MUCILIN by Paradigm Pharmaceuticals Inc / BERLIN PHARMACEUTICAL INDUSTRY COMPANY LIMITED - (BRANCH) MUCILIN Orange
MUCILIN by
Drug Labeling and Warnings
MUCILIN by is a Otc medication manufactured, distributed, or labeled by Paradigm Pharmaceuticals Inc, BERLIN PHARMACEUTICAL INDUSTRY COMPANY LIMITED - (BRANCH). Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
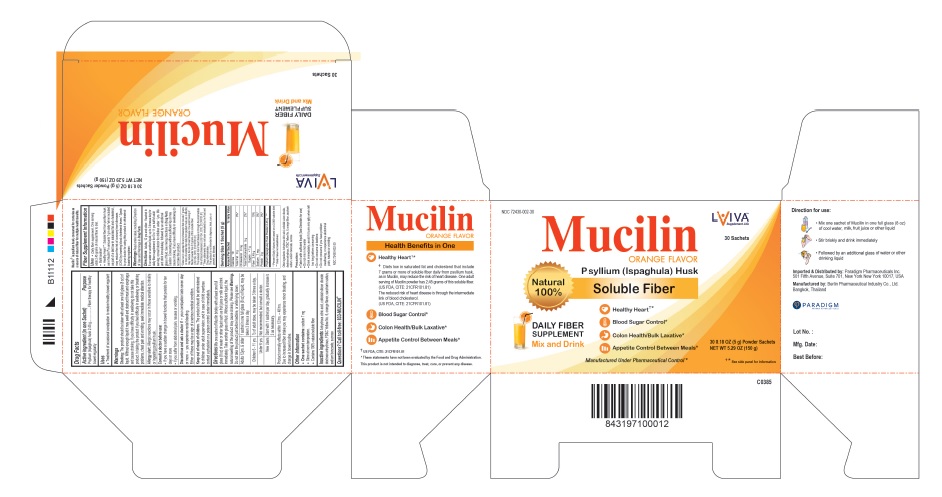
MUCILIN- psyllium (ispaghula) husk powder
Paradigm Pharmaceuticals Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
MUCILIN Orange
Warnings
Choking: The product should be taken with at least one full glass (8 oz) of liquid. Without enough liquid it may swell and obstruct throat and esophagus and cause choking. If you have difficulty in swallowing do not take this product. In taking the product if you feel difficulty in swallowing, breathing problems, chest pain, vomiting, seek immediate medical attention.
Allergy alert: Allergic reactions may occur in those sensitive to inhaling or ingesting psyllium husk.
Consult a doctor before use:
- if you have a sudden change in bowel functions that persists for ten days or more
- if you suffer from abdominal pain, nausea or vomiting.
Discontinue and ask a doctor if
- your constipation lasts seven days or more
- you experience rectal bleeding
Either of these may be a sign of a serious medical condition.
Keep out of reach of children. The product should be administered to children under adult supervision; and in case of child overdose.
Contact a physician or a poison control center immediately.
Directions
Mix one sachet of Mucilin briskly with at least one full glass (8 oz) of water or other liquid such as fruit juice or milk and drink immediately. Take another glass of fluid. Without sufficient liquid, the natural swelling of the product may cause choking. Please see Warnings. Do not take the product just before bedtime, or while lying down.
|
Adults 12yrs. & older |
1 sachet in 8 oz of liquid, may be taken 3 times a day. |
|
Children 6 - 11 yrs. |
½ of adult dose may be taken 3 times a day. |
|
Under 6 yrs. |
Not recommended, but consult a doctor. |
|
New Users |
Start with 1 dose per day, gradually increase to 3 as necessary. |
Product normally effects within 12 hrs. – 48 hrs.
Due to increased fiber intake you may experience minor bloating, and change in bowel routine.
Other Information
One sachet contains: sodium 7 mg
Store at room temperature.
Contains 100% natural soluble fiber
Inactive Ingredients
Anhydrous citric acid, colloidal silicon dioxide, dextrose anhydrous, FD&C Yellow No. 6, orange flavor, saccharin sodium, sodium benzoate, sucrose
| MUCILIN
psyllium (ispaghula) husk powder |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Paradigm Pharmaceuticals Inc (080044989) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| BERLIN PHARMACEUTICAL INDUSTRY COMPANY LIMITED - (BRANCH) | 661748158 | manufacture(72430-002) | |