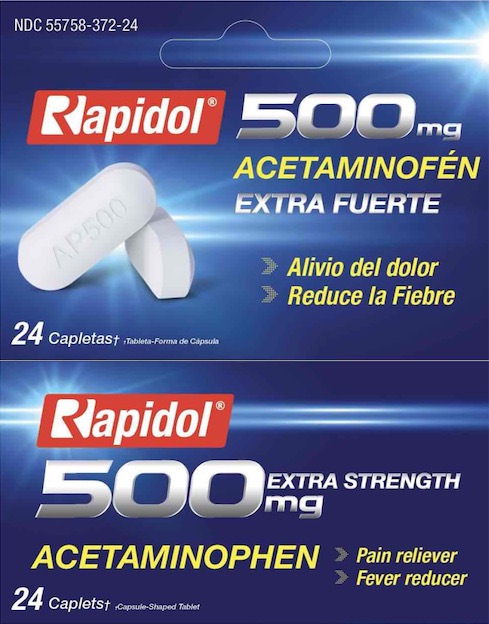
Rapidol Acetaminophen 500 (HPharma)
Dolex Acetaminophen 500mg by
Drug Labeling and Warnings
Dolex Acetaminophen 500mg by is a Otc medication manufactured, distributed, or labeled by Pharmadel LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DOLEX ACETAMINOPHEN 500MG EXTRA STRENGTH- acetaminophen capsule, coated
Pharmadel LLC
----------
Rapidol Acetaminophen 500 (HPharma)
Uses
For the temporary relief of minor aches and pains due to:
- the common cold
- headache
- backache
- muscular aches
- toothache
- minor pain of arthritis
- premenstrual and menstrual cramps
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4000mg in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
■ skin reddening
■ blisters
■ rash.
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- If allergic to of the inactive ingredients in this product
Directions
- do not take more than directed (see OVERDOSE WARNING)
| Age | Dose |
| adults and children 12 years of age and over |
|
| children under 12 years of age |
|
| DOLEX ACETAMINOPHEN 500MG
EXTRA STRENGTH
acetaminophen capsule, coated |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
 |
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Pharmadel LLC (030129680) |
Revised: 8/2025
Document Id: 3c2d68d6-6702-d7f0-e063-6294a90a74ee
Set id: f8c383c1-d1b7-ceaa-e053-6294a90a6053
Version: 4
Effective Time: 20250812
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.