Laxative pills by McKesson (Sunmark) DRUG FACTS
Laxative pills by
Drug Labeling and Warnings
Laxative pills by is a Otc medication manufactured, distributed, or labeled by McKesson (Sunmark). Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LAXATIVE PILLS MAXIMUM STRENGTH- sennosides tablet
McKesson (Sunmark)
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
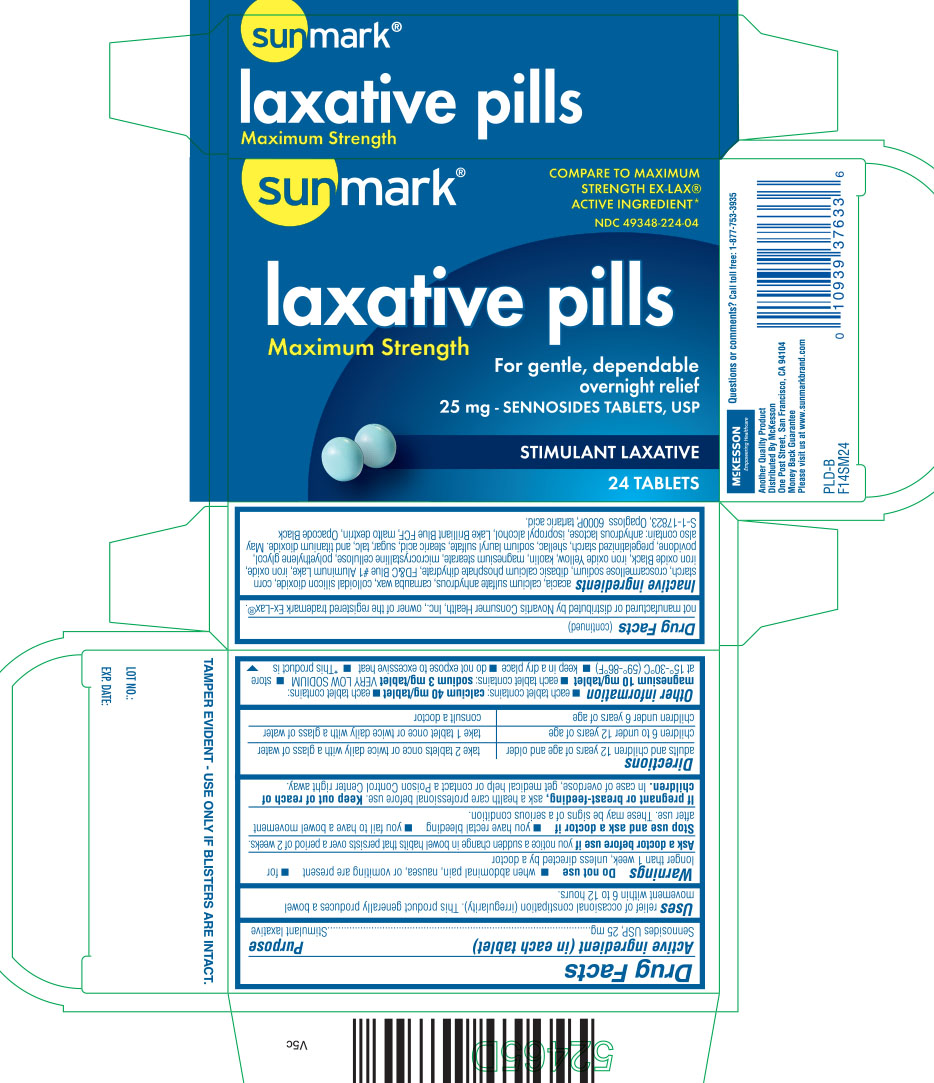
DRUG FACTS
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement within 6 to 12 hours
Warnings
Do not use
- when abdominal pain, nausea, or vomiting are present
- for longer than 1 week, unless directed by a doctor
Ask a doctor before use if
- you notice a sudden change in bowel habits that persists over a period of 2 weeks
Directions
| adults and children 12 years of age and older | take 2 tablets once or twice daily with a glass of water |
| children 6 to under 12 years of age | take 1 tablet once or twice daily with a glass of water |
| children under 6 years of age | consult a doctor |
Other information
- each tablet contains: calcium 40 mg/ tablet
- each tablet contains: magnesium 10 mg/ tablet
- each tablet contains: sodium 3 mg/ tablet VERY LOW SODIUM
- store at 15º - 30ºC (59º-86ºF)
- keep in a dry place
- do not expose to excessive heat
- *This product is not manufactured or distributed by Novartis Consumer Health Inc., owner of the registered trademark Ex-Lax®
Inactive ingredients
acacia, calcium sulfate anhydrous, carnauba wax, colloidal silicon dioxide, corn starch, croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C Blue #1 Aluminum Lake, iron oxide, iron oxide Black, iron oxide Yellow, kaolin, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, pregelatinized starch, shellac, sodium lauryl sulfate, stearic acid, sugar, talc, and titanium dioxide. May also contain: anhydrous lactose, isopropyl alcohol, Lake Brilliant Blue FCF, malto dextrin, Opacode Black S-1-17823, Opagloss 6000P, tartaric acid
Principal Display Panel
Compare to maximum strength EX-LAX® active ingredient*
laxative pills
maximum strength
for gentle, dependable overnight relief
25 mg- Sennosides tablets, USP
stimulant laxative
TAMPER EVIDENT- USE ONLY IF BLISTERS ARE INTACT
Questions or comments? Call toll free: 1-877-753-3935
Another Quality product
Distributed by McKesson
One post street, San Francisco, CA 94104
Money Back guarantee
Please visit us at www.sunmarkbrand.com
| LAXATIVE PILLS
MAXIMUM STRENGTH
sennosides tablet |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - McKesson (Sunmark) (177667227) |