3) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Centers for Disease Control and Prevention (CDC)4 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
Limitations of Authorized Use
- REGEN-COV is not authorized for post-exposure prophylaxis of COVID-19 in geographic regions where exposure is likely to have been to a non-susceptible SARS-CoV-2 variant, based on available information including variant susceptibility to this drug and regional variant frequency.
- FDA's determination and any updates will be available at: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#coviddrugs. 5
- Post-exposure prophylaxis with REGEN-COV (casirivimab and imdevimab) is not a substitute for vaccination against COVID-19.
- REGEN-COV (casirivimab and imdevimab) is not authorized for pre-exposure prophylaxis for prevention of COVID-19.
RECENT MAJOR CHANGES
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
REGEN-COV has been authorized by FDA for the emergency uses described above.
REGEN-COV is not FDA-approved for these uses.
REGEN-COV is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of REGEN-COV under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
Treatment
This EUA is for the use of the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, for the treatment of mild to moderate COVID-19 in adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death [see Limitations of Authorized Use (1.1)].
Post-Exposure Prophylaxis
This EUA is for the use of the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) for post-exposure prophylaxis of COVID-19 in individuals who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
-
not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Center for Disease Control and Prevention (CDC)5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
Criteria for Identifying High Risk Individuals
The following medical conditions or other factors may place adults and pediatric patients (age 12-17 years and weighing at least 40 kg) at higher risk for progression to severe COVID-19:
- Older age (for example, age ≥65 years of age)
- Obesity or being overweight (for example, BMI >25 kg/m2, or if age 12-17, have BMI ≥85th percentile for their age and gender based on CDC growth charts, https://www.cdc.gov/growthcharts/clinical_charts.htm)
- Pregnancy
- Chronic kidney disease
- Diabetes
- Immunosuppressive disease or immunosuppressive treatment
- Cardiovascular disease (including congenital heart disease) or hypertension
- Chronic lung diseases (for example, chronic obstructive pulmonary disease, asthma [moderate-to-severe], interstitial lung disease, cystic fibrosis and pulmonary hypertension)
- Sickle cell disease
- Neurodevelopmental disorders (for example, cerebral palsy) or other conditions that confer medical complexity (for example, genetic or metabolic syndromes and severe congenital anomalies)
- Having a medical-related technological dependence (for example, tracheostomy, gastrostomy, or positive pressure ventilation (not related to COVID-19))
Other medical conditions or factors (for example, race or ethnicity) may also place individual patients at high risk for progression to severe COVID-19 and authorization of REGEN-COV under the EUA is not limited to the medical conditions or factors listed above. For additional information on medical conditions and factors associated with increased risk for progression to severe COVID, see the CDC website: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html. Healthcare providers should consider the benefit-risk for an individual patient.
Available Dosage Forms of REGEN-COV:
REGEN-COV (casirivimab and imdevimab) is available as:
- 1.
A single vial which contains two antibodies co-formulated in a 1:1 ratio of casirivimab and imdevimab or
- 2. Individual antibody solutions in separate vials, which may be supplied in separate cartons or together in a single carton (also referred to as a co-packaged carton), or in a dose pack.
Routes of Administration for REGEN-COV:
REGEN-COV may be administered by intravenous infusion or subcutaneous injection.
FOR TREATMENT, INTRAVENOUS INFUSION IS STRONGLY RECOMMENDED. SUBCUTANEOUS INJECTION IS AN ALTERNATIVE ROUTE OF ADMINISTRATION WHEN INTRAVENOUS INFUSION IS NOT FEASIBLE AND WOULD LEAD TO DELAY IN TREATMENT.
FOR POST-EXPOSURE PROPHYLAXIS, EITHER SUBCUTANEOUS INJECTION OR INTRAVENOUS INFUSION CAN BE USED.
Treatment Dosage
- The authorized dosage is 600 mg of casirivimab and 600 mg of imdevimab administered together as a single intravenous infusion or by subcutaneous injection as soon as possible after positive SARS-CoV-2 viral testing and within 10 days of symptom onset [see Dosage and Administration (2.2) and Clinical Trial Results and Supporting Data for EUA (18.1)].
- The authorized dosage of 600 mg of casirivimab and 600 mg of imdevimab for subcutaneous administration for treatment is selected based on the totality of the scientific evidence, incorporating clinical data, viral load reduction data (pharmacodynamics) and pharmacokinetic data [see Clinical Pharmacology (14.2) and (14.3)].
Post-exposure Prophylaxis Dosage
- The authorized dosage is 600 mg of casirivimab and 600 mg of imdevimab administered by subcutaneous injection or together as a single intravenous infusion as soon as possible following exposure to SARS-CoV-2.
- For individuals in whom repeat dosing is determined to be appropriate for ongoing exposure to SARS-CoV-2 for longer than 4 weeks and who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination, the initial dose is 600 mg of casirivimab and 600 mg of imdevimab by subcutaneous injection or intravenous infusion followed by subsequent repeat dosing of 300 mg of casirivimab and 300 mg of imdevimab by subcutaneous injection or intravenous infusion once every 4 weeks for the duration of ongoing exposure.
- The authorized dosage including dosage for repeat dosing is based on the totality of the scientific evidence including clinical pharmacology data and clinical trial data [see Clinical Trial Results and Supporting Data for EUA (18.2) and Clinical Pharmacology (14.3)].
For Intravenous Infusion:
- Co-formulated casirivimab and imdevimab solution in a vial and casirivimab and imdevimab solutions in individual vials must be diluted prior to intravenous administration.
- Administer casirivimab and imdevimab together as a single intravenous infusion via pump or gravity (see Table 1, Table 2, Table 3 and Table 4).
- Clinically monitor patients during infusion and observe patients for at least 1 hour after infusion is complete.
For Subcutaneous Injection:
- Administer casirivimab and imdevimab using the co-formulated solution in a vial or using the individual vials (see Table 5 and Table 6).
- Clinically monitor patients after injections and observe patients for at least 1 hour after injections.
- For treatment, subcutaneous injection is an alternative route of administration when intravenous administration is not feasible and would lead to delay in treatment. For post-exposure prophylaxis, either subcutaneous injection or intravenous infusion can be administered.
REGEN-COV may only be administered in settings in which health care providers have immediate access to medications to treat a severe infusion or hypersensitivity reactions, such as anaphylaxis, and the ability to activate the emergency medical system (EMS), as necessary.
Health care providers must submit a report on ALL MEDICATION ERRORS and ALL SERIOUS ADVERSE EVENTS potentially related to REGEN-COV. See Sections 8 and 9 of the Full EUA Prescribing Information for reporting instructions below.
- Patients treated with REGEN-COV should continue to self-isolate and use infection control measures (e.g., wear mask, isolate, social distance, avoid sharing personal items, clean and disinfect "high touch" surfaces, and frequent handwashing) according to CDC guidelines.
The authorized dosage may be updated as additional data from clinical trials becomes available.
For information on clinical trials that are testing the use of REGEN-COV in COVID-19, please see www.clinicaltrials.gov.
Contraindications
REGEN-COV is contraindicated in individuals with previous severe hypersensitivity reactions, including anaphylaxis, to REGEN-COV [see Warnings and Precautions (5.1)].
Dosing
Patient Selection for Treatment and Post-Exposure Prophylaxis
This section provides essential information on the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied in individual vials to be administered together in adult and pediatric patients (12 years of age and older weighing at least 40 kg) who are at high risk for progression to severe COVID-19, including hospitalization or death for:
- Treatment of mild to moderate COVID-19 in adult and pediatric patients with positive results of direct SARS-CoV-2 viral testing [see Limitations of Authorized Use (1.1)].
- Post-exposure prophylaxis of COVID-19 in high risk individuals who are:
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Centers for Disease Control and Prevention (CDC)5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
Criteria for Identifying High Risk Individuals
The following medical conditions or other factors may place adults and pediatric patients (age 12-17 years and weighing at least 40 kg) at higher risk for progression to severe COVID-19:
- Older age (for example, age ≥65 years of age)
- Obesity or being overweight (for example, BMI >25 kg/m2, or if age 12-17, have BMI ≥85th percentile for their age and gender based on CDC growth charts, https://www.cdc.gov/growthcharts/clinical_charts.htm)
- Pregnancy
- Chronic kidney disease
- Diabetes
- Immunosuppressive disease or immunosuppressive treatment
- Cardiovascular disease (including congenital heart disease) or hypertension
- Chronic lung diseases (for example, chronic obstructive pulmonary disease, asthma [moderate-to-severe], interstitial lung disease, cystic fibrosis and pulmonary hypertension)
- Sickle cell disease
- Neurodevelopmental disorders (for example, cerebral palsy) or other conditions that confer medical complexity (for example, genetic or metabolic syndromes and severe congenital anomalies)
- Having a medical-related technological dependence (for example, tracheostomy, gastrostomy, or positive pressure ventilation (not related to COVID-19))
Other medical conditions or factors (for example, race or ethnicity) may also place individual patients at high risk for progression to severe COVID-19 and authorization of REGEN-COV under the EUA is not limited to the medical conditions or factors listed above. For additional information on medical conditions and factors associated with increased risk for progression to severe COVID-19, see the CDC website: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html. Healthcare providers should consider the benefit-risk for an individual patient.
Dosage
Treatment:
The dosage in adult and pediatric patients (12 years of age and older weighing at least 40 kg) is 600 mg of casirivimab and 600 mg of imdevimab administered together as a single intravenous infusion or by subcutaneous injection. Casirivimab and imdevimab should be given together as soon as possible after positive results of direct SARS-CoV-2 viral testing and within 10 days of symptom onset.
Post-Exposure Prophylaxis:
The dosage in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) is 600 mg of casirivimab and 600 mg of imdevimab administered by subcutaneous injection or together as a single intravenous infusion. Casirivimab and imdevimab should be given together as soon as possible following exposure to SARS-CoV-2.
For individuals in whom repeat dosing is determined to be appropriate for ongoing exposure to SARS-CoV-2 for longer than 4 weeks and who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination, the initial dose is 600 mg of casirivimab and 600 mg of imdevimab by subcutaneous injection or intravenous infusion followed by subsequent repeat dosing of 300 mg of casirivimab and 300 mg of imdevimab by subcutaneous injection or intravenous infusion once every 4 weeks for the duration of ongoing exposure.
For Intravenous Infusion:
- Casirivimab and imdevimab solution co-formulated in a vial and in individual vials, including co-packaged carton and dose pack, must be diluted prior to intravenous administration.
- Administer casirivimab and imdevimab together as a single intravenous infusion via pump or gravity (see Table 1, Table 2, Table 3 and Table 4).
- Clinically monitor patients during infusion and observe patients for at least 1 hour after infusion is complete.
Dosage Adjustment in Specific Populations
No dosage adjustment is recommended in pregnant or lactating women and in patients with renal impairment [see Full EUA Prescribing Information, Use in Specific Populations (11)].
Preparation and Administration
There are TWO different formulations of REGEN-COV:
- Casirivimab and imdevimab co-formulated solution containing two antibodies in a 1:1 ratio in a vial.
- Casirivimab and imdevimab available as individual antibody solutions in separate vials supplied as follows:
- Individual vials in individual cartons, or
- together in a single carton (as referred to as a co-packaged carton), or
- in a dose pack. The dose pack contains individual vials of casirivimab and imdevimab, configurations that may vary in vial size, strength and appearance and are available in dose pack configurations that include 2, 5, and 8 cartons [see Full EUA Prescribing Information, How Supplied/Storage and Handling (19)].
For treatment, intravenous infusion is strongly recommended. Subcutaneous injection is an alternative route of administration when intravenous infusion is not feasible and would lead to delay in treatment.
For post-exposure prophylaxis, either subcutaneous injection or intravenous infusion can be used.
There are differences in the way the two formulations are prepared. Carefully follow the preparation procedures below.
- Casirivimab and imdevimab co-formulated solution in a vial and casirivimab or imdevimab as individual antibody solutions in separate 11.1 mL vials may be used to prepare more than one dose simultaneously as appropriate, either in intravenous bags or in syringes for subcutaneous injection. Discard any product remaining in the vial.
- Store unopened casirivimab and imdevimab vials in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Unopened vials may be stored in the original carton at room temperature [up to 25°C (77°F)] and must be used within 30 days. If not used in the 30 days, discard vials.
Under the EUA, a single-dose vial may be used to prepare more than one dose.
Preparation for Intravenous Infusion
For treatment, the preferred route of administration for casirivimab and imdevimab is by intravenous infusion after dilution.
Casirivimab and imdevimab solution for intravenous infusion should be prepared by a qualified healthcare professional using aseptic technique:
- Remove the casirivimab and imdevimab vials from refrigerated storage and allow to equilibrate to room temperature for approximately 20 minutes before preparation. Do not expose to direct heat. Do not shake the vials.
- Inspect casirivimab and imdevimab vials visually for particulate matter and discoloration prior to administration. Should either be observed, the vial must be discarded and replaced with a new vial.
- The solution for each vial should be clear to slightly opalescent, colorless to pale yellow.
- Obtain a prefilled intravenous infusion bag containing either 50 mL, 100 mL, 150 mL, or 250 mL of either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
- Withdraw the appropriate amount of casirivimab and imdevimab from each respective vial(s) and inject into a prefilled infusion bag containing either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP (see Table 1 and Table 2). If using one vial to prepare more than one infusion bag, then prepare all infusion bags at the same time. The product is preservative-free, therefore do not store unused solution in vial(s).
- Gently invert infusion bag by hand approximately 10 times to mix. Do not shake.
- This product is preservative-free and therefore, the diluted infusion solution should be administered immediately (see Table 3 and Table 4).
- If immediate administration is not possible, store the diluted casirivimab and imdevimab infusion solution in the refrigerator between 2°C to 8°C (36°F to 46°F) for no more than 36 hours or at room temperature up to 25°C (77°F) for no more than 4 hours. If refrigerated, allow the infusion solution to equilibrate to room temperature for approximately 30 minutes prior to administration.
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag | Preparing Using Co-Formulated Casirivimab and Imdevimab Vial | Preparing Casirivimab and Imdevimab Using Individual Vials* |
|---|---|---|
|
|
||
| 50 mL | Add 10 mL of co-formulated casirivimab and imdevimab (1 vial) into a prefilled 0.9% Sodium Chloride or 5% Dextrose infusion bag and administer as instructed below | Add:
|
| 100 mL | ||
| 150 mL | ||
| 250 mL | ||
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag | Preparing Using Co-Formulated Casirivimab and Imdevimab Vial | Preparing Casirivimab and Imdevimab Using Individual Vials† |
|---|---|---|
|
|
||
| 50 mL | Add 5 mL of co-formulated casirivimab and imdevimab into a prefilled 0.9% Sodium Chloride or 5% Dextrose infusion bag and administer as instructed below | Add:
|
| 100 mL | ||
| 150 mL | ||
| 250 mL | ||
Administration by Intravenous Infusion
Casirivimab and imdevimab infusion solution should be administered by a qualified healthcare professional using aseptic technique.
- Gather the recommended materials for infusion:
- Polyvinyl chloride (PVC), polyethylene (PE)-lined PVC, or polyurethane (PU) infusion set
- In-line or add-on 0.2 micron polyethersulfone (PES) filter
- Attach the infusion set to the intravenous bag.
- Prime the infusion set.
- Administer the entire infusion solution in the bag via pump or gravity through an intravenous line containing a sterile, in-line or add-on 0.2-micron polyethersulfone (PES) filter (see Table 3 and Table 4). Due to potential overfill of prefilled saline bags, the entire infusion solution in the bag should be administered to avoid underdosage.
- The prepared infusion solution should not be administered simultaneously with any other medication. The compatibility of casirivimab and imdevimab injection with intravenous solutions and medications other than 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP is not known.
- After infusion is complete, flush the tubing with either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP to ensure delivery of the required dose.
- Discard unused product.
- Clinically monitor patients during administration and observe patients for at least 1 hour after infusion is complete.
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag used | Maximum Infusion Rate | Minimum Infusion Time |
|---|---|---|
|
|
||
| 50 mL* | 180 mL/hr | 20 minutes |
| 100 mL | 310 mL/hr | 21 minutes |
| 150 mL | 310 mL/hr | 31 minutes |
| 250 mL | 310 mL/hr | 50 minutes |
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag used | Maximum Infusion Rate | Minimum Infusion Time |
|---|---|---|
|
|
||
| 50 mL† | 165 mL/hr | 20 minutes |
| 100 mL | 310 mL/hr | 20 minutes |
| 150 mL | 310 mL/hr | 30 minutes |
| 250 mL | 310 mL/hr | 49 minutes |
Preparation for Subcutaneous Injection
Remove the casirivimab and imdevimab vial(s) from refrigerated storage and allow to equilibrate to room temperature for approximately 20 minutes before preparation. Do not expose to direct heat. Do not shake the vials.
Inspect casirivimab and imdevimab vial(s) visually for particulate matter and discoloration prior to administration. Should either be observed, the vial must be discarded and replaced with a new vial. The solution for each vial should be clear to slightly opalescent, colorless to pale yellow.
- Casirivimab and imdevimab should be prepared using the appropriate number of syringes (see Table 5 and Table 6). Obtain 3 mL or 5 mL polypropylene Luer Lock syringes with luer connection and 21-gauge 1½ inch transfer needles.
- Withdraw the appropriate amount of solution into each syringe (see Table 5 and Table 6). Prepare all syringes at the same time.
- Replace the 21-gauge transfer needle with a 25-gauge or 27-gauge needle for subcutaneous injection.
- This product is preservative-free and therefore, the prepared syringes should be administered immediately. If immediate administration is not possible, store the prepared casirivimab and imdevimab syringes in the refrigerator between 2ºC to 8ºC (36ºF to 46ºF) for no more than 24 hours, or at room temperature up to 25ºC (77ºF) for no more than 8 hours. If refrigerated, allow the syringes to equilibrate to room temperature for approximately 20 minutes prior to administration.
| Prepare 600 mg of Casirivimab and 600 mg of Imdevimab | Preparation of 4 Syringes |
|---|---|
| Using Casirivimab and Imdevimab Co-formulated Vial | Withdraw 2.5 mL solution per syringe into FOUR separate syringes. |
| Using Casirivimab and Imdevimab Individual Vials |
|
| For total of 4 syringes. |
| Prepare 300 mg of Casirivimab and 300 mg of Imdevimab | Preparation of 2 Syringes |
|---|---|
|
|
|
| Using Casirivimab and Imdevimab Co-formulated Vial | Withdraw 2.5 mL solution per syringe into TWO separate syringes. |
| Using Casirivimab and Imdevimab Individual Vials |
|
| For total of 2 syringes. | |
Administration for Subcutaneous Injection
- For the administration of 600 mg of casirivimab and 600 mg of imdevimab, gather 4 syringes (see Table 5) and prepare for subcutaneous injections.
- For the administration of 300 mg of casirivimab and 300 mg of imdevimab, gather 2 syringes (see Table 6) and prepare for subcutaneous injections.
- Administer the subcutaneous injections consecutively, each at a different injection site, into the thigh, back of the upper arm, or abdomen, except for 2 inches (5 cm) around the navel. The waistline should be avoided.
- When administering the subcutaneous injections, it is recommended that providers use different quadrants of the abdomen or upper thighs or back of the upper arms to space apart each 2.5 mL subcutaneous injection of casirivimab and imdevimab. DO NOT inject into skin that is tender, damaged, bruised, or scarred.
- Clinically monitor patients after injections and observe patients for at least 1 hour.
Storage
Store unopened casirivimab and imdevimab vials in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Unopened vials may be stored in the original carton at room temperature [up to 25°C (77°F)] and must be used within 30 days. If not used in the 30 days, discard vials.
Warnings
There are limited clinical data available for REGEN-COV (casirivimab and imdevimab). Serious and unexpected adverse events may occur that have not been previously reported with REGEN-COV use.
Hypersensitivity Including Anaphylaxis and Infusion-Related Reactions
Serious hypersensitivity reactions, including anaphylaxis, have been observed with administration of REGEN-COV (casirivimab and imdevimab). If signs or symptoms of a clinically significant hypersensitivity reaction or anaphylaxis occur, immediately discontinue administration and initiate appropriate medications and/or supportive therapy.
Infusion-related reactions, occurring during the infusion and up to 24 hours after the infusion, have been observed with administration of REGEN-COV. These reactions may be severe or life-threatening.
Signs and symptoms of infusion-related reactions may include:
- fever, difficulty breathing, reduced oxygen saturation, chills, fatigue, arrhythmia (e.g., atrial fibrillation, tachycardia, bradycardia), chest pain or discomfort, weakness, altered mental status, nausea, headache, bronchospasm, hypotension, hypertension, angioedema, throat irritation, rash including urticaria, pruritus, myalgia, vasovagal reactions (e.g., pre-syncope, syncope), dizziness, and diaphoresis.
Consider slowing or stopping the infusion and administer appropriate medications and/or supportive care if an infusion-related reaction occurs.
Hypersensitivity reactions occurring more than 24 hours after the infusion have also been reported with the use of REGEN-COV under Emergency Use Authorization.
Clinical Worsening After REGEN-COV Administration
Clinical worsening of COVID-19 after administration of REGEN-COV has been reported and may include signs or symptoms of fever, hypoxia or increased respiratory difficulty, arrhythmia (e.g., atrial fibrillation, tachycardia, bradycardia), fatigue, and altered mental status. Some of these events required hospitalization. It is not known if these events were related to REGEN-COV use or were due to progression of COVID-19.
Limitations of Benefit and Potential for Risk in Patients with Severe COVID-19
Monoclonal antibodies, such as REGEN-COV, may be associated with worse clinical outcomes when administered to hospitalized patients with COVID-19 requiring high flow oxygen or mechanical ventilation. Therefore, REGEN-COV is not authorized for use in patients [see Limitations of Authorized Use (1.1)]:
- who are hospitalized due to COVID-19, OR
- who require oxygen therapy due to COVID-19, OR
- who require an increase in baseline oxygen flow rate due to COVID-19 in those on chronic oxygen therapy due to underlying non-COVID-19 related comorbidity.
Side Effects
Adverse events have been reported with REGEN-COV (casirivimab and imdevimab) [see Full EUA Prescribing Information, Clinical Trials Experience (6.1)].
Additional adverse events associated with REGEN-COV, some of which may be serious, may become apparent with more widespread use.
INSTRUCTIONS FOR HEALTH CARE PROVIDERS
As the health care provider, you must communicate to your patient or parent/caregiver, as age appropriate, information consistent with the "Fact Sheet for Patients, Parents and Caregivers" (and provide a copy of the Fact Sheet) prior to the patient receiving REGEN-COV (casirivimab and imdevimab), including:
- FDA has authorized the emergency use of REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, for the treatment of mild to moderate COVID-19 in adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death [see Limitations of Authorized Use (1.1)].
- FDA has authorized the emergency use of REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) for post-exposure prophylaxis of COVID-19 in individuals who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Center for Disease Control and Prevention (CDC)5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- The patient or parent/caregiver has the option to accept or refuse REGEN-COV.
- The significant known and potential risks and benefits of REGEN-COV, and the extent to which such risks and benefits are unknown.
- Information on available alternative treatments and the risks and benefits of those alternatives, including clinical trials.
- Patients treated with REGEN-COV should continue to self-isolate and use infection control measures (e.g., wear mask, isolate, social distance, avoid sharing personal items, clean and disinfect "high touch" surfaces, and frequent handwashing) according to CDC guidelines.
For information on clinical trials that are testing the use of REGEN-COV related to COVID-19, please see www.clinicaltrials.gov.
MANDATORY REQUIREMENTS FOR REGEN-COV UNDER EMERGENCY USE AUTHORIZATION:
In order to mitigate the risks of using this unapproved product under EUA and to optimize the potential benefit of REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, the following items are required. Use of REGEN-COV under this EUA is limited to the following (all requirements must be met):
- Treatment of mild to moderate COVID-19 in adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing and who are at high risk for severe COVID-19, including hospitalization or death [see Limitations of Authorized Use (1.1)].
- Post-exposure prophylaxis of COVID-19 in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- a.
not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Center for Disease Control and Prevention (CDC)5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
- a.
not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- As the health care provider, communicate to your patient or parent/caregiver, as age appropriate, information consistent with the "Fact Sheet for Patients, Parents and Caregivers" prior to the patient receiving REGEN-COV. Health care providers (to the extent practicable given the circumstances of the emergency) must document in the patient's medical record that the patient/caregiver has been:
- a. Given the "Fact Sheet for Patients, Parents and Caregivers",
- b. Informed of alternatives to receiving REGEN-COV, and
- c. Informed that REGEN-COV is an unapproved drug that is authorized for use under this Emergency Use Authorization.
- Patients with known hypersensitivity to any ingredient of REGEN-COV must not receive REGEN-COV.
- The prescribing health care provider and/or the provider's designee are/is responsible for mandatory reporting of all medication errors and serious adverse events* potentially related to REGEN-COV treatment within 7 calendar days from the onset of the event. The reports must include unique identifiers and the words "REGEN-COV use for COVID-19 under Emergency Use Authorization (EUA)" in the description section of the report.
- Submit adverse event reports to FDA MedWatch using one of the following methods:
- Complete and submit the report online: www.fda.gov/medwatch/report.htm, or
- Complete and submit a postage-paid FDA Form 3500 (https://www.fda.gov/media/76299/download) and return by:
- Mail to MedWatch, 5600 Fishers Lane, Rockville, MD 20852-9787, or
- Fax (1-800-FDA-0178), or
- Call 1-800-FDA-1088 to request a reporting form
- Submitted reports must include in the field name, "Describe Event, Problem, or Product Use/Medication Error" a statement "REGEN-COV use for COVID-19 under Emergency Use Authorization (EUA)."
- death;
- a life-threatening adverse event;
- inpatient hospitalization or prolongation of existing hospitalization;
- a persistent or significant incapacity or substantial disruption of the ability to conduct normal life functions;
- a congenital anomaly/birth defect;
- a medical or surgical intervention to prevent death, a life-threatening event, hospitalization, disability, or congenital anomaly.
- Submit adverse event reports to FDA MedWatch using one of the following methods:
- The prescribing health care provider and/or the provider's designee is/are responsible for mandatory responses to requests from FDA for information about adverse events and medication errors following receipt of REGEN-COV.
- OTHER REPORTING REQUIREMENTS
- Healthcare facilities and providers must report therapeutics information and utilization data through HHS Protect, Teletracking or National Healthcare Safety Network (NHSN) as directed by the U.S. Department of Health and Human Services.
- In addition, please provide a copy of all FDA MedWatch forms to:
Regeneron Pharmaceuticals, Inc
Fax: 1-888-876-2736
E-mail: medical.information@regeneron.com
Or call Regeneron Pharmaceuticals at 1-844-734-6643 to report adverse events.
APPROVED AVAILABLE ALTERNATIVES
Veklury (remdesivir) is FDA-approved for the treatment of COVID-19 in adults and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, who are not hospitalized and have mild-to-moderate COVID-19, and who are at high risk for progression to severe COVID-19, including hospitalization or death. Veklury is administered via intravenous infusion for a total treatment duration of 3 days.
Although Veklury is an approved alternative treatment of mild-to-moderate COVID-19 in adults and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death, FDA does not consider Veklury to be an adequate alternative to REGEN-COV for this authorized use because it may not be feasible or practical for certain patients (e.g., it requires a 3-day treatment duration).
There is no adequate, approved and available alternative to REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, for post-exposure prophylaxis of COVID-19 in individuals who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per CDC5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
Additional information on COVID-19 treatments can be found at https://www.cdc.gov/coronavirus/2019-ncov/index.html. The health care provider should visit https://clinicaltrials.gov/ to determine whether the patient may be eligible for enrollment in a clinical trial.
AUTHORITY FOR ISSUANCE OF THE EUA
The Secretary of the Department of Health and Human Services (HHS) has declared a public health emergency that justifies the emergency use of drugs and biological products during the COVID-19 pandemic.
FDA has issued this EUA, requested by Regeneron Pharmaceuticals, Inc. for the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, for the treatment of mild to moderate COVID-19 in adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing and who are at high risk for progression to severe COVID-19, including hospitalization or death.6 As a health care provider, you must comply with the mandatory requirements of the EUA (see above).
FDA has issued this EUA, requested by Regeneron Pharmaceuticals, Inc. for the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) for post-exposure prophylaxis of COVID-19 in individuals who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Centers for Disease Control and Prevention (CDC)5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
Although limited scientific information is available, based on the totality of the scientific evidence available to date, it is reasonable to believe that REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, may be effective for the treatment of COVID-19 or for post-exposure prophylaxis of COVID-19 in individuals as specified in this Fact Sheet. You may be contacted and asked to provide information to help with the assessment of the use of the product during this emergency.
This EUA for REGEN-COV will end when the Secretary determines that the circumstances justifying the EUA no longer exist or when there is a change in the approval status of the product such that an EUA may no longer be needed.
CONTACT INFORMATION
For additional information visit www.REGENCOV.com
If you have questions, please contact Regeneron at 1-844-734-6643.
FULL EUA PRESCRIBING INFORMATION
| FULL EUA PRESCRIBING INFORMATION: | 11 USE IN SPECIFIC POPULATIONS |
| CONTENTS* | 11.1 Pregnancy |
| 1 AUTHORIZED USE | 11.2 Lactation |
| 1.1 TREATMENT | 11.3 Pediatric Use |
| 1.2 POST-EXPOSURE PROPHYLAXIS | 11.4 Geriatric Use |
| 2 DOSAGE AND ADMINISTRATION | 11.5 Renal Impairment |
| 2.1 Patient Selection | 11.6 Hepatic Impairment |
| 2.2 Dosage | 11.7 Other Specific Populations |
| 2.3 Dose Adjustment in Specific Populations | 12 OVERDOSAGE |
| 2.4 Dose Preparation and Administration | 13 PRODUCT DESCRIPTION |
| 3 DOSAGE FORMS AND STRENGTHS | 14 CLINICAL PHARMACOLOGY |
| 4 CONTRAINDICATIONS | 14.1 Mechanism of Action |
| 5 WARNINGS AND PRECAUTIONS | 14.2 Pharmacodynamics |
| 5.1 Hypersensitivity including Anaphylaxis and Infusion- | 14.3 Pharmacokinetics |
| Related Reactions | 15 MICROBIOLOGY |
| 5.2 Clinical Worsening After REGEN-COV Administration | 16 NONCLINICAL TOXICOLOGY |
| 5.3 Limitations of Benefit and Potential for Risk in Patients | 17 ANIMAL PHARMACOLOGIC AND EFFICACY DATA |
| with Severe COVID-19 | 18 CLINICAL TRIAL RESULTS AND SUPPORTING DATA FOR EUA |
| 6 OVERALL SAFETY SUMMARY | 18.1 Treatment of Mild to Moderate COVID-19 (COV-2067) |
| 6.1 Clinical Trials Experience | 18.2 Post-exposure Prophylaxis of COVID-19 (COV-2069) |
| 7 PATIENT MONITORING RECOMMENDATIONS | 19 HOW SUPPLIED/STORAGE AND HANDLING |
| 8 ADVERSE REACTIONS AND MEDICATION ERRORS | 20 PATIENT COUNSELING INFORMATION |
| REPORTING REQUIREMENTS AND INSTRUCTIONS | 21 CONTACT INFORMATION |
| 9 OTHER REPORTING REQUIREMENTS | * Sections or subsections omitted from the full prescribing information are |
| 10 DRUG INTERACTIONS | not listed. |
1 AUTHORIZED USE
1.1 TREATMENT
REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, is authorized for use under an EUA for the treatment of mild to moderate COVID-19 in adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death.
Limitations of Authorized Use
- REGEN-COV is not authorized for treatment of mild to moderate COVID-19 in geographic regions where infection is likely to have been caused by a non-susceptible SARS-CoV-2 variant based on available information such as variant susceptibility to this drug and regional variant frequency.
- - FDA's determination and any updates will be available at: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#coviddrugs.1
- REGEN-COV (casirivimab and imdevimab) is not authorized for use in patients:
- - who are hospitalized due to COVID-19, OR
- - who require oxygen therapy due to COVID-19, OR
- - who require an increase in baseline oxygen flow rate due to COVID-19 in those on chronic oxygen therapy due to underlying non-COVID-19 related comorbidity.
- Monoclonal antibodies, such as REGEN-COV, may be associated with worse clinical outcomes when administered to hospitalized patients with COVID-19 requiring high flow oxygen or mechanical ventilation [see Warnings and Precautions (5.2)].
1.2 POST-EXPOSURE PROPHYLAXIS
REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, is authorized for use under an EUA for the post-exposure prophylaxis of COVID-19 in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- not fully vaccinated7 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications8) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Centers for Disease Control and Prevention (CDC)9 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons).
Limitations of Authorized Use
- REGEN-COV is not authorized for post-exposure prophylaxis of COVID-19 in geographic regions where exposure is likely to have been to a non-susceptible SARS-CoV-2 variant, based on available information including variant susceptibility to this drug and regional variant frequency.
- - FDA's determination and any updates will be available at: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#coviddrugs. 5
- Post-exposure prophylaxis with REGEN-COV (casirivimab and imdevimab) is not a substitute for vaccination against COVID-19.
- REGEN-COV (casirivimab and imdevimab) is not authorized for pre-exposure prophylaxis for prevention of COVID-19.
2 DOSAGE AND ADMINISTRATION
2.1 Patient Selection
The optimal dosing regimen for treatment of COVID-19 has not yet been established.
The recommended dosing regimen may be updated as data from clinical trials become available.
Patient Selection for Treatment and Post-Exposure Prophylaxis
Treatment:
This section provides essential information on the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied as individual vials to be administered together, for the treatment of adult and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death [see Limitations of Authorized Use (1.1)].
Post-Exposure Prophylaxis:
This section provides essential information on the unapproved product, REGEN-COV (casirivimab and imdevimab) co-formulated product and REGEN-COV (casirivimab and imdevimab) supplied in individual vials to be administered together, in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) for the post-exposure prophylaxis of COVID-19 in individuals who are at high risk for progression to severe COVID-19, including hospitalization or death, and are:
- not fully vaccinated3 or who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination (for example, individuals with immunocompromising conditions including those taking immunosuppressive medications4) and
- - have been exposed to an individual infected with SARS-CoV-2 consistent with close contact criteria per Centers for Disease Control and Prevention (CDC)5 or
- - who are at high risk of exposure to an individual infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons) [see Limitations of Authorized Use (1.2)].
The following medical conditions or other factors may place adults and pediatric patients (age 12-17 years and weighing at least 40 kg) at higher risk for progression to severe COVID-19:
- Older age (for example, age ≥65 years of age)
- Obesity or being overweight (for example, BMI >25 kg/m2, or if age 12-17, have BMI ≥85th percentile for their age and gender based on CDC growth charts, https://www.cdc.gov/growthcharts/clinical_charts.htm)
- Pregnancy
- Chronic kidney disease
- Diabetes
- Immunosuppressive disease or immunosuppressive treatment
- Cardiovascular disease (including congenital heart disease) or hypertension
- Chronic lung diseases (for example, chronic obstructive pulmonary disease, asthma [moderate-to-severe], interstitial lung disease, cystic fibrosis and pulmonary hypertension)
- Sickle cell disease
- Neurodevelopmental disorders (for example, cerebral palsy) or other conditions that confer medical complexity (for example, genetic or metabolic syndromes and severe congenital anomalies)
- Having a medical-related technological dependence (for example, tracheostomy, gastrostomy, or positive pressure ventilation (not related to COVID-19))
Other medical conditions or factors (for example, race or ethnicity) may also place individual patients at high risk for progression to severe COVID-19 and authorization of REGEN-COV under the EUA is not limited to the medical conditions or factors listed above.
For additional information on medical conditions and factors associated with increased risk for progression to severe COVID-19, see the CDC website: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html. Healthcare providers should consider the benefit-risk for an individual patient.
2.2 Dosage
Treatment:
The dosage in adult and pediatric patients (12 years of age and older weighing at least 40 kg) is 600 mg of casirivimab and 600 mg of imdevimab administered together as a single intravenous infusion or by subcutaneous injection. Casirivimab and imdevimab should be given together as soon as possible after a positive viral test for SARS-CoV-2 and within 10 days of symptom onset.
Post-Exposure Prophylaxis:
The dosage in adult and pediatric individuals (12 years of age and older weighing at least 40 kg) is 600 mg of casirivimab and 600 mg of imdevimab administered by subcutaneous injection or together as a single intravenous infusion. Casirivimab and imdevimab should be given together as soon as possible following exposure to SARS-CoV-2.
For individuals whom repeat dosing is determined to be appropriate for ongoing exposure to SARS-CoV-2 for longer than 4 weeks and who are not expected to mount an adequate immune response to complete SARS-CoV-2 vaccination, the initial dose is 600 mg of casirivimab and 600 mg of imdevimab by subcutaneous injection or intravenous infusion followed by subsequent repeat dosing of 300 mg of casirivimab and 300 mg of imdevimab by subcutaneous injection or intravenous infusion once every 4 weeks for the duration of ongoing exposure.
For Intravenous Infusion:
- Casirivimab and imdevimab solution co-formulated in a vial and in individual vials, including co-packaged carton and dose pack, must be diluted prior to intravenous administration.
- Administer casirivimab and imdevimab together as a single intravenous infusion via pump or gravity (see Table 1, Table 2, Table 3 and Table 4).
- Clinically monitor patients during infusion and observe patients for at least 1 hour after infusion is complete.
2.3 Dose Adjustment in Specific Populations
Pregnancy or Lactation
No dosage adjustment is recommended in pregnant or lactating women [see Use in Specific Populations (11.1, 11.2)].
Pediatric Use
No dosage adjustment is recommended in pediatric patients who weigh at least 40 kg and are older than 12 years of age. REGEN-COV (casirivimab and imdevimab) is not recommended for pediatric patients weighing less than 40 kg or those less than 12 years of age [see Use in Specific Populations (11.3)].
Renal Impairment
No dosage adjustment is recommended in patients with renal impairment [see Use in Specific Populations (11.5)].
2.4 Dose Preparation and Administration
There are TWO different formulations of REGEN-COV:
- Casirivimab and imdevimab co-formulated solution containing two antibodies in a 1:1 ratio in a vial.
- Casirivimab and imdevimab available as individual antibody solutions in separate vials supplied as follows:
- Individual vials in individual cartons, or
- together in a single carton (also referred to as a co-packaged carton), or
- in a dose pack. The dose pack contains individual vials of casirivimab and imdevimab, configurations that may vary in vial size, strength and appearance and are available in dose pack configurations that include 2, 5, and 8 cartons [see Full EUA Prescribing Information, How Supplied/Storage and Handling (19)].
For treatment, intravenous infusion is strongly recommended. Subcutaneous injection is an alternative route of administration when intravenous infusion is not feasible and would lead to delay in treatment.
For post-exposure prophylaxis, either subcutaneous injection or intravenous infusion can be used.
There are differences in the way the two formulations are prepared. Carefully follow the preparation procedures below.
- Casirivimab and imdevimab co-formulated solution in a vial and casirivimab or imdevimab as individual antibody solutions in separate 11.1 mL vials may be used to prepare more than one dose simultaneously as appropriate, either in intravenous bags or in syringes for subcutaneous injection. Discard any product remaining in the vial.
- Store unopened casirivimab and imdevimab vials in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Unopened vials may be stored in the original carton at room temperature [up to 25°C (77°F)] and must be used within 30 days. If not used in the 30 days, discard vials.
Under the EUA, a single-dose vial may be used to prepare more than one dose.
Preparation for Intravenous Infusion
For treatment, the preferred route of administration for casirivimab and imdevimab is by intravenous infusion after dilution.
Casirivimab and imdevimab solution for intravenous infusion should be prepared by a qualified healthcare professional using aseptic technique:
- Remove the casirivimab and imdevimab vials from refrigerated storage and allow to equilibrate to room temperature for approximately 20 minutes before preparation. Do not expose to direct heat. Do not shake the vials.
- Inspect casirivimab and imdevimab vials visually for particulate matter and discoloration prior to administration. Should either be observed, the vial must be discarded and replaced with a new vial.
- The solution for each vial should be clear to slightly opalescent, colorless to pale yellow.
- Obtain a prefilled intravenous infusion bag containing either 50 mL, 100 mL, 150 mL, or 250 mL of either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
- Withdraw the appropriate amount of casirivimab and imdevimab from each respective vial(s) and inject into a prefilled infusion bag containing either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP, (see Table 1 and Table 2). If using one vial to prepare more than one infusion bag, then prepare all infusion bags at the same time. The product is preservative-free, therefore do not store unused solution in vial(s).
- Gently invert infusion bag by hand approximately 10 times to mix. Do not shake.
- This product is preservative-free and therefore, the diluted infusion solution should be administered immediately (see Table 3 and Table 4).
- If immediate administration is not possible, store the diluted casirivimab and imdevimab infusion solution in the refrigerator between 2°C to 8°C (36°F to 46°F) for no more than 36 hours or at room temperature up to 25°C (77°F) for no more than 4 hours. If refrigerated, allow the infusion solution to equilibrate to room temperature for approximately 30 minutes prior to administration.
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag | Preparing Using Co-Formulated Casirivimab and Imdevimab Vial | Preparing Casirivimab and Imdevimab Using Individual Vials* |
|---|---|---|
|
|
||
| 50 mL | Add 10 mL of co-formulated casirivimab and imdevimab (1 vial) into a prefilled 0.9% Sodium Chloride or 5% Dextrose infusion bag and administer as instructed below | Add:
|
| 100 mL | ||
| 150 mL | ||
| 250 mL | ||
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag | Preparing Using Co-Formulated Casirivimab and Imdevimab Vial | Preparing Casirivimab and Imdevimab Using Individual Vials† |
|---|---|---|
|
|
||
| 50 mL | Add 5 mL of co-formulated casirivimab and imdevimab into a prefilled 0.9% Sodium Chloride or 5% Dextrose infusion bag and administer as instructed below | Add:
|
| 100 mL | ||
| 150 mL | ||
| 250 mL | ||
Administration by Intravenous Infusion
Casirivimab and imdevimab infusion solution should be administered by a qualified healthcare professional using aseptic technique.
- Gather the recommended materials for infusion:
- Polyvinyl chloride (PVC), polyethylene (PE)-lined PVC, or polyurethane (PU) infusion set
- In-line or add-on 0.2 micron polyethersulfone (PES) filter
- Attach the infusion set to the intravenous bag.
- Prime the infusion set.
- Administer the entire infusion solution in the bag via pump or gravity through an intravenous line containing a sterile, in-line or add-on 0.2-micron polyethersulfone (PES) filter (see Table 3 and Table 4). Due to potential overfill of prefilled saline bags, the entire infusion solution in the bag should be administered to avoid underdosage.
- The prepared infusion solution should not be administered simultaneously with any other medication. The compatibility of casirivimab and imdevimab injection with intravenous solutions and medications other than 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP is not known.
- After infusion is complete, flush the tubing with either 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP to ensure delivery of the required dose.
- Discard unused product.
- Clinically monitor patients during administration and observe patients for at least 1 hour after infusion is complete.
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag used | Maximum Infusion Rate | Minimum Infusion Time |
|---|---|---|
|
|
||
| 50 mL* | 180 mL/hr | 20 minutes |
| 100 mL | 310 mL/hr | 21 minutes |
| 150 mL | 310 mL/hr | 31 minutes |
| 250 mL | 310 mL/hr | 50 minutes |
| Size of Prefilled 0.9% Sodium Chloride or 5% Dextrose Infusion Bag used | Maximum Infusion Rate | Minimum Infusion Time |
|---|---|---|
|
|
||
| 50 mL† | 165 mL/hr | 20 minutes |
| 100 mL | 310 mL/hr | 20 minutes |
| 150 mL | 310 mL/hr | 30 minutes |
| 250 mL | 310 mL/hr | 49 minutes |
Preparation for Subcutaneous Injection
Remove the casirivimab and imdevimab vial(s) from refrigerated storage and allow to equilibrate to room temperature for approximately 20 minutes before preparation. Do not expose to direct heat. Do not shake the vials.
Inspect casirivimab and imdevimab vial(s) visually for particulate matter and discoloration prior to administration. Should either be observed, the vial must be discarded and replaced with a new vial. The solution for each vial should be clear to slightly opalescent, colorless to pale yellow.
- Casirivimab and imdevimab should be prepared using the appropriate number of syringes (see Table 5 and Table 6). Obtain 3 mL or 5 mL polypropylene Luer Lock syringes with luer connection and 21-gauge 1½ inch transfer needles.
- Withdraw the appropriate amount of solution into each syringe (see Table 5 and Table 6). Prepare all syringes at the same time.
- Replace the 21-gauge transfer needle with a 25-gauge or 27-gauge needle for subcutaneous injection.
- This product is preservative-free and therefore, the prepared syringes should be administered immediately. If immediate administration is not possible, store the prepared casirivimab and imdevimab syringes in the refrigerator between 2ºC to 8ºC (36ºF to 46ºF) for no more than 24 hours, or at room temperature up to 25ºC (77ºF) for no more than 8 hours. If refrigerated, allow the syringes to equilibrate to room temperature for approximately 20 minutes prior to administration.
| Prepare 600 mg of Casirivimab and 600 mg of Imdevimab | Preparation of 4 Syringes |
|---|---|
| Using Casirivimab and Imdevimab Co-formulated Vial | Withdraw 2.5 mL solution per syringe into FOUR separate syringes. |
| Using Casirivimab and Imdevimab Individual Vials |
|
| For total of 4 syringes. |
| Prepare 300 mg of Casirivimab and 300 mg of Imdevimab | Preparation of 2 Syringes |
|---|---|
|
|
|
| Using Casirivimab and Imdevimab Co-formulated Vial | Withdraw 2.5 mL solution per syringe into TWO separate syringes. |
| Using Casirivimab and Imdevimab Individual Vials |
|
| For total of 2 syringes. | |
Administration for Subcutaneous Injection
- For the administration of 600 mg of casirivimab and 600 mg of imdevimab, gather 4 syringes (see Table 5) and prepare for subcutaneous injections.
- For the administration of 300 mg of casirivimab and 300 mg of imdevimab, gather 2 syringes (see Table 6) and prepare for subcutaneous injections.
- Administer the subcutaneous injections consecutively, each at a different injection site, into the thigh, back of the upper arm, or abdomen, except for 2 inches (5 cm) around the navel. The waistline should be avoided.
- When administering the subcutaneous injections, it is recommended that providers use different quadrants of the abdomen or upper thighs or back of the upper arms to space apart each 2.5 mL subcutaneous injection of casirivimab and imdevimab. DO NOT inject into skin that is tender, damaged, bruised, or scarred.
- Clinically monitor patients after injections and observe patients for at least 1 hour.
3 DOSAGE FORMS AND STRENGTHS
REGEN-COV (casirivimab and imdevimab) is available as:
- A single vial which contains two antibodies co-formulated in a 1:1 ratio of casirivimab and imdevimab. Co-formulated casirivimab and imdevimab is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution available as:
- Injection: 600 mg of casirivimab and 600 mg of imdevimab per 10 mL (60 mg/60 mg per mL) in a single-dose10 vial
- Individual antibody solutions in separate single-dose1 vials, which may be supplied in separate cartons or together in a single carton (also referred to as a co-packaged carton), or as dose pack.
- Casirivimab is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution available as:
- Injection: 300 mg/2.5 mL (120 mg/mL) or 1,332 mg/11.1 mL (120 mg/mL)
- Imdevimab is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution available as:
- Injection: 300 mg/2.5 mL (120 mg/mL) or 1,332 mg/11.1 mL (120 mg/mL)
- Each REGEN-COV dose pack contains 1,200 mg of casirivimab [REGN10933] and 1,200 mg of imdevimab [REGN10987] [see How Supplied/Storage and Handling (19)]. Casirivimab and imdevimab vial labels and carton labeling may instead be labeled REGN10933 and REGN10987, respectively.
- Casirivimab is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution available as:
4 CONTRAINDICATIONS
REGEN-COV is contraindicated in individuals with previous severe hypersensitivity reactions, including anaphylaxis, to REGEN-COV [see Warnings and Precautions (5.1)].
5 WARNINGS AND PRECAUTIONS
There are limited clinical data available for REGEN-COV (casirivimab and imdevimab). Serious and unexpected adverse events may occur that have not been previously reported with REGEN-COV use.
5.1 Hypersensitivity including Anaphylaxis and Infusion-Related Reactions
Serious hypersensitivity reactions, including anaphylaxis, have been reported with administration of REGEN-COV (casirivimab and imdevimab). If signs or symptoms of a clinically significant hypersensitivity reaction or anaphylaxis occur, immediately discontinue administration and initiate appropriate medications and/or supportive care.
Infusion-related reactions, occurring during the infusion and up to 24 hours after the infusion, have been observed with administration of REGEN-COV. These reactions may be severe or life threatening.
Signs and symptoms of infusion related reactions may include:
- fever, difficulty breathing, reduced oxygen saturation, chills, nausea, arrhythmia (e.g., atrial fibrillation, tachycardia, bradycardia), chest pain or discomfort, weakness, altered mental status, headache, bronchospasm, hypotension, hypertension, angioedema, throat irritation, rash including urticaria, pruritus, myalgia, vasovagal reactions (e.g., pre-syncope, syncope), dizziness, fatigue, and diaphoresis [see Overall Safety Summary (6.1)].
If an infusion-related reaction occurs, consider slowing or stopping the infusion and administer appropriate medications and/or supportive care.
Hypersensitivity reactions occurring more than 24 hours after the infusion have also been reported with the use of REGEN-COV under Emergency Use Authorization.
5.2 Clinical Worsening After REGEN-COV Administration
Clinical worsening of COVID-19 after administration of REGEN-COV has been reported and may include signs or symptoms of fever, hypoxia or increased respiratory difficulty, arrhythmia (e.g., atrial fibrillation, tachycardia, bradycardia), fatigue, and altered mental status. Some of these events required hospitalization. It is not known if these events were related to REGEN-COV use or were due to progression of COVID-19.
5.3 Limitations of Benefit and Potential for Risk in Patients with Severe COVID-19
Monoclonal antibodies, such as REGEN-COV, may be associated with worse clinical outcomes when administered to hospitalized patients with COVID-19 requiring high flow oxygen or mechanical ventilation. Therefore, REGEN-COV is not authorized for use in patients [see Limitations of Authorized Use (1.1)]:
- who are hospitalized due to COVID-19, OR
- who require oxygen therapy due to COVID-19, OR
- who require an increase in baseline oxygen flow rate due to COVID-19 in those on chronic oxygen therapy due to underlying non-COVID-19 related comorbidity.
6 OVERALL SAFETY SUMMARY
6.1 Clinical Trials Experience
Overall, approximately 16,000 subjects have been exposed to REGEN-COV (casirivimab and imdevimab) in clinical trials in hospitalized and non-hospitalized subjects. Approximately 13,500 subjects received intravenous infusions and 2,500 subjects received subcutaneous injections.
The safety of REGEN-COV (casirivimab and imdevimab) is based on analyses from COV-2067, a Phase 1/2/3 trial of ambulatory (non-hospitalized) subjects with COVID-19; COV-2069, a Phase 3 post-exposure prophylaxis trial for prevention of COVID-19; and COV-2093, a Phase 1 trial evaluating the safety and pharmacokinetics of REGEN-COV repeat subcutaneous dosing every 4 weeks for 24 weeks.
COV-2067
This is a randomized, double-blind, placebo-controlled clinical trial in subjects with mild to moderate COVID-19 who had a sample collected for the first positive SARS-CoV-2 viral infection determination within 3 days prior to the start of the infusion. In the phase 3 portion of the trial, subjects were treated with a single intravenous infusion of 600 mg of casirivimab and 600 mg of imdevimab (n=827), or 1,200 mg of casirivimab and 1,200 mg of imdevimab (n=1,849), or 4,000 mg of casirivimab and 4,000 mg of imdevimab (n=1,012), or placebo (n=1,843). REGEN-COV is not authorized at the 4,000 mg of casirivimab and 4,000 mg of imdevimab dose. The 1,200 mg of casirivimab and 1,200 mg of imdevimab is no longer authorized under this EUA [see Clinical Trial Results and Supporting Data for EUA (18)].
In pooled phase 1/2/3 analysis, infusion-related reactions (adverse event assessed as causally related by the investigator) of grade 2 or higher severity have been observed in 10/4,206 (0.2%) of those who received REGEN-COV at the authorized dose or a higher dose [see Warnings and Precautions (5.1)].
Overall, in Phase 1/2/3, three subjects receiving the 8,000 mg dose of REGEN-COV, and one subject receiving the 1,200 mg of casirivimab and 1,200 mg of imdevimab infusion-related reactions (urticaria, pruritus, flushing, pyrexia, shortness of breath, chest tightness, nausea, vomiting, rash) which resulted in permanent discontinuation of the infusion. All events resolved [see Warnings and Precautions (5.1)].
Anaphylactic reactions have been reported in the clinical program in subjects receiving REGEN-COV. The events began within 1 hour of completion of the infusion, and in at least one case required treatment including epinephrine. The events resolved.
COV-2069
This is a randomized, double-blind, placebo-controlled clinical trial assessing the efficacy and safety of REGEN-COV (casirivimab and imdevimab) for post-exposure prophylaxis of COVID-19 in household contacts of individuals infected with SARS-CoV-2. Subjects who were SARS-CoV-2 negative at baseline were enrolled in Cohort A and received a single dose of 600 mg of casirivimab and 600 mg of imdevimab subcutaneously (n=1,311) or placebo (n=1,306).
Adverse events were reported in 265 subjects (20%) in the REGEN-COV group and 379 subjects (29%) in the placebo group. Injection site reactions (all grade 1 and 2) occurred in 55 subjects (4%) in the REGEN-COV group and 19 subjects (2%) in the placebo group. The most common signs and symptoms of injection site reactions which occurred in at least 1% of subjects in the REGEN-COV group were erythema and pruritus. Hypersensitivity reactions occurred in 2 subjects (0.2%) in the REGEN-COV group and all hypersensitivity reactions were grade 1 in severity. There were no cases of anaphylaxis.
Subjects who were SARS-CoV-2 positive at baseline were enrolled in Cohort B and received a single dose of 600 mg of casirivimab and 600 mg of imdevimab subcutaneously (n=155) or placebo (n=156).
Adverse events were reported in 52 subjects (34%) in the REGEN-COV group and 75 subjects (48%) in the placebo group. Injection site reactions, all of which were grade 1 or 2, occurred in 6 subjects (4%) in the REGEN-COV group and 1 subject (1%) in the placebo group. The most common signs and symptoms of injection site reactions which occurred in at least 1% of subjects in the REGN-COV group were ecchymosis and erythema. There were no cases of hypersensitivity reaction or anaphylaxis.
COV-2093
This is a randomized double-blind, placebo-controlled Phase 1 trial evaluating the safety, pharmacokinetic and immunogenicity of repeated doses of 600 mg of casirivimab and 600 mg of imdevimab administered subcutaneously in healthy adult subjects. In COV-2093, subjects were randomized 3:1 to REGEN-COV (n=729) or placebo (n=240) administered every 4 weeks for 24 weeks. Adverse events were reported in 380 subjects (52%) in the REGEN-COV group and 111 subjects (46%) in the placebo group. Injection site reactions occurred in 12% and 4% of subjects following single dose administration in the REGEN-COV and placebo groups, respectively; the remaining safety findings following subcutaneous administration in the REGEN-COV group were similar to the safety findings observed with intravenous administration of REGEN-COV in COV-2067.
With repeat dosing, injection site reactions occurred in 252 subjects (35%) in the REGEN-COV group and 38 subjects (16%) in the placebo group; all injection site reactions were grade 1 or 2 in severity. Hypersensitivity reactions occurred in 8 subjects (1%) in the REGEN-COV group; and all hypersensitivity reactions were grade 1 or 2 in severity. There were no cases of anaphylaxis.
The authorized dosage for repeat dosing for post-exposure prophylaxis of COVID-19 for certain individuals who remain at high risk of exposure for longer than 4 weeks is the initial dose of 600 mg casirivimab and 600 mg imdevimab followed by 300 mg of casirivimab and 300 mg of imdevimab administered every 4 weeks [see Dosage and Administration (2.2)].
7 PATIENT MONITORING RECOMMENDATIONS
Clinically monitor patients during dose administration and observe patients for at least 1 hour after intravenous infusion or subcutaneous dosing is complete [see Warnings and Precautions (5.1) and Clinical Trials Experience (6.1)].
8 ADVERSE REACTIONS AND MEDICATION ERRORS REPORTING REQUIREMENTS AND INSTRUCTIONS
Clinical trials evaluating the safety of REGEN-COV (casirivimab and imdevimab) are ongoing [see Overall Safety Summary (6)].
Completion of an FDA MedWatch Form to report all medication errors and serious adverse events* occurring during REGEN-COV use and considered to be potentially related to REGEN-COV is mandatory and must be done by the prescribing healthcare provider and/or the provider's designee. These adverse events must be reported within 7 calendar days from the onset of the event:
*Serious Adverse Events are defined as:
- death;
- a life-threatening adverse event;
- inpatient hospitalization or prolongation of existing hospitalization;
- a persistent or significant incapacity or substantial disruption of the ability to conduct normal life functions;
- a congenital anomaly/birth defect;
- a medical or surgical intervention to prevent death, a life-threatening event, hospitalization, disability, or congenital anomaly.
If a serious and unexpected adverse event occurs and appears to be associated with the use of REGEN-COV, the prescribing health care provider and/or the provider's designee must complete and submit a MedWatch form to FDA using one of the following methods:
- Complete and submit the report online: www.fda.gov/medwatch/report.htm, or
- Complete and submit a postage-paid FDA Form 3500 (https://www.fda.gov/media/76299/download) and return by:
- Mail to MedWatch, 5600 Fishers Lane, Rockville, MD 20852-9787, or
- Fax (1-800-FDA-0178), or
- Call 1-800-FDA-1088 to request a reporting form
IMPORTANT: When reporting adverse events or medication errors to MedWatch, please complete the entire form with detailed information. It is important that the information reported to FDA be as detailed and complete as possible. Information that must be included:
- Patient demographics (e.g., patient initials, date of birth)
- Pertinent medical history
- Pertinent details regarding admission and course of illness
- Concomitant medications
- Timing of adverse event(s) in relationship to administration of REGEN-COV
- Pertinent laboratory and virology information
- Outcome of the event and any additional follow-up information if it is available at the time of the MedWatch report. Subsequent reporting of follow-up information should be completed if additional details become available.
The following steps are highlighted to provide the necessary information for safety tracking:
- In section A, box 1, provide the patient's initials in the Patient Identifier
- In section A, box 2, provide the patient's date of birth or age
- In section B, box 5, description of the event:
- a. Write "REGEN-COV use for COVID-19 under Emergency Use Authorization (EUA)" as the first line
- b. Provide a detailed report of medication error and/or adverse event. It is important to provide detailed information regarding the patient and adverse event/medication error for ongoing safety evaluation of this unapproved drug. Please see information to include listed above.
- In section G, box 1, name and address:
- a. Provide the name and contact information of the prescribing health care provider or institutional designee who is responsible for the report
- b. Provide the address of the treating institution (NOT the health care provider's office address).
9 OTHER REPORTING REQUIREMENTS
Healthcare facilities and providers must report therapeutics information and utilization data through HHS Protect, Teletracking or National Healthcare Safety Network (NHSN) as directed by the U.S. Department of Health and Human Services.
In addition, please provide a copy of all FDA MedWatch forms to:
Regeneron Pharmaceuticals, Inc
Fax: 1-888-876-2736
E-mail: medical.information@regeneron.com
Or call Regeneron Pharmaceuticals at 1-844-734-6643 to report adverse events.
10 DRUG INTERACTIONS
REGEN-COV consists of 2 monoclonal antibodies (mAbs), casirivimab and imdevimab, which are not renally excreted or metabolized by cytochrome P450 enzymes; therefore, interactions with concomitant medications that are renally excreted or that are substrates, inducers, or inhibitors of cytochrome P450 enzymes are unlikely.
11 USE IN SPECIFIC POPULATIONS
11.1 Pregnancy
Risk Summary
There are insufficient data to evaluate a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. REGEN-COV (casirivimab and imdevimab) should only be used during pregnancy if the potential benefit outweighs the potential risk for the mother and the fetus.
Nonclinical reproductive toxicity studies have not been conducted with casirivimab and imdevimab. In a tissue cross-reactivity study with casirivimab and imdevimab using human fetal tissues, no binding of clinical concern was detected. Human immunoglobulin G1 (IgG1) antibodies are known to cross the placental barrier; therefore, casirivimab and imdevimab have the potential to be transferred from the mother to the developing fetus. It is unknown whether the potential transfer of casirivimab and imdevimab provides any treatment benefit or risk to the developing fetus. There are maternal and fetal risks associated with untreated COVID-19 in pregnancy (see Clinical Considerations).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
11.2 Lactation
Risk Summary
There are no available data on the presence of casirivimab and/or imdevimab in human milk or animal milk, the effects on the breastfed infant, or the effects of the drug on milk production. Maternal IgG is known to be present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for REGEN-COV (casirivimab and imdevimab) and any potential adverse effects on the breastfed child from REGEN-COV or from the underlying maternal condition. Breastfeeding individuals with COVID-19 should follow practices according to clinical guidelines to avoid exposing the infant to COVID-19.
11.3 Pediatric Use
REGEN-COV is not authorized for use in pediatric patients under 12 years of age or weighing less than 40 kg. The safety and effectiveness of casirivimab and imdevimab are being assessed in pediatric and adolescent patients in an ongoing clinical trial. The recommended dosing regimen is expected to result in comparable serum exposures of casirivimab and imdevimab in patients 12 years of age and older and weighing at least 40 kg as observed in adults, since adults with similar body weight have been included in Trials COV-2067, COV-2069, and COV-2093.
11.4 Geriatric Use
Of the 4,567 subjects with SARS-CoV-2 infection randomized in Trial COV-2067, 14% were 65 years or older, and 4% were 75 years of age or older. Of the 3,029 subjects randomized in Trial COV-2069, 9% were 65 years or older and 2% were 75 years of age or older. Of the 974 subjects randomized in Trial COV-2093, 13% were 65 years or older and 2% were 75 years of age or older. The difference in pharmacokinetics (PK) of casirivimab and imdevimab in geriatric patients compared to younger patients is unknown [see Clinical Trial Results and Supporting Data for EUA (18.1)].
11.5 Renal Impairment
Casirivimab and imdevimab are not eliminated intact in the urine, thus renal impairment is not expected to affect the exposure of casirivimab and imdevimab.
12 OVERDOSAGE
Doses up to 8,000 mg (4,000 mg each of casirivimab and imdevimab, greater than 3 times the recommended dose) have been administered in clinical trials without dose-limiting toxicity. Treatment of overdose should consist of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. There is no specific antidote for overdose with REGEN-COV (casirivimab and imdevimab).
13 PRODUCT DESCRIPTION
Casirivimab, a human immunoglobulin G-1 (IgG1) monoclonal antibody (mAb), is a covalent heterotetramer consisting of 2 heavy chains and 2 light chains produced by recombinant DNA technology in Chinese hamster ovary (CHO) cell suspension culture and has an approximate molecular weight of 145.23 kDa.
Casirivimab injection is a sterile, preservative-free, clear to slightly opalescent and colorless to pale yellow solution in a vial for subcutaneous use or intravenous infusion after dilution available as a 300 mg/2.5 mL (120 mg/mL) or 1,332 mg/11.1 mL (120 mg/mL) solution and must be administered with imdevimab. The vial stoppers are not made with natural rubber latex.
- Casirivimab: Each 2.5 mL of solution contains 300 mg of casirivimab, L-histidine (1.9 mg), L-histidine monohydrochloride monohydrate (2.7 mg), polysorbate 80 (2.5 mg), sucrose (200 mg), and Water for Injection, USP. The pH is 6.0.
- Casirivimab: Each 11.1 mL of solution contains 1,332 mg of casirivimab, L-histidine (8.3 mg), L-histidine monohydrochloride monohydrate (12.1 mg), polysorbate 80 (11.1 mg), sucrose (888 mg), and Water for Injection, USP. The pH is 6.0.
Imdevimab, a human IgG1 mAb, is a covalent heterotetramer consisting of 2 heavy chains and 2 light chains produced by recombinant DNA technology in Chinese hamster ovary (CHO) cell suspension culture and has an approximate molecular weight of 144.14 kDa.
Imdevimab injection is a sterile, preservative-free, clear to slightly opalescent and colorless to pale yellow solution in a vial for subcutaneous use or intravenous infusion after dilution available as a 300 mg/2.5 mL (120 mg/mL) or 1,332 mg/11.1 mL (120 mg/mL) solution and must be administered with casirivimab. The vial stoppers are not made with natural rubber latex.
- Imdevimab: Each 2.5 mL of solution contains 300 mg of imdevimab, L-histidine (1.9 mg), L-histidine monohydrochloride monohydrate (2.7 mg), polysorbate 80 (2.5 mg), sucrose (200 mg), and Water for Injection, USP. The pH is 6.0.
- Imdevimab: Each 11.1 mL of solution contains 1,332 mg of imdevimab, L-histidine (8.3 mg), L-histidine monohydrochloride monohydrate (12.1 mg), polysorbate 80 (11.1 mg), sucrose (888 mg), and Water for Injection, USP. The pH is 6.0.
REGEN-COV (casirivimab and imdevimab solution) injection is a sterile, preservative-free, clear to slightly opalescent, and colorless to pale yellow 10 mL solution in a vial for intravenous infusion after dilution. The vial stoppers are not made with natural rubber latex.
- Each 10 mL of solution contains 600 mg of casirivimab, 600 mg of imdevimab, L-histidine (7.4 mg), L-histidine monohydrochloride monohydrate (10.9 mg), polysorbate 80 (10.0 mg), sucrose (800 mg), and Water for Injection, USP. The pH is 6.0.
14 CLINICAL PHARMACOLOGY
14.1 Mechanism of Action
Casirivimab (IgG1κ) and imdevimab (IgG1λ) are two recombinant human mAbs which are unmodified in the Fc regions. Casirivimab and imdevimab bind to non-overlapping epitopes of the spike protein receptor binding domain (RBD) of SARS-CoV-2 with dissociation constants KD = 45.8 pM and 46.7 pM, respectively. Casirivimab, imdevimab and casirivimab and imdevimab together blocked RBD binding to the human ACE2 receptor with IC50 values of 56.4 pM, 165 pM and 81.8 pM, respectively and prevents viral attachment to host cells [see Microbiology/Resistance Information (15)].
14.2 Pharmacodynamics
Trial COV-2067 evaluated REGEN-COV (casirivimab and imdevimab) with doses of up to 6.66 times the recommended dose (600 mg of casirivimab and 600 mg of imdevimab; 1,200 mg of casirivimab and 1,200 mg of imdevimab; 4,000 mg of casirivimab and 4,000 mg of imdevimab) in ambulatory patients with COVID-19. A flat dose-response relationship for efficacy was identified for REGEN-COV at all doses, based on viral load and clinical outcomes. Similar reductions in viral load (log10 copies/mL) were observed in subjects for the (600 mg of casirivimab and 600 mg of imdevimab) intravenous and (600 mg of casirivimab and 600 mg of imdevimab) subcutaneous doses; however, only limited clinical outcome data are available for the subcutaneous route of administration for the treatment of symptomatic patients.
14.3 Pharmacokinetics
Both casirivimab and imdevimab exhibited linear and dose-proportional pharmacokinetics (PK) between (600 mg of casirivimab and 600 mg of imdevimab) to (4,000 mg of casirivimab and 4,000 mg of imdevimab) doses of REGEN-COV (casirivimab and imdevimab) following intravenous administration of single dose. A summary of PK parameters after a single (600 mg of casirivimab and 600 mg of imdevimab) intravenous dose, for each antibody is provided in Table 7.
| PK Parameter* | Casirivimab | Imdevimab |
|---|---|---|
|
|
||
| Ceoi (mg/L)† | 192 (80.9) | 198 (84.8) |
| C28 (mg/L)‡ | 46.2 (22.3) | 38.5 (19.7) |
A summary of PK parameters after a single 600 mg of casirivimab and 600 mg of imdevimab subcutaneous dose is shown in Table 8.
| PK Parameter*,† | Casirivimab | Imdevimab |
|---|---|---|
|
|
||
| Cmax (mg/L) | 55.6 (22.2) | 52.7 (22.5) |
| tmax (day)‡ | 8.00 (4.00, 87.0) | 7.00 (4.00, 15.0) |
| AUC0-28 (mg∙day/L) | 1060 (363) | 950 (362) |
| AUCinf (mg∙day/L)§ | 2580 (1349) | 1990 (1141) |
| C28 (mg/L)¶ | 30.7 (11.9) | 24.8 (9.58) |
| Half-life (day) | 31.8 (8.35) | 26.9 (6.80) |
For the repeat dose prophylaxis intravenous and subcutaneous regimens, population pharmacokinetic simulations predicted that trough concentrations in serum at steady-state after an initial 600 mg casirivimab and 600 mg imdevimab intravenous or subcutaneous dose followed by monthly (every 4 weeks) 300 mg casirivimab and 300 mg imdevimab intravenous or subcutaneous doses are similar to slightly higher than observed mean day 29 concentrations in serum for a single 600 mg casirivimab and 600 mg imdevimab subcutaneous dose.
Specific Populations
The effect of different covariates (e.g., age, sex, race, body weight, disease severity, hepatic impairment) on the PK of casirivimab and imdevimab is unknown. Renal impairment is not expected to impact the PK of casirivimab and imdevimab, since mAbs with molecular weight >69 kDa are known not to undergo renal elimination. Similarly, dialysis is not expected to impact the PK of casirivimab and imdevimab.
Drug-Drug Interactions
Casirivimab and imdevimab are mAbs which are not renally excreted or metabolized by cytochrome P450 enzymes; therefore, interactions with concomitant medications that are renally excreted or that are substrates, inducers, or inhibitors of cytochrome P450 enzymes are unlikely [see Drug Interactions (10)].
15 MICROBIOLOGY/RESISTANCE INFORMATION
Antiviral Activity
In a SARS-CoV-2 virus neutralization assay in Vero E6 cells, casirivimab, imdevimab, and casirivimab and imdevimab together neutralized SARS-CoV-2 (USA-WA1/2020 isolate) with EC50 values of 37.4 pM (0.006 μg/mL), 42.1 pM (0.006 μg/mL), and 31.0 pM (0.005 μg/mL), respectively.
Antibody-dependent cell-mediated cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP) were assessed using Jurkat target cells expressing SARS-CoV-2 spike protein. Casirivimab, imdevimab and casirivimab and imdevimab together mediated ADCC with human natural killer (NK) effector cells. Casirivimab, imdevimab and casirivimab and imdevimab together mediated ADCP with human macrophages. Casirivimab, imdevimab and casirivimab and imdevimab together did not mediate complement-dependent cytotoxicity in cell-based assays.
Antibody Dependent Enhancement (ADE) of Infection
The potential of casirivimab and of imdevimab to mediate viral entry was assessed in immune cell lines co-incubated with recombinant vesicular stomatitis virus (VSV) virus-like particles (VLP) pseudotyped with SARS-CoV-2 spike protein at concentrations of mAb(s) down to approximately 10-fold below the respective neutralization EC50 values. Casirivimab and imdevimab together and imdevimab alone, but not casirivimab alone, mediated entry of pseudotyped VLP into FcγR2+ Raji and FcγR1+/FcγR2+ THP1 cells (maximum infection in total cells of 1.34% and 0.24%, respectively, for imdevimab; 0.69% and 0.06%, respectively for casirivimab and imdevimab together), but not any other cell lines tested (IM9, K562, Ramos and U937 cells).
Antiviral Resistance
There is a potential risk of treatment failure due to the development of viral variants that are resistant to casirivimab and imdevimab administered together. Prescribing healthcare providers should consider the prevalence of SARS-CoV-2 variants in their area, where data are available, when considering treatment options.
Escape variants were identified following two passages in cell culture of recombinant VSV encoding SARS-CoV-2 spike protein in the presence of casirivimab or imdevimab individually, but not following two passages in the presence of casirivimab and imdevimab together. Variants which showed reduced susceptibility to casirivimab alone included those with spike protein amino acid substitutions K417E (182-fold), K417N (7-fold), K417R (61-fold), Y453F (>438-fold), L455F (80-fold), E484K (25-fold), F486V (>438-fold) and Q493K (>438-fold). Variants which showed reduced susceptibility to imdevimab alone included substitutions K444N (>755-fold), K444Q (>548-fold), K444T (>1,033-fold), and V445A (>548-fold). Casirivimab and imdevimab together showed reduced susceptibility to variants with K444T (6-fold) and V445A (5-fold) substitutions.
In neutralization assays using VSV VLP pseudotyped with spike protein variants identified in circulating SARS-CoV-2, variants with reduced susceptibility to casirivimab alone included those with E406D (51-fold), G476S (5-fold), E484Q (19-fold), G485D (5-fold), F486L (61-fold), F486S (>715-fold), Q493E (446-fold), Q493R (77-fold), and S494P (5-fold) substitutions, and variants with reduced susceptibility to imdevimab alone included those with P337L (5-fold), N439K (463-fold), N439V (4-fold), N440K (28-fold), K444L (153-fold), K444M (1,577-fold), G446V (135-fold), N450D (9-fold), Q498H (17-fold), P499S (206-fold) substitutions. The G476D substitution had an impact (4-fold) on casirivimab and imdevimab together. Substitutions tested concurrently which had reduced susceptibility to casirivimab and imdevimab together included N440K+E484K (21-fold), found in the B.1.619/B.1.625 lineages, and N439K+E484K (23-fold), found in the AV.1 lineage; variants harboring these concurrent substitutions have been detected rarely in the US.
Casirivimab and imdevimab individually and together retained neutralization activity against pseudotyped VLP expressing all spike protein substitutions found in the B.1.1.7 lineage (Alpha; UK origin) and against pseudotyped VLP expressing only N501Y found in B.1.1.7 and other circulating lineages (Table 9). Casirivimab and imdevimab together retained neutralization activity against pseudotyped VLP expressing all spike protein substitutions, or individual substitutions K417N, E484K or N501Y, found in the B.1.351 lineage (Beta; South Africa origin), and all spike protein substitutions or key substitutions K417T, E484K, or N501Y, found in the P.1 lineage (Gamma; Brazil origin), although casirivimab alone, but not imdevimab, had reduced activity against pseudotyped VLP expressing K417N (7-fold) or E484K (25-fold). The E484K substitution is also found in the B.1.526 lineage (Iota; USA [New York] origin). Casirivimab and imdevimab, individually and together, retained neutralization activity against the L452R substitution found in the B.1.427/B.1.429 lineages (Epsilon; USA [California] origin).
Casirivimab and imdevimab, individually and together, retained neutralization activity against pseudotyped VLP expressing L452R+T478K substitutions found in the B.1.617.2 and AY.3 lineages (Delta; India origin). Casirivimab and imdevimab together retained neutralization activity against pseudotyped VLP expressing L452R+E484Q substitutions, found in the B.1.617.1/B.1.617.3 lineages (Kappa/no designation; India origin), although casirivimab alone, but not imdevimab, had reduced activity against pseudotyped VLP expressing L452R+E484Q (7-fold). Casirivimab and imdevimab together retained activity against pseudotyped VLP expressing R346K+E484K+N501Y found in the B.1.621/B.1.621.1 (Mu; Colombia origin) lineage although casirivimab alone, but not imdevimab, had reduced activity against pseudotyped VLP expressing R346K+E484K+N501Y (23-fold).
Casirivimab and imdevimab, individually (>1732-fold and >754-fold, respectively) and together (>1013-fold), demonstrated reduced neutralization activity against VLP pseudotyped with the full spike protein sequence of the B.1.1.529/BA.1 (Omicron; South Africa origin) lineage.
| Lineage with Spike Protein Substitution | Country First Identified | WHO Nomenclature | Key Substitutions | Fold Reduction in Susceptibility |
|---|---|---|---|---|
| Abbreviations: del, deletion; ins, insertion | ||||
|
|
||||
| B.1.1.7 | UK | Alpha | N501Y* | no change† |
| B.1.351 | South Africa | Beta | K417N+E484K+N501Y‡ | no change† |
| P.1 | Brazil | Gamma | K417T+E484K+N501Y§ | no change† |
| B.1.617.2/AY.3 | India | Delta | L452R+T478K | no change† |
| B.1.427/B.1.429 | USA (California) | Epsilon | L452R | no change† |
| B.1.526¶ | USA (New York) | Iota | E484K | no change† |
| B.1.617.1/B.1.617.3 | India | Kappa/no designation | L452R+E484Q | no change† |
| B.1.621/B.1.621.1 | Colombia | Mu | R346K+E484K+N501Y | no change† |
| B.1.1.529/BA.1 | South Africa | Omicron | G339D+S371L+S373P+S375F+K417N+N440K, G446S+S477N+T478K+E484A+Q493R+G496S+Q498R+N501Y+Y505H# | >1013-foldÞ |
Due to the large reduction of pseudotyped VLP neutralization activity against spike protein from the B.1.1.529/BA.1 (Omicron;South Africa origin) variant, it is unlikely that casirivimab and imdevimab together will be active against this variant.
Casirivimab and imdevimab together retained activity against authentic SARS-CoV-2 variants of B.1.1.7 (Alpha), B.1.351 (Beta), P.1 (Gamma), B.1.617.1 (Kappa) lineages (Table 10), although casirivimab alone, but not imdevimab, had reduced activity against B.1.351 (5-fold), P.1 (>371-fold) and B.1.617.1 (6-fold) variants.
It is not known how pseudotyped VLP or authentic SARS-CoV-2 data correlate with clinical outcomes.
| SARS-CoV-2 Lineage | Country First Identified | WHO Nomenclature | Key Substitutions* | Fold Reduction in Susceptibility |
|---|---|---|---|---|
|
|
||||
| B.1.1.7 | UK | Alpha | N501Y | no change† |
| B.1.351 | South Africa | Beta | K417N+E484K+N501Y | no change† |
| P.1 | Brazil | Gamma | K417T+E484K+N501Y | no change† |
| B.1.617.2 | India | Delta | L452R+T478K | no change† |
| B.1.617.1 | India | Kappa | L452R+E484Q | no change† |
In clinical trial COV-2067, interim data indicated only one variant (G446V) occurring at an allele fraction ≥15%, which was detected in 3/66 subjects who had nucleotide sequencing data, each at a single time point (two at baseline in subjects from placebo and 2,400 mg casirivimab and imdevimab groups, and one at Day 25 in a subject from the 8,000 mg casirivimab and imdevimab group). The G446V variant had reduced susceptibility to imdevimab of 135-fold compared to wild-type in a pseudotyped VSV VLP neutralization assay but retained susceptibility to casirivimab alone and casirivimab and imdevimab together.
It is possible that resistance-associated variants to casirivimab and imdevimab together could have cross-resistance to other mAbs targeting the receptor binding domain of SARS-CoV-2. The clinical impact is not known.
16 NONCLINICAL TOXICOLOGY
Carcinogenicity, genotoxicity, and reproductive toxicology studies have not been conducted with casirivimab and imdevimab.
In a toxicology study in cynomolgus monkeys, casirivimab and imdevimab had no adverse effects when administered intravenously or subcutaneously. Non-adverse liver findings (minor transient increases in AST and ALT) were observed.
In tissue cross-reactivity studies with casirivimab and imdevimab using human adult and fetal tissues, no binding of clinical concern was detected.
17 ANIMAL PHARMACOLOGIC AND EFFICACY DATA
Casirivimab and imdevimab administered together has been assessed in rhesus macaque and Syrian golden hamster treatment models of SARS-CoV-2 infection. Therapeutic administration of casirivimab and imdevimab together at 25 mg/kg or 150 mg/kg into rhesus macaques (n=4 for each dosing group) 1-day post infection resulted in approximately 1-2 log10 reductions in genomic and sub-genomic viral RNA in nasopharyngeal swabs and oral swabs at Day 4 post-challenge in most animals, and reduced lung pathology relative to placebo-treated animals. Therapeutic administration of casirivimab and imdevimab together at 5 mg/kg and 50 mg/kg doses to hamsters 1-day post infection resulted in reduced weight loss relative to placebo treated animals. In the prophylactic setting in rhesus macaques, administration of 50 mg/kg casirivimab and imdevimab together prior to challenge with SARS-CoV-2 demonstrated reduction in viral RNA via nasopharyngeal, oral swabs and bronchioalveolar lavage fluid, as well as a reduction in lung inflammation. In the prophylactic setting in hamsters, administration of 0.5 mg/kg, 5 mg/kg, or 50 mg/kg casirivimab and imdevimab together prior to challenge with SARS-CoV-2 protected against weight loss, and reduced percentage of lung area showing pneumonia pathology and severity of lung inflammation, indicative of reduced morbidity in this model. The applicability of these findings to a clinical setting is not known.
18 CLINICAL TRIAL RESULTS AND SUPPORTING DATA FOR EUA
18.1 Treatment of Mild to Moderate COVID-19 (COV-2067)
The data supporting this EUA are based on the analysis of Phase 1/2/3 from trial, COV-2067 (NCT04425629). This is a randomized, double-blinded, placebo-controlled clinical trial evaluating REGEN-COV (casirivimab and imdevimab) for the treatment of subjects with mild to moderate COVID-19 (subjects with COVID-19 symptoms who are not hospitalized). Cohort 1 enrolled adult subjects who were not hospitalized and had 1 or more COVID-19 symptoms that were at least mild in severity. Treatment was initiated within 3 days of obtaining a positive SARS-CoV-2 viral infection determination. Subjects in the Phase 3 primary efficacy analysis met the criteria for high risk for progression to severe COVID-19, as shown in Section 2.
In the Phase 3 trial, 4,567 subjects with at least one risk factor for severe COVID-19 were randomized to a single intravenous infusion of 600 mg of casirivimab and 600 mg of imdevimab (n=838), 1,200 mg of casirivimab and 1,200 mg of imdevimab (n=1,529), 4,000 mg of casirivimab and 4,000 mg of imdevimab (n=700), or placebo (n=1,500) groups. The two REGEN-COV doses at the start of Phase 3 were 4,000 mg and 1,200 mg of each component; however, based on Phase 1/2 efficacy analyses showing that the 4,000 mg and 1,200 mg doses of each component were similar, the Phase 3 portion of the protocol was amended to compare 1,200 mg dose of each component vs. placebo and 600 mg dose of each component vs. placebo. Comparisons were between subjects randomized to the specific REGEN-COV dose and subjects who were concurrently randomized to placebo.
At baseline, in all randomized subjects with at least one risk factor, the median age was 50 years (with 13% of subjects ages 65 years or older), 52% of the subjects were female, 84% were White, 36% were Hispanic or Latino, and 5% were Black or African American. In subjects with available baseline symptom data, 15% had mild symptoms, 42% had moderate, 42% had severe symptoms, and 2% reported no symptoms at baseline; the median duration of symptoms was 3 days; mean viral load was 6.2 log10 copies/mL at baseline. The baseline demographics and disease characteristics were well balanced across the casirivimab and imdevimab and placebo treatment groups.
The primary endpoint was the proportion of subjects with ≥1 COVID-19-related hospitalization or all-cause death through Day 29, in subjects with a positive SARS-CoV-2 RT-qPCR result from nasopharyngeal (NP) swab at randomization, and with at least one risk factor for severe COVID-19, i.e., the modified full analysis set (mFAS). In the mFAS, events (COVID-19-related hospitalization or all-cause death through Day 29) occurred in 7 (1.0%) subjects treated with 600 mg of casirivimab and 600 mg of imdevimab compared to 24 (3%) subjects concurrently randomized to placebo, demonstrating a 70% reduction in COVID-19-related hospitalization or all-cause death compared to placebo (p=0.0024). Events occurred in 18 (1.3%) subjects treated with 1,200 mg of casirivimab and 1,200 mg of imdevimab compared to 62 (5%) subjects concurrently randomized to placebo, demonstrating a 71% reduction compared to placebo (REGEN-COV 1% vs placebo 5%, p<0.0001). In the 1,200 mg analysis, there was 1 death each in the REGEN-COV and placebo arm (p=1.0); and in 2,400 mg analysis, there were 1 and 3 deaths, respectively, in the REGEN-COV and placebo arms (p=0.3721). Overall, similar effects were observed for 600 mg of casirivimab and 600 mg of imdevimab and 1,200 mg of casirivimab and 1,200 mg of imdevimab doses, indicating the absence of a dose effect; therefore the 600 mg of casirivimab and 600 mg of imdevimab dose is authorized and the 1,200 mg of casirivimab and 1,200 mg of imdevimab dose is no longer authorized under this EUA (See Table 11). Results were consistent across subgroups of patients defined by nasopharyngeal viral load >106 copies/mL at baseline or serologic status.
| 600 mg of casirivimab and 600 mg of imdevimab (intravenous) | Placebo | 1,200 mg of casirivimab and 1,200 mg of imdevimab (intravenous) | Placebo | |
|---|---|---|---|---|
| n=736 | n=748 | n=1,355 | n=1,341 | |
| # of subjects with at least 1 event (COVID-19-related hospitalization or all-cause death) | 7 (1.0%) | 24 (3.2%) | 18 (1.3%) | 62 (4.6%) |
| Risk reduction | 70% (p=0.0024) | 71% (p<0.0001) |
||
Treatment with REGEN-COV resulted in a statistically significant reduction in the LS mean viral load (log10 copies/mL) from baseline to Day 7 compared to placebo (-0.71 log10 copies/mL for 600 mg dose of casirivimab and 600 mg of imdevimab and -0.86 log10 copies/mL for 2,400 mg; p<0.0001). Reductions were observed in the overall mFAS population and in other subgroups, including those with baseline viral load >106 copies/mL or who were seronegative at baseline. Consistent effects were observed for the individual doses, indicating the absence of a dose effect. Figure 1 shows the mean change from baseline in SARS-COV-2 viral load to Day 15.
Figure 1: Change from Baseline in SARS-COV-2 Viral Load (log10 copies/mL) to Day 15 (COV-2067)
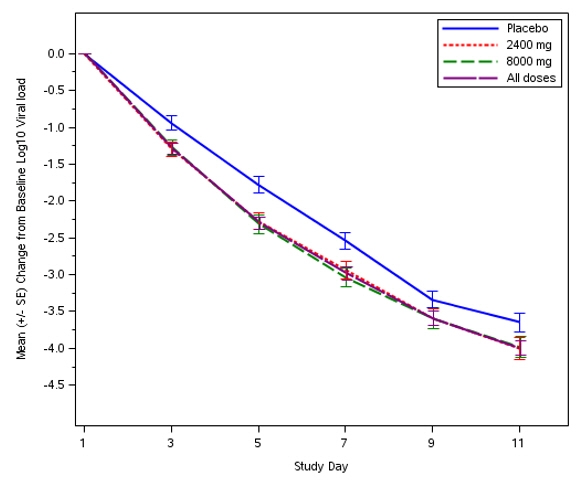
REGEN-COV 1.2 g IV = 600 mg of casirivimab and 600 mg of imdevimab administered intravenously
REGEN-COV 2.4 g IV = 1,200 mg of casirivimab and 1,200 mg of imdevimab administered intravenously
The median time to symptom resolution, as recorded in a trial-specific daily symptom diary, was 10 days for REGEN-COV-treated subjects, as compared with 14 days for placebo-treated subjects (p=0.0001 for 600 mg of casirivimab and 600 mg of imdevimab vs. placebo; p<0.0001 for 1,200 mg of casirivimab and 1,200 mg of imdevimab vs. placebo). Symptoms assessed were fever, chills, sore throat, cough, shortness of breath/difficulty breathing, nausea, vomiting, diarrhea, headache, red/watery eyes, body aches, loss of taste/smell, fatigue, loss of appetite, confusion, dizziness, pressure/tight chest, chest pain, stomachache, rash, sneezing, sputum/phlegm, runny nose. Time to COVID-19 symptom resolution was defined as time from randomization to the first day during which the subject scored 'no symptom' (score of 0) on all of the above symptoms except cough, fatigue, and headache, which could have been 'mild/moderate symptom' (score of 1) or 'no symptom' (score of 0).
18.2 Post-exposure Prophylaxis of COVID-19 (COV-2069)
The data supporting this EUA for post-exposure prophylaxis of COVID-19 are based on the efficacy analysis of data from the Phase 3 COV-2069 trial (NCT04452318). This is a randomized, double-blind, placebo-controlled clinical trial studying REGEN-COV (casirivimab and imdevimab) for post-exposure prophylaxis of COVID-19 in household contacts of individuals infected with SARS-CoV-2 (index case).
The trial enrolled subjects who were asymptomatic and who lived in the same household with a SARS-CoV-2 infected patient. Subjects were randomized 1:1 to a single dose of 600 mg of casirivimab and 600 mg of imdevimab or placebo administered subcutaneously within 96 hours of collection of the index cases' positive SARS-CoV-2 diagnostic test sample.
Subjects with a negative SARS-CoV-2 RT-qPCR test result at baseline (n=2,067) were enrolled and randomized in Cohort A. The primary analysis population included subjects who were SARS-CoV-2 RT-qPCR negative and seronegative at baseline. Of the 1,505 subjects in the primary analysis population, 753 subjects were randomized to receive REGEN-COV and 752 subjects were randomized to placebo. Following randomization and dosing, subjects had SARS-CoV-2 RT-qPCR testing via a nasopharyngeal swab every 7 days as well as weekly interviews with the investigator for assessment of COVID-19 symptoms during the 28-day efficacy assessment period. No data were collected on the type or extent of exposure to the index case.
For the primary analysis population at baseline, the median age was 44 years (with 9% of subjects ages 65 years or older), 54% of the subjects were female, 86% were White, 41% were Hispanic or Latino, and 9% were Black. The baseline demographics and disease characteristics were well balanced across the casirivimab and imdevimab and placebo treatment groups.
The primary efficacy endpoint was the proportion of subjects who developed RT qPCR-confirmed COVID-19 through Day 29. In the primary analysis population (RT-qPCR negative and seronegative at baseline), there was an 81% risk reduction in the development of COVID-19 with REGEN-COV treatment versus placebo [11/753 (1%) and 59/752 (8%); adjusted odds ratio 0.17; p<0.0001]. Figure 2 shows the cumulative incidence of COVID-19 through Day 29. Similar results were obtained in a sensitivity analysis that included RT-qPCR negative subjects at baseline, regardless of baseline serological status, where there was an 82% risk reduction in RT-qPCR-confirmed COVID-19 with REGEN-COV treatment versus placebo. There was a 66% risk reduction in the proportion of participants with any RT-qPCR-confirmed SARS-CoV-2 infection (symptomatic or asymptomatic) with REGEN-COV treatment versus placebo [36/753 (5%) and 107/752 (14%); adjusted odds ratio 0.31; p<0.0001].
Figure 2: Cumulative Incidence of Symptomatic COVID-19 (COV-2069 Cohort A)

In a post-hoc analysis in the subgroup of subjects who met the criteria for high risk for progression to severe COVID-19 (as shown in Section 2), there was a 76% risk reduction in COVID-19 with REGEN-COV treatment versus placebo [10/570 (2%) vs 42/567 (7%); adjusted odds ratio 0.22; p<0.0001].
In Cohort B, asymptomatic subjects with a positive SARS-CoV-2 RT-qPCR test result at baseline (n=311) were enrolled and randomized 1:1 to REGEN-COV or placebo. In a post-hoc analysis of the overall combined Cohort A and Cohort B (regardless of serology status at baseline), there was a 62% risk reduction in COVID-19 with REGEN-COV treatment versus placebo [46/1201 (4%) vs 119/1177 (10%); adjusted odds ratio 0.35; p<0.0001].
19 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Co-formulated casirivimab and imdevimab injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution supplied in a vial. Refer to Table 12.
Casirivimab injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution supplied in a vial. Refer to Table 13.
Imdevimab injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution supplied in a vial. Refer to Table 13.
REGEN-COV (casirivimab and imdevimab) injection is available as:
- 1.
A single vial which contains two antibodies co-formulated in a 1:1 ratio of casirivimab and imdevimab (Table 12).
- 2. Individual antibody solutions in separate vials, which may be supplied in separate cartons (Table 13) or together in a single carton (also referred to as a co-packaged carton) (Table 14), or in a dose pack (Table 15).
| Antibody | Concentration | Package Size | NDC Number |
|---|---|---|---|
| REGEN-COV (casirivimab and imdevimab) | 600 mg/600 mg per 10 mL (60 mg/60 mg per mL) | 1 vial per carton | 61755-039-01 |
INDIVIDUAL CASIRIVIMAB AND IMDEVIMAB SOLUTIONS MUST BE ADMINISTERED TOGETHER.
| Antibody | Concentration | Package Size | NDC Number |
|---|---|---|---|
| Casirivimab REGN10933 | 1,332 mg/11.1 mL (120 mg/mL) | 1 vial per carton | 61755-024-01 |
| 300 mg/2.5 mL (120 mg/mL) | 1 vial per carton | 61755-026-01 | |
| Imdevimab REGN10987 | 1,332 mg/11.1 mL (120 mg/mL) | 1 vial per carton | 61755-025-01 |
| 300 mg/2.5 mL (120 mg/mL) | 1 vial per carton | 61755-027-01 |
Each co-packaged carton contains 1 vial of casirivimab and 1 vial of imdevimab. Refer to Table 14.
| Co-Packaged Carton Contents | Co-Packaged Components | Concentration | Co-Packaged Carton NDC Number |
|---|---|---|---|
| 2 Vials | 1 vial of casirivimab
(NDC: 61755-024-00) | 1,332 mg/11.1 mL (120 mg/mL) | 61755-042-02 |
| 1 vial of imdevimab
(NDC: 61755-025-00) | 1,332 mg/11.1 mL (120 mg/mL) |
||
| 2 Vials | 1 vial of casirivimab
(NDC: 61755-026-00) | 300 mg/2.5 mL (120 mg/mL) | 61755-045-02 |
| 1 vial of imdevimab
(NDC: 61755-027-00) | 300 mg/2.5 mL (120 mg/mL) |
Each REGEN-COV dose pack contains sufficient number of vials of casirivimab [REGN10933] and imdevimab [REGN10987] to prepare up to two treatment doses (600 mg of casirivimab and 600 mg of imdevimab). Refer to Table 15.
| REGEN-COV Dose Pack Size | REGEN-COV Dose Pack Components | Concentration | REGEN-COV Dose Pack NDC Number |
|---|---|---|---|
| 2 Cartons | 1 vial of casirivimab REGN10933 (NDC: 61755-024-01) | 1,332 mg/11.1 mL (120 mg/mL) | 61755-035-02 |
| 1 vial of imdevimab REGN10987 (NDC: 61755-025-01) | 1,332 mg/11.1 mL (120 mg/mL) |
||
| 8 Cartons | 4 vials of casirivimab REGN10933 (NDC: 61755-026-01) | 300 mg/2.5 mL (120 mg/mL) | 61755-036-08 |
| 4 vials of imdevimab REGN10987 (NDC: 61755-027-01) | 300 mg/2.5 mL (120 mg/mL) |
||
| 5 Cartons | 1 vial of casirivimab REGN10933 (NDC: 61755-024-01) | 1,332 mg/11.1 mL (120 mg/mL) | 61755-037-05 |
| 4 vials of imdevimab REGN10987 (NDC: 61755-027-01) | 300 mg/2.5 mL (120 mg/mL) |
||
| 5 Cartons | 4 vials of casirivimab REGN10933 (NDC: 61755-026-01) | 300 mg/2.5 mL (120 mg/mL) | 61755-038-05 |
| 1 vial of imdevimab REGN10987 (NDC: 61755-025-01) | 1,332 mg/11.1 mL (120 mg/mL) |
Storage and Handling
Casirivimab is preservative-free. Discard any unused portion.
Imdevimab is preservative-free. Discard any unused portion.
Store unopened casirivimab and imdevimab vials in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Unopened vials may be stored in the original carton at room temperature [up to 25°C (77°F)] and must be used within 30 days. If not used in the 30 days, discard vials.
DO NOT FREEZE. DO NOT SHAKE. DO NOT EXPOSE TO DIRECT LIGHT.
Solution in vial requires dilution prior to intravenous administration. The prepared infusion solution is intended to be used immediately. If immediate administration is not possible, store diluted casirivimab and imdevimab infusion solution in the refrigerator at 2°C to 8°C (36°F to 46°F) for no more than 36 hours or at room temperature up to 25°C (77°F) for no more than 4 hours. If refrigerated, allow the infusion solution to equilibrate to room temperature for approximately 30 minutes prior to administration.
The prepared syringes should be administered immediately. If immediate administration is not possible, store the prepared casirivimab and imdevimab syringes in the refrigerator between 2ºC to 8ºC (36ºF to 46ºF) for no more than 24 hours, or at room temperature up to 25ºC (77ºF) for no more than 8 hours. If refrigerated, allow the syringes to equilibrate to room temperature for approximately 20 minutes prior to administration.
20 PATIENT COUNSELING INFORMATION
Patients treated with REGEN-COV (casirivimab and imdevimab) should continue to self-isolate and use infection control measures (e.g., wear mask, isolate, social distance, avoid sharing personal items, clean and disinfect "high touch" surfaces, and frequent handwashing) according to CDC guidelines. Also see Fact Sheet for Patients, Parents and Caregivers.
21 CONTACT INFORMATION
For additional information visit www.REGENCOV.com
If you have questions, please contact Regeneron at 1-844-734-6643.
Manufactured by:
Regeneron Pharmaceuticals, Inc.
777 Old Saw Mill River Road
Tarrytown, NY 10591-6707
©2021 Regeneron Pharmaceuticals, Inc. All rights reserved.
Revised: 12/2021
FACT SHEET FOR PATIENTS, PARENTS AND CAREGIVERS
EMERGENCY USE AUTHORIZATION (EUA) OF REGEN-COV™ (casirivimab and imdevimab) FOR CORONAVIRUS DISEASE 2019 (COVID-19)
You are being given a medicine called REGEN-COV (casirivimab and imdevimab) for the treatment or post-exposure prevention of coronavirus disease 2019 (COVID-19). SARS-CoV-2 is the virus that causes COVID-19. This Fact Sheet contains information to help you understand the potential risks and potential benefits of taking REGEN-COV.
Receiving REGEN-COV may benefit certain people with COVID-19 and may help prevent certain people who have been exposed to someone who is infected with SARS-CoV-2 from getting SARS-CoV-2 infection, or may prevent certain people who are at high risk of exposure to someone who is infected with SARS-CoV-2 from getting SARS-CoV-2 infection.
Read this Fact Sheet for information about REGEN-COV. Talk to your healthcare provider if you have questions. It is your choice to receive REGEN-COV or stop at any time.
WHAT IS COVID-19?
COVID-19 is caused by a virus called a coronavirus, SARS-CoV-2. People can get COVID-19 through contact with another person who has the virus.
COVID-19 illnesses have ranged from very mild (including some with no reported symptoms) to severe, including illness resulting in death. While information so far suggests that most COVID-19 illness is mild, serious illness can happen and may cause some of your other medical conditions to become worse. People of all ages with severe, long-lasting (chronic) medical conditions like heart disease, lung disease, and diabetes, for example, and other conditions including obesity, seem to be at higher risk of being hospitalized for COVID-19. Older age, with or without other conditions, also places people at higher risk of being hospitalized for COVID-19.
WHAT ARE THE SYMPTOMS OF COVID-19?
The symptoms of COVID-19 include fever, cough, and shortness of breath, which may appear 2 to 14 days after exposure. Serious illness including breathing problems can occur and may cause your other medical conditions to become worse.
WHAT IS REGEN-COV (casirivimab and imdevimab)?
REGEN-COV is an investigational medicine used in adults and adolescents (12 years of age and older who weigh at least 88 pounds (40 kg)) who are at high risk for severe COVID-19, including hospitalization or death for:
- treatment of mild to moderate symptoms of COVID-19
- post-exposure prevention of COVID-19 in persons who are:
- not fully vaccinated against COVID-19 (Individuals are considered to be fully vaccinated 2 weeks after their second vaccine dose in a 2-dose series [such as the Pfizer or Moderna vaccines], or 2 weeks after a single-dose vaccine [such as Johnson & Johnson's Janssen vaccine]), or,
- are not expected to build up enough of an immune response to the complete COVID-19 vaccination (for example, someone with immunocompromising conditions, including someone who is taking immunosuppressive medications), and
- have been exposed to someone who is infected with SARS-CoV-2. Close contact with someone who is infected with SARS-CoV-2 is defined as being within 6 feet for a total of 15 minutes or more, providing care at home to someone who is sick, having direct physical contact with the person (hugging or kissing, for example), sharing eating or drinking utensils, or being exposed to respiratory droplets from an infected person (sneezing or coughing, for example). For additional details, go to https://www.cdc.gov/coronavirus/2019-ncov/if-you-are-sick/quarantine.html, or
- someone who is at high risk of being exposed to someone who is infected with SARS-CoV-2 because of occurrence of SARS-CoV-2 infection in other individuals in the same institutional setting (for example, nursing homes, prisons).
REGEN-COV is investigational because it is still being studied. There is limited information known about the safety and effectiveness of using REGEN-COV to treat people with COVID-19 or to prevent COVID-19 in people who are at high risk of being exposed to someone who is infected with SARS-CoV-2. REGEN-COV is not authorized for pre-exposure prophylaxis for prevention of COVID-19.
The FDA has authorized the emergency use of REGEN-COV for the treatment of COVID-19 and the post-exposure prevention of COVID-19 under an Emergency Use Authorization (EUA). For more information on EUA, see the "What is an Emergency Use Authorization (EUA)?" section at the end of this Fact Sheet.
WHO SHOULD NOT TAKE REGEN-COV?
Do not take REGEN-COV if you have had a severe allergic reaction to REGEN-COV.
WHAT SHOULD I TELL MY HEALTH CARE PROVIDER BEFORE I RECEIVE REGEN-COV?
Tell your healthcare provider about all of your medical conditions, including if you:
- Have any allergies
- Have had a severe allergic reaction including anaphylaxis to REGEN-COV previously
- Have received a COVID-19 vaccine
- Have any serious illnesses
- Are pregnant or plan to become pregnant
- Are breastfeeding or plan to breastfeed
- Are taking any medications (prescription, over-the-counter, vitamins, and herbal products)
HOW WILL I RECEIVE REGEN-COV (casirivimab and imdevimab)?
- REGEN-COV consists of two investigational medicines, casirivimab and imdevimab, given together at the same time through a vein (intravenous or IV) or injected in the tissue just under the skin (subcutaneous injections). Your healthcare provider will determine the most appropriate way for you to be given REGEN-COV.
- Treatment: If you are receiving an intravenous infusion, the infusion will take 20 to 50 minutes or longer. Your healthcare provider will determine the duration of your infusion.
- If your healthcare provider determines that you are unable to receive REGEN-COV as an intravenous infusion which would lead to a delay in treatment, then as an alternative, REGEN-COV can be given in the form of subcutaneous injections. If you are receiving subcutaneous injections, your dose will be provided as multiple injections given in separate locations around the same time.
- Post-exposure prevention: If you are receiving subcutaneous injections, your dose will be provided as multiple injections given in separate locations around the same time. If you are receiving an intravenous infusion, the infusion will take 20 to 50 minutes or longer.
- After the initial dose, if your healthcare provider determines that you need to receive additional doses of REGEN-COV for ongoing protection, the additional intravenous or subcutaneous doses would be administered monthly.
WHAT ARE THE IMPORTANT POSSIBLE SIDE EFFECTS OF REGEN-COV (casirivimab and imdevimab)?
Possible side effects of REGEN-COV are:
- Allergic reactions. Allergic reactions can happen during and after infusion or injection of REGEN-COV. Tell your healthcare provider right away or seek immediate medical attention if you get any of the following signs and symptoms of allergic reactions: fever, chills, nausea, headache, shortness of breath, low or high blood pressure, rapid or slow heart rate, chest discomfort or pain, weakness, confusion, feeling tired, wheezing, swelling of your lips, face, or throat, rash including hives, itching, muscle aches, feeling faint, dizziness and sweating. These reactions may be severe or life threatening.
- Worsening symptoms after treatment: You may experience new or worsening symptoms after infusion or injection, including fever, difficulty breathing, rapid or slow heart rate, tiredness, weakness or confusion. If these symptoms occur, contact your healthcare provider or seek immediate medical attention as some of these symptoms have required hospitalization. It is unknown if these symptoms are related to treatment or are due to the progression of COVID-19.
The side effects of getting any medicine by vein may include brief pain, bleeding, bruising of the skin, soreness, swelling, and possible infection at the infusion site. The side effects of getting any medicine by subcutaneous injection may include pain, bruising of the skin, soreness, swelling, and possible infection at the injection site.
These are not all the possible side effects of REGEN-COV. Not a lot of people have been given REGEN-COV. Serious and unexpected side effects may happen. REGEN-COV is still being studied so it is possible that all of the risks are not known at this time.
It is possible that REGEN-COV could interfere with your body's own ability to fight off a future infection of SARS-CoV-2. Similarly, REGEN-COV may reduce your body's immune response to a vaccine for SARS-CoV-2. Specific studies have not been conducted to address these possible risks. Talk to your healthcare provider if you have any questions.
WHAT OTHER TREATMENT CHOICES ARE THERE?
Like REGEN-COV (casirivimab and imdevimab), FDA may allow for the emergency use of other medicines to treat people with COVID-19. Go to https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization for information on the emergency use of other medicines that are not approved by FDA that are used to treat people with COVID-19. Your healthcare provider may talk with you about clinical trials you may be eligible for.
It is your choice to be treated or not to be treated with REGEN-COV. Should you decide not to receive REGEN-COV or stop it at any time, it will not change your standard medical care.
WHAT OTHER PREVENTION CHOICES ARE THERE?
Vaccines to prevent COVID-19 are also available under Emergency Use Authorization. Use of REGEN-COV does not replace vaccination against COVID-19. REGEN-COV is not authorized for pre-exposure prophylaxis for prevention of COVID-19.
WHAT IF I AM PREGNANT OR BREASTFEEDING?
There is limited experience using REGEN-COV (casirivimab and imdevimab) in pregnant women or breastfeeding mothers. For a mother and unborn baby, the benefit of receiving REGEN-COV may be greater than the risk of using the product. If you are pregnant or breastfeeding, discuss your options and specific situation with your healthcare provider.
HOW DO I REPORT SIDE EFFECTS WITH REGEN-COV (casirivimab and imdevimab)?
Tell your healthcare provider right away if you have any side effect that bothers you or does not go away.
Report side effects to FDA MedWatch at www.fda.gov/medwatch or call 1-800-FDA-1088 or call 1-844-734-6643.
HOW CAN I LEARN MORE?
- Ask your health care provider.
- Visit www.REGENCOV.com
- Visit https://www.covid19treatmentguidelines.nih.gov/
- Contact your local or state public health department.
WHAT IS AN EMERGENCY USE AUTHORIZATION (EUA)?
The United States FDA has made REGEN-COV (casirivimab and imdevimab) available under an emergency access mechanism called an EUA. The EUA is supported by a Secretary of Health and Human Service (HHS) declaration that circumstances exist to justify the emergency use of drugs and biological products during the COVID-19 pandemic.
REGEN-COV has not undergone the same type of review as an FDA-approved product. In issuing an EUA under the COVID-19 public health emergency, the FDA must determine, among other things, that based on the totality of scientific evidence available, it is reasonable to believe that the product may be effective for diagnosing, treating, or preventing COVID-19, or a serious or life-threatening disease or condition caused by COVID-19; that the known and potential benefits of the product, when used to diagnose, treat, or prevent such disease or condition, outweigh the known and potential risks of such product; and that there are no adequate, approved and available alternatives. All of these criteria must be met to allow for the medicine to be used in the treatment of COVID-19 or prevention of COVID-19 during the COVID-19 pandemic.
The EUA for REGEN-COV is in effect for the duration of the COVID-19 declaration justifying emergency use of these products, unless terminated or revoked (after which the products may no longer be used).
Manufactured by:
Regeneron Pharmaceuticals, Inc.
777 Old Saw Mill River Road
Tarrytown, NY 10591-6707
©2022 Regeneron Pharmaceuticals, Inc. All rights reserved.
Revised: 01/2022
PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Label - Casirivimab
701208
12345-00
Casirivimab
Injection
NDC: 61755-026-00
Rx only
300 mg/2.5 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH IMDEVIMAB
Mfd by: Regeneron Pharmaceuticals, Inc.
LOT/EXP
PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Label - Casirivimab - Roche
Casirivimab
120 mg/mL
concentrate for solution
for infusion
300 mg/2.5 mL
IV after dilution
Must administer with
imdevimab
Roche
EXP
Lot
10238448 IE (EG)
PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Carton - Casirivimab
NDC: 61755-026-01
Rx only
Casirivimab
Injection
300 mg/2.5 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH IMDEVIMAB
Contains 1 Single-Dose Vial
Discard unused portion

PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Box - Casirivimab
Casirivimab (REGN10933)
300 mg/2.5 mL (120 mg/mL)
NDC: 61755-026-01
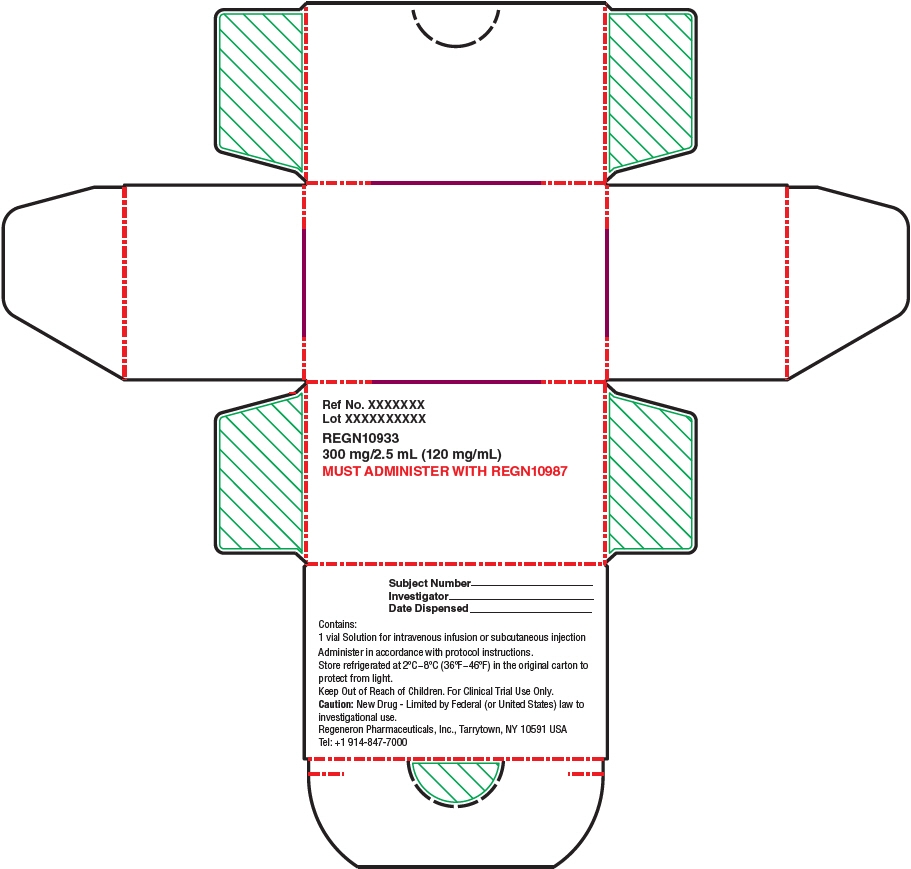
PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Modified Vial Label - REGN10933
701208
Subject Number
Solution for intravenous infusion or subcutaneous injection.
Administer in accordance with protocol instructions.
Store refrigerated at 2°C–8°C (36°F–46°F)
in original carton to protect from light.
Keep Out of Reach of Children. For Clinical Trial Use Only.
Caution: New Drug - Limited by Federal (or United States)
law to investigational use. Regeneron Pharmaceuticals, Inc.
Tarrytown, NY 10591 USA Tel: +1 914-847-7000
Lot

PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Modified Vial Carton - REGN10933
Subject Number
Investigator
Date Dispensed
Contains:
1 vial Solution for intravenous infusion or subcutaneous injection
Administer in accordance with protocol instructions.
Store refrigerated at 2°C–8°C (36°F–46°F) in the original carton to
protect from light.
Keep Out of Reach of Children. For Clinical Trial Use Only.
Caution: New Drug - Limited by Federal (or United States) law to
investigational use.
Regeneron Pharmaceuticals, Inc., Tarrytown, NY 10591 USA
Tel: +1 914-847-7000
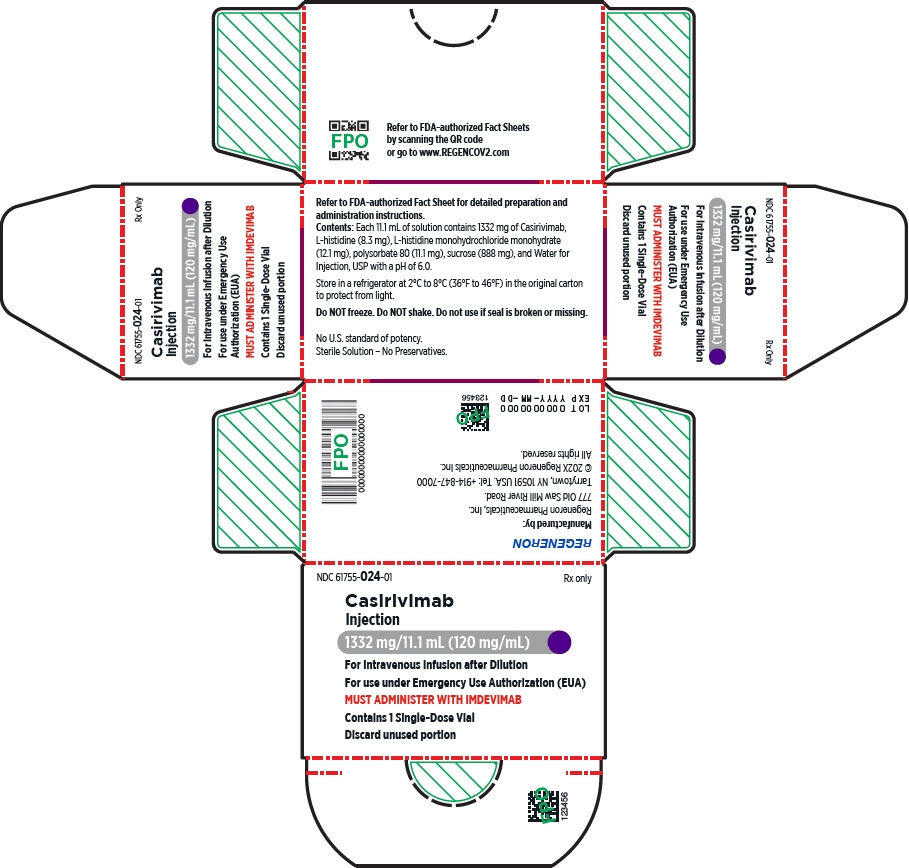
PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Label - Casirivimab
701210
12345-00
Casirivimab
Injection
NDC: 61755-024-00
Rx only
1332 mg/11.1 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH IMDEVIMAB
Mfd by: Regeneron Pharmaceuticals, Inc.

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Label - Casirivimab - Roche
Casirivimab 120 mg/mL
concentrate for solution for
infusion
1332 mg/11.1 mL
IV after dilution
Must administer with
imdevimab
Roche
EXP
Lot
10238449 IE (EG)

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Carton - Casirivimab
NDC: 61755-024-01
Rx only
Casirivimab
Injection
1332 mg/11.1 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH IMDEVIMAB
Contains 1 Single-Dose Vial
Discard unused portion

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Box - Casirivimab
Casirivimab (REGN10933)
1332 mg/11.1 mL (120 mg/mL)
NDC: 61755-024-01

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Initial Clinical Vial Label - REGN10933
701210
Subject Number
Solution for intravenous administration.
Administer in accordance with protocol instructions.
Store refrigerated at 2°C–8°C (36°F-46°F)
in the original carton to protect from light.
Keep Out of Reach of Children. For Clinical Trial Use Only.
Caution: New Drug – Limited by Federal (or United States) law
to investigational use. Regeneron Pharmaceuticals, Inc.
Tarrytown, NY 10591 USA Tel: +1 914-847-7000
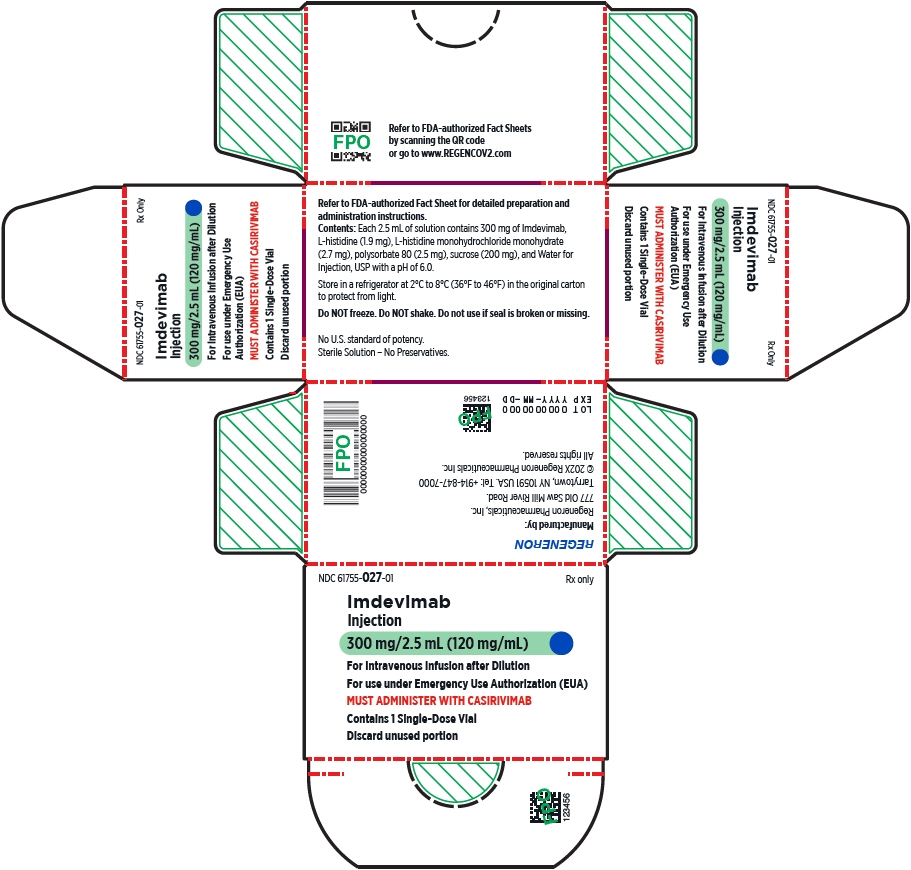
PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Initial Clinical Vial Carton - REGN10933
Ref No. XXXXXXX
Lot XXXXXXXXXX
120 mg/mL 11.1 mL
REGN10933

PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Label - Imdevimab
701208
12345-00
Imdevimab
Injection
NDC: 61755-027-00
Rx only
300 mg/2.5 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH CASIRIVIMAB
Mfd by: Regeneron Pharmaceuticals, Inc.
LOT/EXP
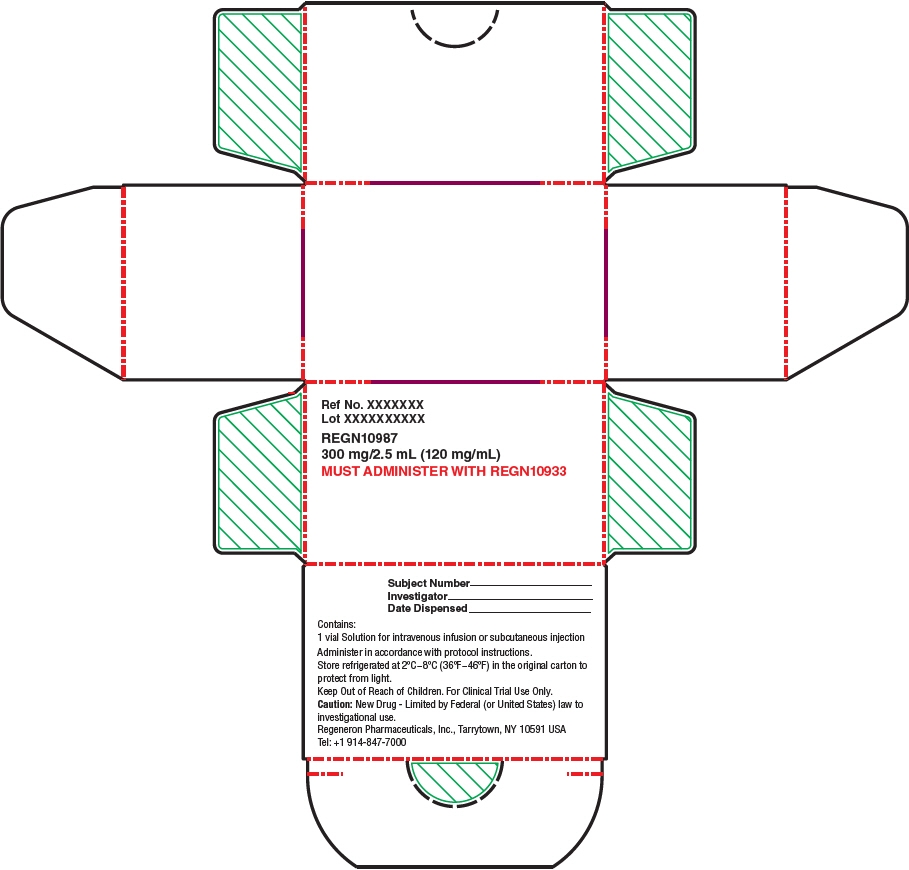
PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Label - Imdevimab - Roche
Imdevimab
120 mg/mL
concentrate for solution
for infusion
300 mg/2.5 mL
IV after dilution
Must administer with
casirivimab
Roche
EXP
Lot
10238451 IE (EG)

PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Carton - Imdevimab
NDC: 61755-027-01
Rx only
Imdevimab
Injection
300 mg/2.5 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH CASIRIVIMAB
Contains 1 Single-Dose Vial
Discard unused portion

PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Vial Box - Imdevimab
Imdevimab (REGN10987)
300 mg/2.5 mL (120 mg/mL)
NDC: 61755-027-01
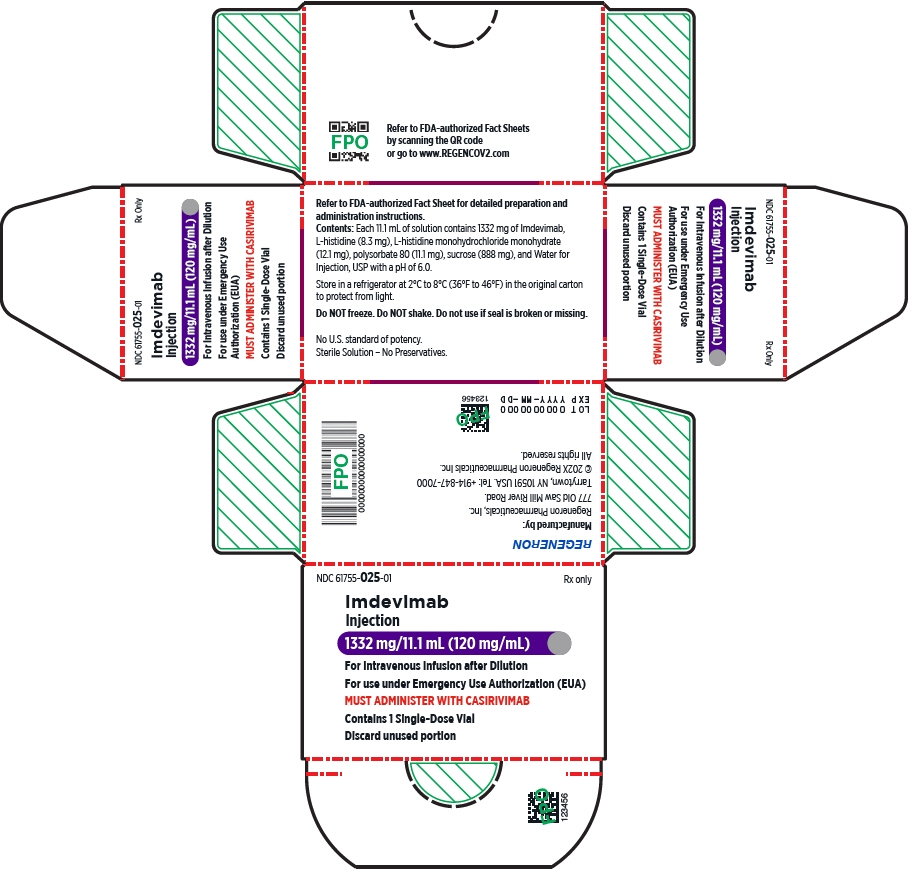
PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Modified Vial Label - REGN10987
701208
Subject Number
Solution for intravenous infusion or subcutaneous injection.
Administer in accordance with protocol instructions.
Store refrigerated at 2°C–8°C (36°F–46°F)
in original carton to protect from light.
Keep Out of Reach of Children. For Clinical Trial Use Only.
Caution: New Drug - Limited by Federal (or United States)
law to investigational use. Regeneron Pharmaceuticals, Inc.
Tarrytown, NY 10591 USA Tel: +1 914-847-7000
Lot

PRINCIPAL DISPLAY PANEL - 300 mg/2.5 mL Modified Vial Carton - REGN10987
Subject Number
Investigator
Date Dispensed
Contains:
1 vial Solution for intravenous infusion or subcutaneous injection
Administer in accordance with protocol instructions.
Store refrigerated at 2°C–8°C (36°F–46°F) in the original carton to
protect from light.
Keep Out of Reach of Children. For Clinical Trial Use Only.
Caution: New Drug - Limited by Federal (or United States) law to
investigational use.
Regeneron Pharmaceuticals, Inc., Tarrytown, NY 10591 USA
Tel: +1 914-847-7000

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Label - Imdevimab
701210
12345-00
Imdevimab
Injection
NDC: 61755-025-00
Rx only
1332 mg/11.1 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH CASIRIVIMAB
Mfd by: Regeneron Pharmaceuticals, Inc.

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Label - Imdevimab - Roche
Imdevimab 120 mg/mL
concentrate for solution for
infusion
1332 mg/11.1 mL
IV after dilution
Must administer with
casirivimab
Roche
EXP
Lot
10238452 IE (EG)

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Carton - Imdevimab
NDC: 61755-025-01
Rx only
Imdevimab
Injection
1332 mg/11.1 mL (120 mg/mL)
For Intravenous Infusion after Dilution
For use under Emergency Use Authorization (EUA)
MUST ADMINISTER WITH CASIRIVIMAB
Contains 1 Single-Dose Vial
Discard unused portion

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Vial Box - Imdevimab
Imdevimab (REGN10987)
1332 mg/11.1 mL (120 mg/mL)
NDC: 61755-025-01

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Initial Clinical Vial Label - REGN10987
701210
Subject Number
Solution for intravenous administration.
Administer in accordance with protocol instructions.
Store refrigerated at 2°C–8°C (36°F–46°F)
in original carton to protect from light.
Keep Out of Reach of Children. For Clinical Trial Use Only.
Caution: New Drug – Limited by Federal (or United States) law
to investigational use. Regeneron Pharmaceuticals, Inc.
Tarrytown, NY 10591 USA Tel: +1 914-847-7000

PRINCIPAL DISPLAY PANEL - 1332 mg/11.1 mL Initial Clinical Vial Carton - REGN10987
Ref No. XXXXXXX
Lot XXXXXXXXXX
120 mg/mL 11.1 mL
REGN10987
PRINCIPAL DISPLAY PANEL - 600 mg/10 mL Vial Label
702310
13443-00
NDC: 61755-039-00
Rx only
REGEN-COV™
(casirivimab and imdevimab)
Injection
600 mg / 600 mg per 10 mL
(60 mg / 60 mg per mL)
For use under Emergency Use Authorization (EUA)
One 10 mL Single-Dose Vial. Discard unused portion.
Mfd by: Regeneron Pharmaceuticals, Inc.

PRINCIPAL DISPLAY PANEL - 600 mg/10 mL Vial Carton
NDC: 61755-039-01
Rx only
REGEN-COV™
(casirivimab and imdevimab)
Injection
600 mg / 600 mg per 10 mL
(60 mg / 60 mg per mL)
Refer to FDA-authorized Fact Sheets for detailed instructions
on dosage, preparation, and route of administration.
For use under Emergency Use Authorization (EUA)
One 10 mL Single-Dose Vial. Discard unused portion.

PRINCIPAL DISPLAY PANEL - Dose Pack - 61755-035-02
NDC: 61755-035-02
REGEN-COV™
[casirivimab (REGN10933) with imdevimab (REGN10987)]
This dose pack provides one complete dose, and contains:
- 1 vial of casirivimab 1332 mg/11.1 mL (120 mg/mL)
- 1 vial of imdevimab 1332 mg/11.1 mL (120 mg/mL)
Must dilute and administer together via intravenous infusion.
Refer to FDA-authorized Fact Sheet for detailed preparation
and administration instructions.
For Use under Emergency Authorization (EUA).
Do not open this dose pack until time of dose preparation.
Store dose pack refrigerated between 2°C - 8°C (36°F to 46°F).
Do Not Freeze.
Dose Pack Lot #: XXX
13451
REGENERON
Bar code for Manufacturer internal use only.
TEL: +1 844-734-6643

PRINCIPAL DISPLAY PANEL - Dose Pack - 61755-036-08
NDC: 61755-036-08
REGEN-COV™
[casirivimab (REGN10933) with imdevimab (REGN10987)]
This dose pack provides one complete dose, and contains:
- 4 vials of casirivimab 300 mg/2.5 mL (120 mg/mL)
- 4 vials of imdevimab 300 mg/2.5 mL (120 mg/mL)
Must dilute and administer together via intravenous infusion.
Refer to FDA-authorized Fact Sheet for detailed preparation
and administration instructions.
For Use under Emergency Authorization (EUA).
Do not open this dose pack until time of dose preparation.
Store dose pack refrigerated between 2°C - 8°C (36°F to 46°F).
Do Not Freeze.
Dose Pack Lot #: XXX
13452
REGENERON
Bar code for Manufacturer internal use only.
TEL: +1 844-734-6643

PRINCIPAL DISPLAY PANEL - Dose Pack - 61755-037-05
NDC: 61755-037-05
REGEN-COV™
[casirivimab (REGN10933) with imdevimab (REGN10987)]
This dose pack provides one complete dose, and contains:
- 1 vial of casirivimab 1332 mg/11.1 mL (120 mg/mL)
- 4 vials of imdevimab 300 mg/2.5 mL (120 mg/mL)
Must dilute and administer together via intravenous infusion.
Refer to FDA-authorized Fact Sheet for detailed preparation
and administration instructions.
For Use under Emergency Authorization (EUA).
Do not open this dose pack until time of dose preparation.
Store dose pack refrigerated between 2°C - 8°C (36°F to 46°F).
Do Not Freeze.
Dose Pack Lot #: XXX
13453
REGENERON
Bar code for Manufacturer internal use only.
TEL: +1 844-734-6643

PRINCIPAL DISPLAY PANEL - Dose Pack - 61755-038-05
NDC: 61755-038-05
REGEN-COV™
[casirivimab (REGN10933) with imdevimab (REGN10987)]
This dose pack provides one complete dose, and contains:
- 4 vials of casirivimab 300 mg/2.5 mL (120 mg/mL)
- 1 vial of imdevimab 1332 mg/11.1 mL (120 mg/mL)
Must dilute and administer together via intravenous infusion.
Refer to FDA-authorized Fact Sheet for detailed preparation
and administration instructions.
For Use under Emergency Authorization (EUA).
Do not open this dose pack until time of dose preparation.
Store dose pack refrigerated between 2°C - 8°C (36°F to 46°F).
Do Not Freeze.
Dose Pack Lot #: XXX
13454
REGENERON
Bar code for Manufacturer internal use only.
TEL: +1 844-734-6643

| CASIRIVIMAB
casirivimab injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CASIRIVIMAB
casirivimab injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| IMDEVIMAB
imdevimab injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| IMDEVIMAB
imdevimab injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| REGEN-COV
casirivimab and imdevimab injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| REGEN-COV
casirivimab and imdevimab kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| REGEN-COV
casirivimab and imdevimab kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| REGEN-COV
casirivimab and imdevimab kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| REGEN-COV
casirivimab and imdevimab kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CASIRIVIMAB WITH IMDEVIMAB
casirivimab and imdevimab kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CASIRIVIMAB WITH IMDEVIMAB
casirivimab and imdevimab kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Regeneron Pharmaceuticals, Inc. (194873139) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Catalent Indiana, LLC | 172209277 | ANALYSIS(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) , MANUFACTURE(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) , LABEL(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) , PACK(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Integrated commercial solutions, LLC | 081015291 | LABEL(61755-026, 61755-024, 61755-027, 61755-025, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) , PACK(61755-026, 61755-024, 61755-027, 61755-025, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Regeneron Pharmaceuticals, Inc | 945589711 | LABEL(61755-026, 61755-024, 61755-027, 61755-025, 61755-035, 61755-036, 61755-037, 61755-038, 61755-042, 61755-045) , ANALYSIS(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038) , API MANUFACTURE(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038) , MANUFACTURE(61755-026, 61755-024, 61755-027, 61755-025, 61755-039, 61755-035, 61755-036, 61755-037, 61755-038) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| EMERGENT BIOSOLUTIONS | 050783398 | LABEL(61755-039) , PACK(61755-039) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Genentech, Inc | 833220176 | MANUFACTURE(61755-042, 61755-045) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Genentech, Inc | 004074162 | API MANUFACTURE(61755-042, 61755-045) , ANALYSIS(61755-042, 61755-045) | |