Quazepam by Atland Pharmaceuticals, LLC QUAZEPAM tablet
Quazepam by
Drug Labeling and Warnings
Quazepam by is a Prescription medication manufactured, distributed, or labeled by Atland Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use QUAZEPAM TABLETS safely and effectively. See full prescribing information for QUAZEPAM TABLETS.
QUAZEPAM TABLETS for oral use C-IV
Initial U.S. Approval: 1985WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death [see Warnings and Precautions (5.1), Drug Interactions (7).]
- Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation.
RECENT MAJOR CHANGES
Warnings and Precautions ( 5.2) 12/2018 INDICATIONS AND USAGE
Quazepam, a gamma-aminobutyric (GABAA) agonist, is indicated for the treatment of insomnia characterized by difficulty falling asleep, frequent nocturnal awakenings, and/or early morning awakenings. ( 1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
15 mg functionally scored tablet, oral ( 3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- CNS depressant effects: Impaired alertness and motor coordination, including risk of daytime impairment. Caution patients against driving and other activities requiring complete mental alertness ( 5.2)
- Benzodiazepine withdrawal syndrome: avoid abrupt discontinuation in at-risk patients ( 5.3)
- The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. ( 5.4)
- Severe anaphylactic/anaphylactoid reactions: Angioedema and anaphylaxis have been reported. Do not rechallenge if such reactions occur. ( 5.5)
- Sleep driving and other complex behaviors while not fully awake. Risk increases with dose and concomitant CNS depressants and alcohol. Immediately evaluate any new onset behavioral changes ( 5.6)
- Worsening of depression or suicidal thinking may occur: Prescribe the least number of tablets feasible to avoid intentional overdose ( 5.7)
ADVERSE REACTIONS
Most common adverse reactions (>1%): drowsiness, headache, fatigue, dizziness, dry mouth, dyspepsia ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Atland Pharmaceuticals at 1-844-416-4284 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. ( 6)
DRUG INTERACTIONS
- CNS Depressants: downward dose adjustment may be necessary due to additive effects ( 7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 12/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS
RECENT MAJOR CHANGES
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Special Populations
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risks from Concomitant Use with Opioids
5.2 CNS-Depressant Effects and Daytime Impairment
5.3 Benzodiazepine Withdrawal Syndrome
5.4 Need to Evaluate for Co-morbid Diagnoses
5.5 Severe Anaphylactic and Anaphylactoid Reactions
5.6 Abnormal Thinking and Behavior Changes
5.7 Worsening of Depression
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse and Dependence
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED / STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death [see Warnings and Precautions (5.1), Drug Interactions (7).]
- Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation.
-
1 INDICATIONS AND USAGE
Quazepam is indicated for the treatment of insomnia characterized by difficulty in falling asleep, frequent nocturnal awakenings, and/or early morning awakenings. The effectiveness of quazepam has been established in placebo-controlled clinical studies of 5 nights duration in acute and chronic insomnia. The sustained effectiveness of quazepam has been established in chronic insomnia in a sleep lab (polysomnographic) study of 28 nights duration. Because insomnia is often transient and intermittent, the prolonged administration of Quazepam Tablets is generally not necessary or recommended. Since insomnia may be a symptom of several other disorders, the possibility that the complaint may be related to a condition for which there is a more specific treatment should be considered.
- 2 DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Quazepam Tablets are contraindicated in patients with known hypersensitivity to quazepam or other benzodiazepines. Rare cases of angioedema involving the tongue, glottis or larynx have been reported in patients after taking the first or subsequent doses of quazepam. Some patients have had additional symptoms such as dyspnea, throat closing, or nausea and vomiting that suggest anaphylaxis. Patients who develop such reactions should not be rechallenged with quazepam.
Contraindicated in patients with established or suspected sleep apnea, or with pulmonary insufficiency.
-
5 WARNINGS AND PRECAUTIONS
5.1 Risks from Concomitant Use with Opioids
Concomitant use of benzodiazepines, including quazepam, and opioids may result in profound sedation, respiratory depression, coma, and death. Because of these risks, reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioids alone. If a decision is made to prescribe quazepam concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. In patients already receiving an opioid analgesic, prescribe a lower initial dose of quazepam than indicated in the absence of an opioid and titrate based on clinical response. If an opioid is initiated in a patient already taking quazepam, prescribe a lower initial dose of the opioid and titrate based upon clinical response.
Advise both patients and caregivers about the risks of respiratory depression and sedation when quazepam is used with opioids. Advise patients not to drive or operate heavy machinery until the effects of concomitant use with the opioid have been determined. [see Drug Interactions (7), Patient Counseling (17)].
5.2 CNS-Depressant Effects and Daytime Impairment
Quazepam is a central nervous system (CNS) depressant and can impair daytime function in some patients even when used as prescribed. Prescribers should monitor for excess depressant effects, but impairment can occur in the absence of subjective symptoms, and may not be reliably detected by ordinary clinical exam (i.e. less than formal psychomotor testing). While pharmacodynamics tolerance or adaptation to some adverse depressant effects of quazepam may develop, patients using quazepam should be cautioned against driving or engaging in other hazardous activities or activities requiring complete mental alertness.
Additive effects occur with concomitant use of other CNS depressants (e.g., other benzodiazepines, opioids, tricyclic antidepressants, alcohol), including daytime use. Downward dose adjustment of quazepam and concomitant CNS depressants should be considered. The potential for adverse drug interactions continues for several days following discontinuation of quazepam, until serum levels of both active parent drug and psychoactive metabolites decline.
Use of quazepam with other sedative-hypnotics is not recommended. Alcohol generally should not be used during treatment with quazepam. The risk of next-day psychomotor impairment is increased if quazepam is taken with less than a full night of sleep remaining (7 to 8 hours); if higher than the recommended dose is taken; if co-administered with other CNS depressants [see Dosage and Administration (2)].
Because quazepam can cause drowsiness and a decreased level of consciousness, patients particularly the elderly, are at higher risk of falls.
5.3 Benzodiazepine Withdrawal Syndrome
A withdrawal syndrome similar to that from alcohol (e.g., convulsions, tremor, abdominal and muscle cramps, vomiting, and sweating) can occur following abrupt discontinuation of quazepam. The more severe withdrawal effects are usually limited to patients taking higher than recommended doses over an extended time. Abrupt discontinuation should be avoided in such patients, and the dose gradually tapered. Prescribers should monitor patients for tolerance, abuse, and dependence.
Milder withdrawal symptoms (e.g., dysphoria and insomnia) can occur following abrupt discontinuation of benzodiazepines taken at therapeutic levels for short periods [See Drug Abuse and Dependence (9)].
5.4 Need to Evaluate for Co-morbid Diagnoses
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after a careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. Worsening of insomnia or the emergence of new thinking or behavior abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with sedative-hypnotic drugs.
5.5 Severe Anaphylactic and Anaphylactoid Reactions
Rare cases of angioedema involving the tongue, glottis or larynx have been reported in patients after taking the first or subsequent doses of sedative-hypnotics, including quazepam. Some patients have had additional symptoms such as dyspnea, throat closing, or nausea and vomiting that suggest anaphylaxis.
Some patients have required medical therapy in the emergency department. If angioedema involves the tongue, glottis or larynx, airway obstruction may occur and be fatal. Patients who develop angioedema after treatment with quazepam should not be rechallenged with the drug.
5.6 Abnormal Thinking and Behavior Changes
Abnormal thinking and behavior changes have been reported in patients treated with sedative-hypnotics including quazepam. Some of these changes include decreased inhibition (e.g., aggressiveness and extroversion that seemed out of character), bizarre behavior, and depersonalization. Visual and auditory hallucinations have also been reported. Amnesia, and other neuro-psychiatric symptoms may occur.
Paradoxical reactions such as stimulation, agitation, increased muscle spasticity, and sleep disturbances may occur unpredictably.
Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake, with amnesia for the event) have been reported with use of sedative-hypnotics. These behaviors can occur with initial treatment or in patients previously tolerant of quazepam or other sedative-hypnotics. Although these behaviors can occur with use at therapeutic doses, risk is increased by higher doses or concomitant use of alcohol or other CNS depressants. Due to risk to the patient and community, quazepam should be discontinued if "sleep-driving" occurs.
Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with sleep-driving, patients usually do not remember these events.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- CNS-depressant effects and next-day impairment [see Warnings and Precautions (5.2)]
- Benzodiazepine withdrawal syndrome [see Warnings and Precautions (5.3)]
- Abnormal thinking and behavior changes, and complex behaviors [see Warnings and Precautions (5.6)]
- Worsening of depression [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The table shows adverse reactions occurring at an incidence of 1% or greater in relatively short-duration, placebo-controlled clinical trials of quazepam.
Quazepam 15 mg PLACEBO NUMBER OF PATIENTS 267 268 % OF PATIENTS REPORTING Central Nervous System Daytime Drowsiness 12 3 Headache 5 2 Fatigue 2 0 Dizziness 2 <1 Autonomic Nervous System Dry Mouth 2 <1 Gastrointestinal System Dyspepsia 1 <1 A double-blind, controlled sleep laboratory study (N=30) in elderly patients compared the effects of quazepam 7.5 mg and 15 mg to that of placebo over a period of 7 days. Both the 7.5 mg and 15 mg doses appeared to be well tolerated. Caution must be used in interpreting this data due to the small size of the study.
-
7 DRUG INTERACTIONS
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and monitor patients closely for respiratory depression and sedation.
Benzodiazepines, including quazepam, produce additive CNS depressant effects when co-administered with ethanol or other CNS depressants (e.g. psychotropic medications, anticonvulsants, antihistamines). Downward dose adjustment of quazepam and/or concomitant CNS depressants may be necessary because of additive effects.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
There are no adequate and well-controlled studies in pregnant women. Administration of benzodiazepines immediately prior to or during childbirth can result in a syndrome of hypothermia, hypotonia, respiratory depression, and difficulty feeding. In addition, infants born to mothers who have taken benzodiazepines during the later stages of pregnancy can develop dependence, and subsequently withdrawal, during the postnatal period. Although administration of quazepam to pregnant animals did not indicate a risk for adverse effects on morphological development at clinically relevant doses, data for other benzodiazepines suggest the possibility of adverse developmental effects (long-term effects on neurobehavioral and immunological function) in animals following prenatal exposure to benzodiazepines. Quazepam should be used during pregnancy only if the potential benefit justifies the potential risk.
Developmental toxicity studies of quazepam in mice at doses up to 400 times the human dose (15 mg) revealed no major drug-related malformations. Minor fetal skeletal variations that occurred were delayed ossification of the sternum, vertebrae, distal phalanges and supraoccipital bones, at doses approximately 70 and 400 times the human dose. A developmental toxicity study of quazepam in New Zealand rabbits at doses up to approximately 130 times the human dose demonstrated no effect on fetal morphology or development of offspring.
8.3 Nursing Mothers
Quazepam and its metabolites are excreted in human milk. Caution should be exercised when administering quazepam to a nursing woman.
8.5 Geriatric Use
Quazepam may cause confusion and over-sedation in the elderly. Elderly patients generally should be started on a low dose of quazepam and observed closely. Elderly and debilitated patients may be more sensitive to benzodiazepines, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. A double-blind controlled sleep laboratory study (N=30) compared the effects of quazepam 7.5 mg and 15 mg to that of placebo over a period of 7 days. Both the 7.5 mg and 15 mg doses appeared to be well tolerated. Caution must be used in interpreting this data due to the small size of the study.
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Quazepam is classified as a Schedule IV controlled substance by federal regulation.
9.2 Abuse and Dependence
Addiction-prone individuals (e.g. history of drug addiction or alcoholism) should be under careful surveillance when receiving quazepam because of increased risk of abuse and dependence. Benzodiazepine withdrawal symptoms can occur following discontinuation of quazepam [see Warnings and Precautions (5.2)].
Abuse and addiction are separate and distinct from physical dependence and tolerance. Abuse is characterized by misuse of the drug for non-medical purposes, often in combination with other psychoactive substances. Physical dependence is a state of adaptation that is manifested by a specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug and/or administration of an antagonist. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug’s effects over time. Tolerance may occur to both the desired and undesired effects of drugs and may develop at different rates for different effects.
Addiction is a primary, chronic, neurobiological disease with genetic, psychosocial, and environmental factors influencing its development and manifestations. It is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, utilizing a multidisciplinary approach, but relapse is common.
-
10 OVERDOSAGE
Contact a poison control center for up-to-date information on the management of benzodiazepine overdose.
Manifestations of quazepam overdose include somnolence, confusion, and coma. General supportive measures should be employed, along with immediate gastric lavage. Dialysis is of limited value. Flumazenil may be useful, but can contribute to the appearance of neurological symptoms including convulsions. Hypotension may be treated by appropriate medical intervention. Animal experiments suggest that forced diuresis or hemodialysis are of little value in treating quazepam overdose. As with the management of intentional overdose with any drug, the possibility of multiple drug ingestion should be considered.
-
11 DESCRIPTION
Quazepam Tablets contains quazepam, a trifluoroethyl benzodiazepine hypnotic agent, having the chemical name 7-chloro-5- (o-fluoro-phenyl)-1,3-dihydro-1-(2,2,2- trifluoroethyl)-2H-1,4-benzodiazepine-2-thione and the following structural
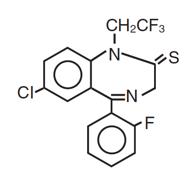
Quazepam has the empirical formula C 17H 11ClF 4N 2S, and a molecular weight of 386.8. It is a white crystalline compound, soluble in ethanol and insoluble in water.
Each Quazepam Tablet contains 15 mg of quazepam. The inactive ingredients for Quazepam Tablets include cellulose, corn starch, FD&C Yellow No. 6, lactose, magnesium stearate, silicon dioxide, and sodium lauryl sulfate.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Quazepam, like other central nervous system agents of the 14-benzodiazepine class, presumably exerts its effects by binding to stereo-specific receptors at several sites within the central nervous system (CNS). The exact mechanism of action is unknown.
12.3 Pharmacokinetics
Absorption
Quazepam is rapidly (absorption half-life of about 30 minutes) and well absorbed from the gastrointestinal tract. The peak plasma concentration of quazepam is approximately 20 ng/mL after a 15 mg dose and occurs at about 2 hours.
Metabolism
Quazepam, the active parent compound, is extensively metabolized in the liver; two of the plasma metabolites are 2-oxoquazepam and N-desalkyl-2-oxoquazepam. All three compounds show CNS depressant activity.
Distribution
The degree of plasma protein binding for quazepam and its two major metabolites is greater than 95%.
Elimination
Following administration of 14C-quazepam, 31% of the dose appeared in the urine and 23% in the feces over five days; only trace amounts of unchanged drug were present in the urine.
The mean elimination half-life of quazepam and 2-oxoquazepam is 39 hours and that of N-desalkyl-2-oxoquazepam is 73 hours. Steady-state levels of quazepam and 2-oxoquazepam are attained by the seventh daily dose and that of N-desalkyl-2-oxoquazepam by the thirteenth daily dose.
Special Populations
Geriatrics: The pharmacokinetics of quazepam and 2-oxoquazepam in geriatric subjects are comparable to those seen in young adults; as with desalkyl metabolites of other benzodiazepines, the elimination half-life of N-desalkyl-2-oxoquazepam in geriatric patients is about twice that of young adults.
Drug Interactions
Bupropion (a CYP2B6 substrate): Co-administration of a single dose of 150 mg Bupropion Hydrochloride XL with steady state quazepam did not significantly affect the AUC and Cmax of bupropion or its primary metabolite, hydroxybupropion.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Quazepam showed no evidence of carcinogenicity in oral carcinogenicity studies in mice and hamsters.
Mutagenesis
Quazepam was negative in the bacterial reverse mutation (Ames) assay and equivocal in the mouse lymphoma tk assay.
Impairment of Fertility
Reproduction studies in mice conducted with quazepam at doses equal to 60 and
180 times the human dose of 15 mg produced slight reductions in fertility rate. Similar reductions in fertility rate have been reported in mice dosed with other benzodiazepines, and is believed to be related to the sedative effects of these drugs at high doses
-
14 CLINICAL STUDIES
The effectiveness of quazepam was established in placebo-controlled clinical studies of 5 nights duration in acute and chronic insomnia. The sustained effectiveness of quazepam was established in chronic insomnia in a sleep laboratory (polysomnographic) study of 28 nights duration. In the sleep laboratory study, quazepam significantly decreased sleep latency and total wake time, and significantly increased total sleep time and percent sleep time, for one or more nights.
Quazepam 15 mg was effective on the first night of administration. Sleep latency, total wake time and wake time after sleep onset were still decreased and percent sleep time was still increased for several nights after the drug was discontinued. Percent slow wave sleep was decreased, and REM sleep was essentially unchanged. No transient sleep disturbance, such as “rebound insomnia,” was observed after withdrawal of the drug in sleep laboratory studies in 12 patients using 15 mg doses.
A double-blind, controlled sleep laboratory study (N=30) in elderly patients compared the effects of quazepam 7.5 mg and 15 mg to that of placebo over a period of 7 days. Both the 7.5 mg and 15 mg doses appeared to be effective. Caution must be used in interpreting this data due to the small size of the study.
-
16 HOW SUPPLIED / STORAGE AND HANDLING
Quazepam Tablets, 15 mg, functionally scored, capsule-shaped, light orange, slightly white speckled tablets, impressed with the product identification number 15 on one side of the tablet, and the product name (DORAL) on the other.
15 mg Bottles of 30 NDC: 71993-265-30 15 mg Bottles of 100 NDC: 71993-265-10 Store Quazepam Tablets at controlled room temperature 20°-25°C (68°-77°F).
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling ( Medication Guide).
Inform patients and caregivers that potentially fatal additive effects may occur if quazepam is used with opioids and not to use such drugs concomitantly unless supervised by a healthcare provider [see Warnings and Precautions (5.1), Drug Interactions (7)]
Inform patients about the benefits and risks of quazepam, stressing the importance of use as directed. Assist patients in understanding the Medication Guide and instruct them to read it with each prescription refill.
CNS depressant Effects and Next-Day Impairment
Tell patients that quazepam can cause next-day impairment, even in the absence of symptoms. Caution patients against driving or engaging in other hazardous activities or activities requiring complete mental alertness when using quazepam. Tell patients that daytime impairment may persist for several days following discontinuation of quazepam. Advise patients that increased drowsiness and decreased consciousness may increase the risk of falls in some patients.
Withdrawal
Instruct patients to contact you before stopping or decreasing the dose of quazepam, because withdrawal symptoms can occur.
Abnormal thinking and behavior change
Instruct patients that sedative hypnotics can cause abnormal thinking and behavior change, including “sleep-driving” and other complex behaviors while not being fully awake (preparing and eating food, making phone calls, or having sex). Tell patients to call you immediately if they develop any of these symptoms.
Severe Allergic Reactions
Inform patients that severe allergic reactions can occur from quazepam. Describe the signs/symptoms of these reactions and advise patients to seek medical attention immediately if these occur.
Suicide
Tell patients that quazepam can worsen depression, and to immediately report any suicidal thoughts.
Alcohol and other drugs
Ask patients about alcohol consumption, medicines they are taking now, and drugs they may be taking without a prescription. Advise patients that alcohol generally should not be used during treatment with quazepam.
Pregnancy
Instruct patients to inform you if they are nursing or pregnant, or may become pregnant while taking quazepam.
Tolerance, Abuse, and Dependence
Tell patients not to increase the dose of quazepam on their own, and to inform you if they believe the drug “does not work”.
Manufactured for:
Atland Pharmaceuticals
Sylacauga, AL 35150Printed in USA. 500495-06 Rev. 12/2019
-
MEDICATION GUIDE
Medication Guide
quazepam (DOOR-al)
(quazepam) Tablets, C-IVWhat is the most important information I should know about DORAL?
- DORAL is a benzodiazepine medicine. Taking benzodiazepines with opioid medicines, alcohol, or other central nervous system depressants (including street drugs) can cause severe drowsiness, breathing problems (respiratory depression), coma and death.
- DORAL may cause serious side effects that you may not know are happening to you. These side effects include:
- sleepiness during the day
- not thinking clearly
- acting strangely, confused, or upset
- “sleep-walking” or doing other activities when you are asleep like:
- eating
- talking
- having sex
- driving a car
Call your healthcare provider right away if you find out that you have done any of the above activities after taking DORAL.
- Do not take DORAL unless you are able to stay in bed a full night (7 to 8 hours) before you must be active again.
- Do not take more DORAL than prescribed.
What is DORAL?
- DORAL is a prescription medicine used to treat certain types of insomnia including difficulty falling asleep, waking up often during the night, or waking up early in the morning.
- DORAL is a federal controlled substance (C-IV) because it can be abused or lead to dependence. Keep DORAL in a safe place to prevent misuse and abuse. Selling or giving away DORAL may harm others, and is against the law. Tell your healthcare provider if you have ever abused or been dependent on alcohol, prescription medicines or street drugs.
- It is not known if DORAL is safe and effective in children.
Do not take DORAL if you:
- are allergic to quazepam or any of the ingredients in DORAL. See the end of this Medication Guide for a complete list of ingredients in DORAL.
- have had an allergic reaction to other sleep medicines or sedatives such as benzodiazepines. Symptoms of a serious allergic reaction can include:
- swelling of your face, lips, and throat that may cause difficulty breathing or swallowing
- nausea and vomiting
- have sleep apnea, breathing or lung problems
Before you take DORAL, tell your healthcare provider about all of your medical conditions, including if you:
- have a history of depression, mental illness or, suicidal thoughts
- have a history of drug or alcohol abuse or addiction
- have lung disease or breathing problems
- are pregnant or plan to become pregnant. It is not known if DORAL can harm your unborn baby.
- are breastfeeding, or plan to breastfeed. DORAL can pass through your breast milk and may harm your baby. Talk to your healthcare provider about the best way to feed your baby if you take DORAL.
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Taking DORAL with certain other medicines can cause side effects or affect how well DORAL or the other medicines work.
Do not start or stop other medicines without talking to your healthcare provider.
Do not take DORAL with other medicines that can make you sleepy unless your healthcare provider tells you to.
How should I take DORAL?
- See “What is the most important information I should know about DORAL?”
- Take DORAL exactly as your healthcare providers tell you to take it.
- DORAL comes in 15 mg tablets. Your healthcare provider may start your DORAL dose at 7.5 mg which is half a tablet. Talk to your healthcare provider or pharmacist about your dose schedule.
- If you take too much DORAL or overdose, get emergency treatment right away.
What should I avoid while taking DORAL?
- Do not drive, operate machinery, do other dangerous activities or do anything that needs you to be alert until you know how DORAL affects you. You may still feel drowsy the next day after taking DORAL.
- You should not drink alcohol while you are taking DORAL.
What are the possible side effects of DORAL?
DORAL may cause serious side effects, including:
- See “What is the most important information I should know about DORAL?”
- Withdrawal symptoms. You may have withdrawal symptoms if you stop taking DORAL suddenly. Withdrawal symptoms can be serious and include seizures. Mild withdrawal symptoms include a depressed mood and trouble sleeping. Talk to your healthcare provider about slowly stopping DORAL to avoid withdrawal symptoms.
- Other conditions. Call your healthcare provider if your insomnia worsens or is not better within 7 to 10 days. This may mean that there is another condition causing your sleep problem.
- Severe allergic reactions. Symptoms include swelling of the tongue or throat, and trouble breathing. Other symptoms may include nausea and vomiting. Get emergency medical help right away if you have these symptoms after taking DORAL.
- Abnormal thoughts and behavior. Symptoms include more outgoing or aggressive behavior than normal, confusion, agitation, hallucinations, worsening of depression, and suicidal thoughts.
- Depression. Pre-existing depression may emerge or worsen during use of benzodiazepines including DORAL.
- Abuse and dependence. Taking DORAL can cause physical and psychological dependence. Physical and psychological dependence is not the same as drug addiction. Your healthcare provider can tell you more about the differences between physical and psychological dependence and drug addiction.
Call your healthcare provider right away if you have any of the above side effects while taking DORAL. The most common side effects of DORAL include:
- drowsiness
- headache
- feeling very tired
- dizziness
- dry mouth
- upset stomach
After you stop taking a sleep medicine, you may have symptoms for the next 1 to 2 days such as:
- trouble sleeping
- nausea
- flushing
- lightheadedness
- uncontrolled crying
- vomiting
- stomach cramps
- panic attack
- nervousness
- stomach area pain
These are not all the possible side effects of DORAL. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store DORAL?
- Store at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep DORAL and all medicines out of the reach of children.
General information about the safe and effective use of Doral.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use DORAL for a condition for which it was not prescribed. Do not give DORAL to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about DORAL that is written for healthcare professionals.
What are the ingredients in DORAL?
Active Ingredient: quazepam
Inactive Ingredients: cellulose, corn starch, FD&C Yellow No.6, lactose, magnesium stearate, silicon dioxide, and sodium lauryl sulfate
Distributed by Galt Pharmaceuticals, Atlanta, GA 30339 USA. If you would like more information, call Galt Pharmaceuticals at 1-855-965-2783 or visit www.doralrx.com.
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 04/2019 - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
QUAZEPAM
quazepam tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 71993-265 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength QUAZEPAM (UNII: JF8V0828ZI) (QUAZEPAM - UNII:JF8V0828ZI) QUAZEPAM 15 mg Inactive Ingredients Ingredient Name Strength POWDERED CELLULOSE (UNII: SMD1X3XO9M) STARCH, CORN (UNII: O8232NY3SJ) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color orange (light to medium orange) Score 2 pieces Shape CAPSULE Size 12mm Flavor Imprint Code 15;DORAL Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71993-265-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 03/20/2018 2 NDC: 71993-265-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/20/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018708 03/20/2018 Labeler - Atland Pharmaceuticals, LLC (080942150)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
