JARDIANCE- empagliflozin tablet, film coated
Jardiance by
Drug Labeling and Warnings
Jardiance by is a Prescription medication manufactured, distributed, or labeled by Boehringer Ingelheim Pharmaceuticals, Inc., Boehringer Ingelheim Pharma GmbH and Co. KG, West-Ward Columbus Inc., Sixarp, LLC, Boehringer Ingelheim Promeco, S.A. de C.V., Bidachem S.p.a., Rottendorf Pharma GmbH, BOEHRINGER INGELHEIM HELLAS SINGLE MEMBER S.A., F.I.S. FABBRICA ITALIANA SINTETICI S.P.A.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use JARDIANCE safely and effectively. See full prescribing information for JARDIANCE.
JARDIANCE® (empagliflozin) tablets, for oral use
Initial U.S. Approval: 2014RECENT MAJOR CHANGES
Warnings and Precautions, Ketoacidosis (5.2) 1/2020 INDICATIONS AND USAGE
JARDIANCE is a sodium-glucose co-transporter 2 (SGLT2) inhibitor indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus,
- to reduce the risk of cardiovascular death in adult patients with type 2 diabetes mellitus and established cardiovascular disease. (1)
Limitations of Use: Not for the treatment of type 1 diabetes mellitus or diabetic ketoacidosis (1)
DOSAGE AND ADMINISTRATION
- The recommended dose of JARDIANCE is 10 mg once daily, taken in the morning, with or without food (2.1)
- Dose may be increased to 25 mg once daily (2.1)
- Assess renal function before initiating JARDIANCE. Do not initiate JARDIANCE if eGFR is below 45 mL/min/1.73 m2 (2.2)
- Discontinue JARDIANCE if eGFR falls persistently below 45 mL/min/1.73 m2 (2.2)
DOSAGE FORMS AND STRENGTHS
Tablets: 10 mg, 25 mg (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Hypotension: Before initiating JARDIANCE assess and correct volume status in patients with renal impairment, the elderly, in patients with low systolic blood pressure, and in patients on diuretics. Monitor for signs and symptoms during therapy. (5.1)
- Ketoacidosis: Assess patients who present with signs and symptoms of metabolic acidosis for ketoacidosis, regardless of blood glucose level. If suspected, discontinue JARDIANCE, evaluate and treat promptly. Before initiating JARDIANCE, consider risk factors for ketoacidosis. Patients on JARDIANCE may require monitoring and temporary discontinuation of therapy in clinical situations known to predispose to ketoacidosis. (5.2)
- Acute Kidney Injury and Impairment in Renal Function: Consider temporarily discontinuing in settings of reduced oral intake or fluid losses. If acute kidney injury occurs, discontinue and promptly treat. Monitor renal function during therapy. (5.3)
- Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated (5.4)
- Hypoglycemia: Consider lowering the dose of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating JARDIANCE (5.5)
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment. (5.6)
- Genital Mycotic Infections: Monitor and treat as appropriate (5.7)
- Hypersensitivity Reactions: Discontinue JARDIANCE, treat promptly, and monitor until signs and symptoms resolve (5.8)
- Increased LDL-C: Monitor and treat as appropriate (5.9)
ADVERSE REACTIONS
- The most common adverse reactions associated with JARDIANCE (5% or greater incidence) were urinary tract infections and female genital mycotic infections (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Boehringer Ingelheim Pharmaceuticals, Inc. at 1-800-542-6257 or 1-800-459-9906 TTY, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters (8.1)
- Lactation: JARDIANCE is not recommended when breastfeeding (8.2)
- Geriatric Patients: Higher incidence of adverse reactions related to volume depletion and reduced renal function (5.1, 5.3, 8.5)
- Patients with Renal Impairment: Higher incidence of adverse reactions related to reduced renal function (2.2, 5.3, 8.6)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 1/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Patients with Renal Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension
5.2 Ketoacidosis
5.3 Acute Kidney Injury and Impairment in Renal Function
5.4 Urosepsis and Pyelonephritis
5.5 Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues
5.6 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene)
5.7 Genital Mycotic Infections
5.8 Hypersensitivity Reactions
5.9 Increased Low-Density Lipoprotein Cholesterol (LDL-C)
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Diuretics
7.2 Insulin or Insulin Secretagogues
7.3 Positive Urine Glucose Test
7.4 Interference with 1,5-anhydroglucitol (1,5-AG) Assay
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Glycemic Control
14.2 Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dose of JARDIANCE is 10 mg once daily in the morning, taken with or without food. In patients tolerating JARDIANCE, the dose may be increased to 25 mg [see Clinical Studies (14)].
In patients with volume depletion, correcting this condition prior to initiation of JARDIANCE is recommended [see Warnings and Precautions (5.1), Use in Specific Populations (8.5) and Patient Counseling Information (17)].
2.2 Patients with Renal Impairment
Assessment of renal function is recommended prior to initiation of JARDIANCE and periodically thereafter.
JARDIANCE should not be initiated in patients with an eGFR less than 45 mL/min/1.73 m2.
No dose adjustment is needed in patients with an eGFR greater than or equal to 45 mL/min/1.73 m2.
JARDIANCE should be discontinued if eGFR is persistently less than 45 mL/min/1.73 m2[see Warnings and Precautions (5.1, 5.3) and Use in Specific Populations (8.6)].
-
3 DOSAGE FORMS AND STRENGTHS
JARDIANCE tablets available as:
- 10 mg pale yellow, round, biconvex and bevel-edged, film-coated tablets debossed with "S 10" on one side and the Boehringer Ingelheim company symbol on the other side.
- 25 mg pale yellow, oval, biconvex, film-coated tablets debossed with "S 25" on one side and the Boehringer Ingelheim company symbol on the other side.
-
4 CONTRAINDICATIONS
- History of serious hypersensitivity reaction to empagliflozin or any of the excipients in JARDIANCE [see Warnings and Precautions (5.8)].
- Severe renal impairment, end-stage renal disease, or dialysis [see Use in Specific Populations (8.6)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension
JARDIANCE causes intravascular volume contraction. Symptomatic hypotension may occur after initiating JARDIANCE [see Adverse Reactions (6.1)] particularly in patients with renal impairment, the elderly, in patients with low systolic blood pressure, and in patients on diuretics. Before initiating JARDIANCE, assess for volume contraction and correct volume status if indicated. Monitor for signs and symptoms of hypotension after initiating therapy and increase monitoring in clinical situations where volume contraction is expected [see Use in Specific Populations (8.5)].
5.2 Ketoacidosis
Reports of ketoacidosis, a serious life-threatening condition requiring urgent hospitalization have been identified in postmarketing surveillance in patients with type 1 and type 2 diabetes mellitus receiving sodium glucose co-transporter-2 (SGLT2) inhibitors, including JARDIANCE. Fatal cases of ketoacidosis have been reported in patients taking JARDIANCE. JARDIANCE is not indicated for the treatment of patients with type 1 diabetes mellitus [see Indications and Usage (1)].
Patients treated with JARDIANCE who present with signs and symptoms consistent with severe metabolic acidosis should be assessed for ketoacidosis regardless of presenting blood glucose levels, as ketoacidosis associated with JARDIANCE may be present even if blood glucose levels are less than 250 mg/dL. If ketoacidosis is suspected, JARDIANCE should be discontinued, patient should be evaluated, and prompt treatment should be instituted. Treatment of ketoacidosis may require insulin, fluid and carbohydrate replacement.
In many of the postmarketing reports, and particularly in patients with type 1 diabetes, the presence of ketoacidosis was not immediately recognized and institution of treatment was delayed because presenting blood glucose levels were below those typically expected for diabetic ketoacidosis (often less than 250 mg/dL). Signs and symptoms at presentation were consistent with dehydration and severe metabolic acidosis and included nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. In some but not all cases, factors predisposing to ketoacidosis such as insulin dose reduction, acute febrile illness, reduced caloric intake, surgery, pancreatic disorders suggesting insulin deficiency (e.g., type 1 diabetes, history of pancreatitis or pancreatic surgery), and alcohol abuse were identified.
Before initiating JARDIANCE, consider factors in the patient history that may predispose to ketoacidosis including pancreatic insulin deficiency from any cause, caloric restriction, and alcohol abuse.
For patients who undergo scheduled surgery, consider temporarily discontinuing JARDIANCE for at least 3 days prior to surgery [see Clinical Pharmacology (12.2, 12.3)].
Consider monitoring for ketoacidosis and temporarily discontinuing JARDIANCE in other clinical situations known to predispose to ketoacidosis (e.g., prolonged fasting due to acute illness or post-surgery). Ensure risk factors for ketoacidosis are resolved prior to restarting JARDIANCE.
Educate patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue JARDIANCE and seek medical attention immediately if signs and symptoms occur.
5.3 Acute Kidney Injury and Impairment in Renal Function
JARDIANCE causes intravascular volume contraction [see Warnings and Precautions (5.1)] and can cause renal impairment [see Adverse Reactions (6.1)]. There have been postmarketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients receiving SGLT2 inhibitors, including JARDIANCE; some reports involved patients younger than 65 years of age.
Before initiating JARDIANCE, consider factors that may predispose patients to acute kidney injury including hypovolemia, chronic renal insufficiency, congestive heart failure and concomitant medications (diuretics, ACE inhibitors, ARBs, NSAIDs). Consider temporarily discontinuing JARDIANCE in any setting of reduced oral intake (such as acute illness or fasting) or fluid losses (such as gastrointestinal illness or excessive heat exposure); monitor patients for signs and symptoms of acute kidney injury. If acute kidney injury occurs, discontinue JARDIANCE promptly and institute treatment.
JARDIANCE increases serum creatinine and decreases eGFR. Patients with hypovolemia may be more susceptible to these changes. Renal function abnormalities can occur after initiating JARDIANCE [see Adverse Reactions (6.1)]. Renal function should be evaluated prior to initiation of JARDIANCE and monitored periodically thereafter. More frequent renal function monitoring is recommended in patients with an eGFR below 60 mL/min/1.73 m2. Use of JARDIANCE is not recommended when eGFR is persistently less than 45 mL/min/1.73 m2 and is contraindicated in patients with an eGFR less than 30 mL/min/1.73 m2 [see Dosage and Administration (2.2), Contraindications (4) and Use in Specific Populations (8.6)].
5.4 Urosepsis and Pyelonephritis
There have been postmarketing reports of serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization in patients receiving SGLT2 inhibitors, including JARDIANCE. Treatment with SGLT2 inhibitors increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6)].
5.5 Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues
Insulin and insulin secretagogues are known to cause hypoglycemia. The risk of hypoglycemia is increased when JARDIANCE is used in combination with insulin secretagogues (e.g., sulfonylurea) or insulin [see Adverse Reactions (6.1)]. Therefore, a lower dose of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia when used in combination with JARDIANCE.
5.6 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene)
Reports of necrotizing fasciitis of the perineum (Fournier's gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including JARDIANCE. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with JARDIANCE presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue JARDIANCE, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
5.7 Genital Mycotic Infections
JARDIANCE increases the risk for genital mycotic infections [see Adverse Reactions (6.1)]. Patients with a history of chronic or recurrent genital mycotic infections were more likely to develop genital mycotic infections. Monitor and treat as appropriate.
5.8 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions, (e.g., angioedema) in patients treated with JARDIANCE. If a hypersensitivity reaction occurs, discontinue JARDIANCE; treat promptly per standard of care, and monitor until signs and symptoms resolve. JARDIANCE is contraindicated in patients with a previous serious hypersensitivity reaction to empagliflozin or any of the excipients in JARDIANCE [see Contraindications (4)].
5.9 Increased Low-Density Lipoprotein Cholesterol (LDL-C)
Increases in LDL-C can occur with JARDIANCE [see Adverse Reactions (6.1)]. Monitor and treat as appropriate.
-
6 ADVERSE REACTIONS
The following important adverse reactions are described below and elsewhere in the labeling:
- Hypotension [see Warnings and Precautions (5.1)]
- Ketoacidosis [see Warnings and Precautions (5.2)]
- Acute Kidney Injury and Impairment in Renal Function [see Warnings and Precautions (5.3)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.6)]
- Genital Mycotic Infections [see Warnings and Precautions (5.7)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.8)]
- Increased Low-Density Lipoprotein Cholesterol (LDL-C) [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Pool of Placebo-Controlled Trials evaluating JARDIANCE 10 and 25 mg
The data in Table 1 are derived from a pool of four 24-week placebo-controlled trials and 18-week data from a placebo-controlled trial with insulin. JARDIANCE was used as monotherapy in one trial and as add-on therapy in four trials [see Clinical Studies (14)].
These data reflect exposure of 1976 patients to JARDIANCE with a mean exposure duration of approximately 23 weeks. Patients received placebo (N=995), JARDIANCE 10 mg (N=999), or JARDIANCE 25 mg (N=977) once daily. The mean age of the population was 56 years and 3% were older than 75 years of age. More than half (55%) of the population was male; 46% were White, 50% were Asian, and 3% were Black or African American. At baseline, 57% of the population had diabetes more than 5 years and had a mean hemoglobin A1c (HbA1c) of 8%. Established microvascular complications of diabetes at baseline included diabetic nephropathy (7%), retinopathy (8%), or neuropathy (16%). Baseline renal function was normal or mildly impaired in 91% of patients and moderately impaired in 9% of patients (mean eGFR 86.8 mL/min/1.73 m2).
Table 1 shows common adverse reactions (excluding hypoglycemia) associated with the use of JARDIANCE. The adverse reactions were not present at baseline, occurred more commonly on JARDIANCE than on placebo and occurred in greater than or equal to 2% of patients treated with JARDIANCE 10 mg or JARDIANCE 25 mg.
Table 1 Adverse Reactions Reported in ≥2% of Patients Treated with JARDIANCE and Greater than Placebo in Pooled Placebo-Controlled Clinical Studies of JARDIANCE Monotherapy or Combination Therapy Number (%) of Patients Placebo
N=995JARDIANCE 10 mg
N=999JARDIANCE 25 mg
N=977aPredefined adverse event grouping, including, but not limited to, urinary tract infection, asymptomatic bacteriuria, cystitis
bFemale genital mycotic infections include the following adverse reactions: vulvovaginal mycotic infection, vaginal infection, vulvitis, vulvovaginal candidiasis, genital infection, genital candidiasis, genital infection fungal, genitourinary tract infection, vulvovaginitis, cervicitis, urogenital infection fungal, vaginitis bacterial. Percentages calculated with the number of female subjects in each group as denominator: placebo (N=481), JARDIANCE 10 mg (N=443), JARDIANCE 25 mg (N=420).
cPredefined adverse event grouping, including, but not limited to, polyuria, pollakiuria, and nocturia
dMale genital mycotic infections include the following adverse reactions: balanoposthitis, balanitis, genital infections fungal, genitourinary tract infection, balanitis candida, scrotal abscess, penile infection. Percentages calculated with the number of male subjects in each group as denominator: placebo (N=514), JARDIANCE 10 mg (N=556), JARDIANCE 25 mg (N=557).Urinary tract infectiona 7.6% 9.3% 7.6% Female genital mycotic infectionsb 1.5% 5.4% 6.4% Upper respiratory tract infection 3.8% 3.1% 4.0% Increased urinationc 1.0% 3.4% 3.2% Dyslipidemia 3.4% 3.9% 2.9% Arthralgia 2.2% 2.4% 2.3% Male genital mycotic infectionsd 0.4% 3.1% 1.6% Nausea 1.4% 2.3% 1.1% Thirst (including polydipsia) was reported in 0%, 1.7%, and 1.5% for placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively.
Volume Depletion
JARDIANCE causes an osmotic diuresis, which may lead to intravascular volume contraction and adverse reactions related to volume depletion. In the pool of five placebo-controlled clinical trials, adverse reactions related to volume depletion (e.g., blood pressure (ambulatory) decreased, blood pressure systolic decreased, dehydration, hypotension, hypovolemia, orthostatic hypotension, and syncope) were reported by 0.3%, 0.5%, and 0.3% of patients treated with placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively. JARDIANCE may increase the risk of hypotension in patients at risk for volume contraction [see Warnings and Precautions (5.1) and Use in Specific Populations (8.5, 8.6)].
Increased Urination
In the pool of five placebo-controlled clinical trials, adverse reactions of increased urination (e.g., polyuria, pollakiuria, and nocturia) occurred more frequently on JARDIANCE than on placebo (see Table 1). Specifically, nocturia was reported by 0.4%, 0.3%, and 0.8% of patients treated with placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively.
Acute Impairment in Renal Function
Treatment with JARDIANCE was associated with increases in serum creatinine and decreases in eGFR (see Table 2). Patients with moderate renal impairment at baseline had larger mean changes [see Warnings and Precautions (5.3) and Use in Specific Populations (8.5, 8.6)].
In a long-term cardiovascular outcome trial, the acute impairment in renal function was observed to reverse after treatment discontinuation suggesting acute hemodynamic changes play a role in the renal function changes observed with empagliflozin.
Table 2 Changes from Baseline in Serum Creatinine and eGFRa in the Pool of Four 24-week Placebo-Controlled Studies and Renal Impairment Study aObserved cases on treatment.
bSubset of patients from renal impairment study with eGFR 30 to less than 60 mL/min/1.73 m2
cApproximately 3 weeks after end of treatment.Pool of 24-Week Placebo-Controlled Studies Placebo JARDIANCE 10 mg JARDIANCE 25 mg Baseline Mean N 825 830 822 Creatinine (mg/dL) 0.84 0.85 0.85 eGFR (mL/min/1.73 m2) 87.3 87.1 87.8 Week 12 Change N 771 797 783 Creatinine (mg/dL) 0.00 0.02 0.01 eGFR (mL/min/1.73 m2) -0.3 -1.3 -1.4 Week 24 Change N 708 769 754 Creatinine (mg/dL) 0.00 0.01 0.01 eGFR (mL/min/1.73 m2) -0.3 -0.6 -1.4 Moderate Renal Impairmentb Placebo JARDIANCE 25 mg Baseline Mean N 187 -- 187 Creatinine (mg/dL) 1.49 -- 1.46 eGFR (mL/min/1.73 m2) 44.3 -- 45.4 Week 12 Change N 176 -- 179 Creatinine (mg/dL) 0.01 -- 0.12 eGFR (mL/min/1.73 m2) 0.1 -- -3.8 Week 24 Change N 170 -- 171 Creatinine (mg/dL) 0.01 -- 0.10 eGFR (mL/min/1.73 m2) 0.2 -- -3.2 Week 52 Change N 164 -- 162 Creatinine (mg/dL) 0.02 -- 0.11 eGFR (mL/min/1.73 m2) -0.3 -- -2.8 Post-treatment Changec N 98 -- 103 Creatinine (mg/dL) 0.03 -- 0.02 eGFR (mL/min/1.73 m2) 0.16 -- 1.48 Hypoglycemia
The incidence of hypoglycemia by study is shown in Table 3. The incidence of hypoglycemia increased when JARDIANCE was administered with insulin or sulfonylurea [see Warnings and Precautions (5.5)].
Table 3 Incidence of Overalla and Severeb Hypoglycemic Events in Placebo-Controlled Clinical Studiesc aOverall hypoglycemic events: plasma or capillary glucose of less than or equal to 70 mg/dL
bSevere hypoglycemic events: requiring assistance regardless of blood glucose
cTreated set (patients who had received at least one dose of study drug)
dInsulin dose could not be adjusted during the initial 18 week treatment periodMonotherapy (24 weeks) Placebo (n=229) JARDIANCE 10 mg (n=224) JARDIANCE 25 mg (n=223) Overall (%) 0.4% 0.4% 0.4% Severe (%) 0% 0% 0% In Combination with Metformin (24 weeks) Placebo + Metformin
(n=206)JARDIANCE 10 mg + Metformin
(n=217)JARDIANCE 25 mg + Metformin
(n=214)Overall (%) 0.5% 1.8% 1.4% Severe (%) 0% 0% 0% In Combination with Metformin + Sulfonylurea
(24 weeks)Placebo (n=225) JARDIANCE 10 mg + Metformin + Sulfonylurea (n=224) JARDIANCE 25 mg + Metformin + Sulfonylurea (n=217) Overall (%) 8.4% 16.1% 11.5% Severe (%) 0% 0% 0% In Combination with Pioglitazone +/- Metformin
(24 weeks)Placebo (n=165) JARDIANCE 10 mg + Pioglitazone +/- Metformin
(n=165)JARDIANCE 25 mg + Pioglitazone +/- Metformin
(n=168)Overall (%) 1.8% 1.2% 2.4% Severe (%) 0% 0% 0% In Combination with Basal Insulin +/- Metformin
(18 weeksd)Placebo (n=170) JARDIANCE 10 mg
(n=169)JARDIANCE 25 mg
(n=155)Overall (%) 20.6% 19.5% 28.4% Severe (%) 0% 0% 1.3% In Combination with MDI Insulin +/-Metformin
(18 weeksd)Placebo (n=188) JARDIANCE 10 mg
(n=186)JARDIANCE 25 mg
(n=189)Overall (%) 37.2% 39.8% 41.3% Severe (%) 0.5% 0.5% 0.5% Genital Mycotic Infections
In the pool of five placebo-controlled clinical trials, the incidence of genital mycotic infections (e.g., vaginal mycotic infection, vaginal infection, genital infection fungal, vulvovaginal candidiasis, and vulvitis) was increased in patients treated with JARDIANCE compared to placebo, occurring in 0.9%, 4.1%, and 3.7% of patients randomized to placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively. Discontinuation from study due to genital infection occurred in 0% of placebo-treated patients and 0.2% of patients treated with either JARDIANCE 10 or 25 mg.
Genital mycotic infections occurred more frequently in female than male patients (see Table 1).
Phimosis occurred more frequently in male patients treated with JARDIANCE 10 mg (less than 0.1%) and JARDIANCE 25 mg (0.1%) than placebo (0%).
Urinary Tract Infections
In the pool of five placebo-controlled clinical trials, the incidence of urinary tract infections (e.g., urinary tract infection, asymptomatic bacteriuria, and cystitis) was increased in patients treated with JARDIANCE compared to placebo (see Table 1). Patients with a history of chronic or recurrent urinary tract infections were more likely to experience a urinary tract infection. The rate of treatment discontinuation due to urinary tract infections was 0.1%, 0.2%, and 0.1% for placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively.
Urinary tract infections occurred more frequently in female patients. The incidence of urinary tract infections in female patients randomized to placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg was 16.6%, 18.4%, and 17.0%, respectively. The incidence of urinary tract infections in male patients randomized to placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg was 3.2%, 3.6%, and 4.1%, respectively [see Warnings and Precautions (5.4) and Use in Specific Populations (8.5)].
Laboratory Tests
Increase in Low-Density Lipoprotein Cholesterol (LDL-C)
Dose-related increases in low-density lipoprotein cholesterol (LDL-C) were observed in patients treated with JARDIANCE. LDL-C increased by 2.3%, 4.6%, and 6.5% in patients treated with placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively [see Warnings and Precautions (5.9)]. The range of mean baseline LDL-C levels was 90.3 to 90.6 mg/dL across treatment groups.
Increase in Hematocrit
In a pool of four placebo-controlled studies, median hematocrit decreased by 1.3% in placebo and increased by 2.8% in JARDIANCE 10 mg and 2.8% in JARDIANCE 25 mg treated patients. At the end of treatment, 0.6%, 2.7%, and 3.5% of patients with hematocrits initially within the reference range had values above the upper limit of the reference range with placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of JARDIANCE. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Ketoacidosis [see Warnings and Precautions (5.2)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Necrotizing Fasciitis of the Perineum (Fournier's gangrene) [see Warnings and Precautions (5.6)]
- Angioedema [see Warnings and Precautions (5.8)]
- Skin Reactions (e.g., rash, urticaria)
-
7 DRUG INTERACTIONS
7.1 Diuretics
Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion [see Warnings and Precautions (5.1)].
7.2 Insulin or Insulin Secretagogues
Coadministration of empagliflozin with insulin or insulin secretagogues increases the risk for hypoglycemia [see Warnings and Precautions (5.5)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on animal data showing adverse renal effects, JARDIANCE is not recommended during the second and third trimesters of pregnancy.
Limited data available with JARDIANCE in pregnant women are not sufficient to determine a drug-associated risk for major birth defects and miscarriage. There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy [see Clinical Considerations].
In animal studies, adverse renal changes were observed in rats when empagliflozin was administered during a period of renal development corresponding to the late second and third trimesters of human pregnancy. Doses approximately 13-times the maximum clinical dose caused renal pelvic and tubule dilatations that were reversible. Empagliflozin was not teratogenic in rats and rabbits up to 300 mg/kg/day, which approximates 48-times and 128-times, respectively, the maximum clinical dose of 25 mg when administered during organogenesis [see Data].
The estimated background risk of major birth defects is 6-10% in women with pre-gestational diabetes with a HbA1c >7 and has been reported to be as high as 20-25% in women with HbA1c >10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk: Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, stillbirth, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
Data
Animal Data
Empagliflozin dosed directly to juvenile rats from postnatal day (PND) 21 until PND 90 at doses of 1, 10, 30 and 100 mg/kg/day caused increased kidney weights and renal tubular and pelvic dilatation at 100 mg/kg/day, which approximates 13-times the maximum clinical dose of 25 mg, based on AUC. These findings were not observed after a 13 week drug-free recovery period. These outcomes occurred with drug exposure during periods of renal development in rats that correspond to the late second and third trimester of human renal development.
In embryo-fetal development studies in rats and rabbits, empagliflozin was administered for intervals coinciding with the first trimester period of organogenesis in humans. Doses up to 300 mg/kg/day, which approximates 48-times (rats) and 128-times (rabbits) the maximum clinical dose of 25 mg (based on AUC), did not result in adverse developmental effects. In rats, at higher doses of empagliflozin causing maternal toxicity, malformations of limb bones increased in fetuses at 700 mg/kg/day or 154-times the 25 mg maximum clinical dose. Empagliflozin crosses the placenta and reaches fetal tissues in rats. In the rabbit, higher doses of empagliflozin resulted in maternal and fetal toxicity at 700 mg/kg/day, or 139-times the 25 mg maximum clinical dose.
In pre- and postnatal development studies in pregnant rats, empagliflozin was administered from gestation day 6 through to lactation day 20 (weaning) at up to 100 mg/kg/day (approximately 16 times the 25 mg maximum clinical dose) without maternal toxicity. Reduced body weight was observed in the offspring at greater than or equal to 30 mg/kg/day (approximately 4 times the 25 mg maximum clinical dose).
8.2 Lactation
Risk Summary
There is no information regarding the presence of JARDIANCE in human milk, the effects of JARDIANCE on the breastfed infant or the effects on milk production. Empagliflozin is present in the milk of lactating rats [see Data]. Since human kidney maturation occurs in utero and during the first 2 years of life when lactational exposure may occur, there may be risk to the developing human kidney.
Because of the potential for serious adverse reactions in a breastfed infant, including the potential for empagliflozin to affect postnatal renal development, advise women that use of JARDIANCE is not recommended while breastfeeding.
Data
Empagliflozin was present at a low level in rat fetal tissues after a single oral dose to the dams at gestation day 18. In rat milk, the mean milk to plasma ratio ranged from 0.634 -5, and was greater than one from 2 to 24 hours post-dose. The mean maximal milk to plasma ratio of 5 occurred at 8 hours post-dose, suggesting accumulation of empagliflozin in the milk. Juvenile rats directly exposed to empagliflozin showed a risk to the developing kidney (renal pelvic and tubular dilatations) during maturation.
8.4 Pediatric Use
The safety and effectiveness of JARDIANCE in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
No JARDIANCE dosage change is recommended based on age [see Dosage and Administration (2)]. In studies assessing the efficacy of empagliflozin in improving glycemic control in patients with type 2 diabetes, a total of 2721 (32%) patients treated with empagliflozin were 65 years of age and older, and 491 (6%) were 75 years of age and older. JARDIANCE is expected to have diminished glycemic efficacy in elderly patients with renal impairment [see Use in Specific Populations (8.6)]. The risk of volume depletion-related adverse reactions increased in patients who were 75 years of age and older to 2.1%, 2.3%, and 4.4% for placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg. The risk of urinary tract infections increased in patients who were 75 years of age and older to 10.5%, 15.7%, and 15.1% in patients randomized to placebo, JARDIANCE 10 mg, and JARDIANCE 25 mg, respectively [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
8.6 Renal Impairment
The efficacy and safety of JARDIANCE were evaluated in a study of patients with mild and moderate renal impairment [see Clinical Studies (14.1)]. In this study, 195 patients exposed to JARDIANCE had an eGFR between 60 and 90 mL/min/1.73 m2, 91 patients exposed to JARDIANCE had an eGFR between 45 and 60 mL/min/1.73 m2 and 97 patients exposed to JARDIANCE had an eGFR between 30 and 45 mL/min/1.73 m2. The glucose lowering benefit of JARDIANCE 25 mg decreased in patients with worsening renal function. The risks of renal impairment [see Warnings and Precautions (5.3)], volume depletion adverse reactions and urinary tract infection-related adverse reactions increased with worsening renal function.
In a large cardiovascular outcomes study, there were 1819 patients with eGFR below 60 mL/min/1.73 m2. The cardiovascular death findings in this subgroup were consistent with the overall findings [see Clinical Studies (14.2)].
The efficacy and safety of JARDIANCE have not been established in patients with severe renal impairment, with ESRD, or receiving dialysis. JARDIANCE is not expected to be effective in these patient populations [see Dosage and Administration (2.2), Contraindications (4) and Warnings and Precautions (5.1, 5.3)].
8.7 Hepatic Impairment
JARDIANCE may be used in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
In the event of an overdose with JARDIANCE, contact the Poison Control Center. Employ the usual supportive measures (e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring, and institute supportive treatment) as dictated by the patient's clinical status. Removal of empagliflozin by hemodialysis has not been studied.
-
11 DESCRIPTION
JARDIANCE tablets contain empagliflozin, an orally-active inhibitor of the sodium-glucose co-transporter 2 (SGLT2).
The chemical name of empagliflozin is D-Glucitol,1,5-anhydro-1-C-[4-chloro-3-[[4-[[(3S)-tetrahydro-3-furanyl]oxy]phenyl]methyl]phenyl]-, (1S).
Its molecular formula is C23H27ClO7 and the molecular weight is 450.91. The structural formula is:
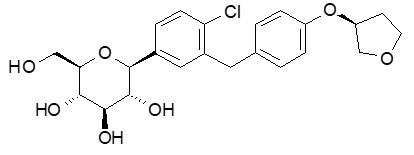
Empagliflozin is a white to yellowish, non-hygroscopic powder. It is very slightly soluble in water, sparingly soluble in methanol, slightly soluble in ethanol and acetonitrile; soluble in 50% acetonitrile/water; and practically insoluble in toluene.
Each film-coated tablet of JARDIANCE contains 10 mg or 25 mg of empagliflozin (free base) and the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, hydroxypropyl cellulose, croscarmellose sodium, colloidal silicon dioxide and magnesium stearate. In addition, the film coating contains the following inactive ingredients: hypromellose, titanium dioxide, talc, polyethylene glycol, and yellow ferric oxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Sodium-glucose co-transporter 2 (SGLT2) is the predominant transporter responsible for reabsorption of glucose from the glomerular filtrate back into the circulation. Empagliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, empagliflozin reduces renal reabsorption of filtered glucose and lowers the renal threshold for glucose, and thereby increases urinary glucose excretion.
12.2 Pharmacodynamics
Urinary Glucose Excretion
In patients with type 2 diabetes, urinary glucose excretion increased immediately following a dose of JARDIANCE and was maintained at the end of a 4-week treatment period averaging at approximately 64 grams per day with 10 mg empagliflozin and 78 grams per day with 25 mg JARDIANCE once daily [see Clinical Studies (14)]. Data from single oral doses of empagliflozin in healthy subjects indicate that, on average, the elevation in urinary glucose excretion approaches baseline by about 3 days for the 10 mg and 25 mg doses.
Urinary Volume
In a 5-day study, mean 24-hour urine volume increase from baseline was 341 mL on Day 1 and 135 mL on Day 5 of empagliflozin 25 mg once daily treatment.
Cardiac Electrophysiology
In a randomized, placebo-controlled, active-comparator, crossover study, 30 healthy subjects were administered a single oral dose of JARDIANCE 25 mg, JARDIANCE 200 mg (8 times the maximum dose), moxifloxacin, and placebo. No increase in QTc was observed with either 25 mg or 200 mg empagliflozin.
12.3 Pharmacokinetics
Absorption
The pharmacokinetics of empagliflozin has been characterized in healthy volunteers and patients with type 2 diabetes and no clinically relevant differences were noted between the two populations. After oral administration, peak plasma concentrations of empagliflozin were reached at 1.5 hours post-dose. Thereafter, plasma concentrations declined in a biphasic manner with a rapid distribution phase and a relatively slow terminal phase. The steady state mean plasma AUC and Cmax were 1870 nmol∙h/L and 259 nmol/L, respectively, with 10 mg empagliflozin once daily treatment, and 4740 nmol∙h/L and 687 nmol/L, respectively, with 25 mg empagliflozin once daily treatment. Systemic exposure of empagliflozin increased in a dose-proportional manner in the therapeutic dose range. The single-dose and steady-state pharmacokinetic parameters of empagliflozin were similar, suggesting linear pharmacokinetics with respect to time.
Administration of 25 mg empagliflozin after intake of a high-fat and high-calorie meal resulted in slightly lower exposure; AUC decreased by approximately 16% and Cmax decreased by approximately 37%, compared to fasted condition. The observed effect of food on empagliflozin pharmacokinetics was not considered clinically relevant and empagliflozin may be administered with or without food.
Distribution
The apparent steady-state volume of distribution was estimated to be 73.8 L based on a population pharmacokinetic analysis. Following administration of an oral [14C]-empagliflozin solution to healthy subjects, the red blood cell partitioning was approximately 36.8% and plasma protein binding was 86.2%.
Metabolism
No major metabolites of empagliflozin were detected in human plasma and the most abundant metabolites were three glucuronide conjugates (2-O-, 3-O-, and 6-O-glucuronide). Systemic exposure of each metabolite was less than 10% of total drug-related material. In vitro studies suggested that the primary route of metabolism of empagliflozin in humans is glucuronidation by the uridine 5'-diphospho-glucuronosyltransferases UGT2B7, UGT1A3, UGT1A8, and UGT1A9.
Elimination
The apparent terminal elimination half-life of empagliflozin was estimated to be 12.4 h and apparent oral clearance was 10.6 L/h based on the population pharmacokinetic analysis. Following once-daily dosing, up to 22% accumulation, with respect to plasma AUC, was observed at steady-state, which was consistent with empagliflozin half-life. Following administration of an oral [14C]-empagliflozin solution to healthy subjects, approximately 95.6% of the drug-related radioactivity was eliminated in feces (41.2%) or urine (54.4%). The majority of drug-related radioactivity recovered in feces was unchanged parent drug and approximately half of drug-related radioactivity excreted in urine was unchanged parent drug.
Specific Populations
Renal Impairment
In patients with mild (eGFR: 60 to less than 90 mL/min/1.73 m2), moderate (eGFR: 30 to less than 60 mL/min/1.73 m2), and severe (eGFR: less than 30 mL/min/1.73 m2) renal impairment and subjects with kidney failure/end stage renal disease (ESRD) patients, AUC of empagliflozin increased by approximately 18%, 20%, 66%, and 48%, respectively, compared to subjects with normal renal function. Peak plasma levels of empagliflozin were similar in subjects with moderate renal impairment and kidney failure/ESRD compared to patients with normal renal function. Peak plasma levels of empagliflozin were roughly 20% higher in subjects with mild and severe renal impairment as compared to subjects with normal renal function. Population pharmacokinetic analysis showed that the apparent oral clearance of empagliflozin decreased, with a decrease in eGFR leading to an increase in drug exposure. However, the fraction of empagliflozin that was excreted unchanged in urine, and urinary glucose excretion, declined with decrease in eGFR.
Hepatic Impairment
In subjects with mild, moderate, and severe hepatic impairment according to the Child-Pugh classification, AUC of empagliflozin increased by approximately 23%, 47%, and 75%, and Cmax increased by approximately 4%, 23%, and 48%, respectively, compared to subjects with normal hepatic function.
Effects of Age, Body Mass Index, Gender, and Race
Based on the population PK analysis, age, body mass index (BMI), gender and race (Asians versus primarily Whites) do not have a clinically meaningful effect on pharmacokinetics of empagliflozin [see Use in Specific Populations (8.5)].
Drug Interactions
In vitro Assessment of Drug Interactions
Empagliflozin does not inhibit, inactivate, or induce CYP450 isoforms. In vitro data suggest that the primary route of metabolism of empagliflozin in humans is glucuronidation by the uridine 5'-diphospho-glucuronosyltransferases UGT1A3, UGT1A8, UGT1A9, and UGT2B7. Empagliflozin does not inhibit UGT1A1, UGT1A3, UGT1A8, UGT1A9, or UGT2B7. Therefore, no effect of empagliflozin is anticipated on concomitantly administered drugs that are substrates of the major CYP450 isoforms or UGT1A1, UGT1A3, UGT1A8, UGT1A9, or UGT2B7. The effect of UGT induction (e.g., induction by rifampicin or any other UGT enzyme inducer) on empagliflozin exposure has not been evaluated.
Empagliflozin is a substrate for P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP), but it does not inhibit these efflux transporters at therapeutic doses. Based on in vitro studies, empagliflozin is considered unlikely to cause interactions with drugs that are P-gp substrates. Empagliflozin is a substrate of the human uptake transporters OAT3, OATP1B1, and OATP1B3, but not OAT1 and OCT2. Empagliflozin does not inhibit any of these human uptake transporters at clinically relevant plasma concentrations and, therefore, no effect of empagliflozin is anticipated on concomitantly administered drugs that are substrates of these uptake transporters.
In vivo Assessment of Drug Interactions
No dose adjustment of JARDIANCE is recommended when coadministered with commonly prescribed medicinal products based on results of the described pharmacokinetic studies. Empagliflozin pharmacokinetics were similar with and without coadministration of metformin, glimepiride, pioglitazone, sitagliptin, linagliptin, warfarin, verapamil, ramipril, and simvastatin in healthy volunteers and with or without coadministration of hydrochlorothiazide and torsemide in patients with type 2 diabetes (see Figure 1). The observed increases in overall exposure (AUC) of empagliflozin following coadministration with gemfibrozil, rifampicin, or probenecid are not clinically relevant. In subjects with normal renal function, coadministration of empagliflozin with probenecid resulted in a 30% decrease in the fraction of empagliflozin excreted in urine without any effect on 24-hour urinary glucose excretion. The relevance of this observation to patients with renal impairment is unknown.
Figure 1 Effect of Various Medications on the Pharmacokinetics of Empagliflozin as Displayed as 90% Confidence Interval of Geometric Mean AUC and Cmax Ratios [reference lines indicate 100% (80% - 125%)] aempagliflozin, 50 mg, once daily; bempagliflozin, 25 mg, single dose; cempagliflozin, 25 mg, once daily; dempagliflozin, 10 mg, single dose 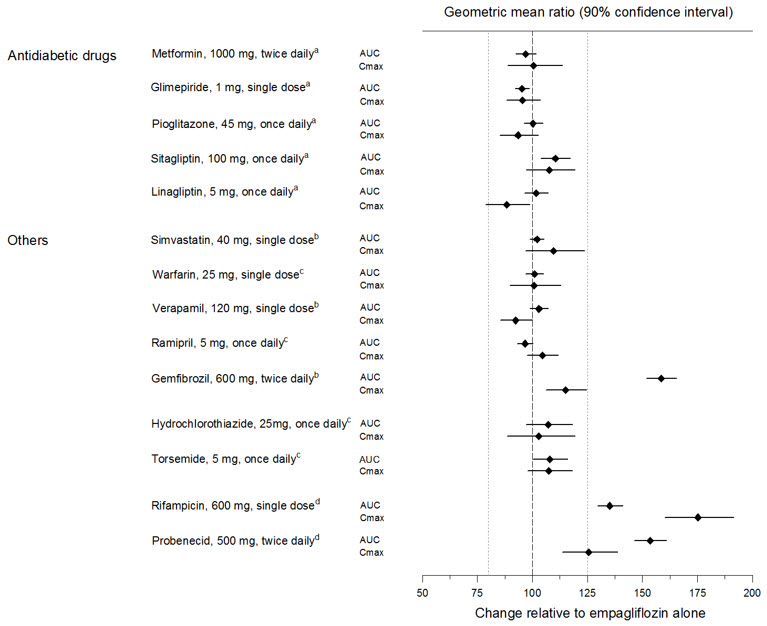
Empagliflozin had no clinically relevant effect on the pharmacokinetics of metformin, glimepiride, pioglitazone, sitagliptin, linagliptin, warfarin, digoxin, ramipril, simvastatin, hydrochlorothiazide, torsemide, and oral contraceptives when coadministered in healthy volunteers (see Figure 2).
Figure 2 Effect of Empagliflozin on the Pharmacokinetics of Various Medications as Displayed as 90% Confidence Interval of Geometric Mean AUC and Cmax Ratios [reference lines indicate 100% (80% - 125%)] aempagliflozin, 50 mg, once daily; bempagliflozin, 25 mg, once daily; cempagliflozin, 25 mg, single dose; dadministered as simvastatin; eadministered as warfarin racemic mixture; fadministered as Microgynon®; gadministered as ramipril 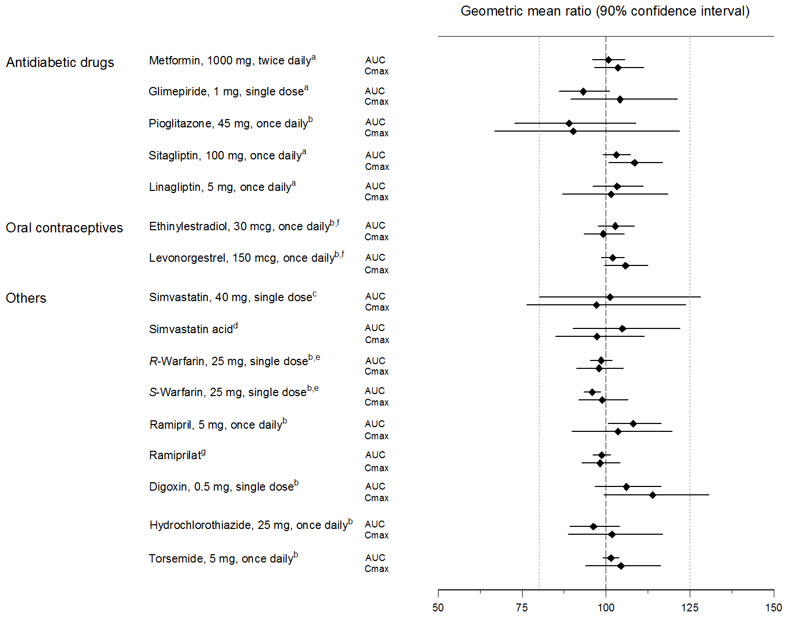
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenesis was evaluated in 2-year studies conducted in CD-1 mice and Wistar rats. Empagliflozin did not increase the incidence of tumors in female rats dosed at 100, 300, or 700 mg/kg/day (up to 72 times the exposure from the maximum clinical dose of 25 mg). In male rats, hemangiomas of the mesenteric lymph node were increased significantly at 700 mg/kg/day or approximately 42 times the exposure from a 25 mg clinical dose. Empagliflozin did not increase the incidence of tumors in female mice dosed at 100, 300, or 1000 mg/kg/day (up to 62 times the exposure from a 25 mg clinical dose). Renal tubule adenomas and carcinomas were observed in male mice at 1000 mg/kg/day, which is approximately 45 times the exposure of the maximum clinical dose of 25 mg. These tumors may be associated with a metabolic pathway predominantly present in the male mouse kidney.
-
14 CLINICAL STUDIES
14.1 Glycemic Control
JARDIANCE has been studied as monotherapy and in combination with metformin, sulfonylurea, pioglitazone, linagliptin, and insulin. JARDIANCE has also been studied in patients with type 2 diabetes with mild or moderate renal impairment.
In patients with type 2 diabetes, treatment with JARDIANCE reduced hemoglobin A1c (HbA1c), compared to placebo. The reduction in HbA1c for JARDIANCE compared with placebo was observed across subgroups including gender, race, geographic region, baseline BMI and duration of disease.
Monotherapy
A total of 986 patients with type 2 diabetes participated in a double-blind, placebo-controlled study to evaluate the efficacy and safety of JARDIANCE monotherapy.
Treatment-naïve patients with inadequately controlled type 2 diabetes entered an open-label placebo run-in for 2 weeks. At the end of the run-in period, patients who remained inadequately controlled and had an HbA1c between 7 and 10% were randomized to placebo, JARDIANCE 10 mg, JARDIANCE 25 mg, or a reference comparator.
At Week 24, treatment with JARDIANCE 10 mg or 25 mg daily provided statistically significant reductions in HbA1c (p-value <0.0001), fasting plasma glucose (FPG), and body weight compared with placebo (see Table 4 and Figure 3).
Table 4 Results at Week 24 From a Placebo-Controlled Monotherapy Study of JARDIANCE JARDIANCE 10 mg N=224 JARDIANCE 25 mg N=224 Placebo N=228 aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 9.4%, 9.4%, and 30.7% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively.
bANCOVA derived p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region. Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.)
cFPG (mg/dL); for JARDIANCE 10 mg, n=223, for JARDIANCE 25 mg, n=223, and for placebo, n=226HbA1c (%)a Baseline (mean) 7.9 7.9 7.9 Change from baseline (adjusted mean) -0.7 -0.8 0.1 Difference from placebo (adjusted mean) (97.5% CI) -0.7b (-0.9, -0.6) -0.9b (-1.0, -0.7) -- Patients [n (%)] achieving HbA1c <7% 72 (35%) 88 (44%) 25 (12%) FPG (mg/dL)c Baseline (mean) 153 153 155 Change from baseline (adjusted mean) -19 -25 12 Difference from placebo (adjusted mean) (95% CI) -31 (-37, -26) -36 (-42, -31) -- Body Weight Baseline (mean) in kg 78 78 78 % change from baseline (adjusted mean) -2.8 -3.2 -0.4 Difference from placebo (adjusted mean) (95% CI) -2.5b (-3.1, -1.9) -2.8b (-3.4, -2.2) -- Figure 3 Adjusted Mean HbA1c Change at Each Time Point (Completers) and at Week 24 (mITT Population) - LOCF 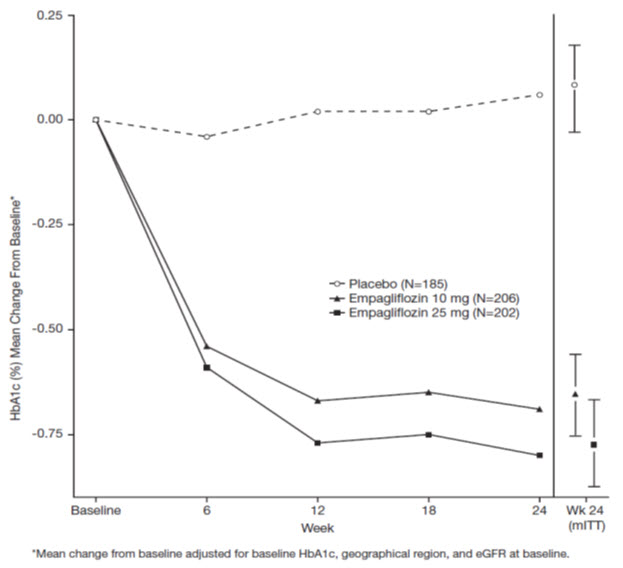
At Week 24, the systolic blood pressure was statistically significantly reduced compared to placebo by -2.6 mmHg (placebo-adjusted, p-value=0.0231) in patients randomized to 10 mg of JARDIANCE and by -3.4 mmHg (placebo-corrected, p-value=0.0028) in patients randomized to 25 mg of JARDIANCE.
Add-On Combination Therapy with Metformin
A total of 637 patients with type 2 diabetes participated in a double-blind, placebo-controlled study to evaluate the efficacy and safety of JARDIANCE in combination with metformin.
Patients with type 2 diabetes inadequately controlled on at least 1500 mg of metformin per day entered an open-label 2 week placebo run-in. At the end of the run-in period, patients who remained inadequately controlled and had an HbA1c between 7 and 10% were randomized to placebo, JARDIANCE 10 mg, or JARDIANCE 25 mg.
At Week 24, treatment with JARDIANCE 10 mg or 25 mg daily provided statistically significant reductions in HbA1c (p-value <0.0001), FPG, and body weight compared with placebo (see Table 5).
Table 5 Results at Week 24 From a Placebo-Controlled Study for JARDIANCE used in Combination with Metformin JARDIANCE
10 mg + Metformin
N=217JARDIANCE
25 mg + Metformin
N=213Placebo + Metformin
N=207aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 9.7%, 14.1%, and 24.6% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively.
bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region. Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.)
cFPG (mg/dL); for JARDIANCE 10 mg, n=216, for JARDIANCE 25 mg, n=213, and for placebo, n=207HbA1c (%)a Baseline (mean) 7.9 7.9 7.9 Change from baseline (adjusted mean) -0.7 -0.8 -0.1 Difference from placebo + metformin (adjusted mean) (95% CI) -0.6b (-0.7, -0.4) -0.6b (-0.8, -0.5) -- Patients [n (%)] achieving HbA1c <7% 75 (38%) 74 (39%) 23 (13%) FPG (mg/dL)c Baseline (mean) 155 149 156 Change from baseline (adjusted mean) -20 -22 6 Difference from placebo + metformin (adjusted mean) -26 -29 -- Body Weight Baseline mean in kg 82 82 80 % change from baseline (adjusted mean) -2.5 -2.9 -0.5 Difference from placebo (adjusted mean) (95% CI) -2.0b (-2.6, -1.4) -2.5b (-3.1, -1.9) -- At Week 24, the systolic blood pressure was statistically significantly reduced compared to placebo by -4.1 mmHg (placebo-corrected, p-value <0.0001) for JARDIANCE 10 mg and -4.8 mmHg (placebo-corrected, p-value <0.0001) for JARDIANCE 25 mg.
Initial Combination Therapy with Metformin
A total of 1364 patients with type 2 diabetes participated in a double-blind, randomized, active-controlled study to evaluate the efficacy and safety of JARDIANCE in combination with metformin as initial therapy compared to the corresponding individual components.
Treatment-naïve patients with inadequately controlled type 2 diabetes entered an open-label placebo run-in for 2 weeks. At the end of the run-in period, patients who remained inadequately controlled and had an HbA1c between 7 and 10.5% were randomized to one of 8 active-treatment arms: JARDIANCE 10 mg or 25 mg; metformin 1000 mg, or 2000 mg; JARDIANCE 10 mg in combination with 1000 mg or 2000 mg metformin; or JARDIANCE 25 mg in combination with 1000 mg or 2000 mg metformin.
At Week 24, initial therapy of JARDIANCE in combination with metformin provided statistically significant reductions in HbA1c (p-value <0.01) compared to the individual components (see Table 6).
Table 6 Glycemic Parameters at 24 Weeks in a Study Comparing JARDIANCE and Metformin to the Individual Components as Initial Therapy JARDIANCE
10 mg +
Metformin
1000 mga
N=161JARDIANCE
10 mg +
Metformin
2000 mga
N=167JARDIANCE
25 mg +
Metformin
1000 mga
N=165JARDIANCE
25 mg +
Metformin
2000 mga
N=169JARDIANCE
10 mg
N=169JARDIANCE
25 mg
N=163Metformin
1000 mga
N=167Metformin
2000 mga
N=162aMetformin total daily dose, administered in two equally divided doses per day.
bp-value ≤0.0062 (modified intent to treat population [observed case] MMRM model included treatment, renal function, region, visit, visit by treatment interaction, and baseline HbA1c).
cp-value ≤0.0056 (modified intent to treat population [observed case] MMRM model included treatment, renal function, region, visit, visit by treatment interaction, and baseline HbA1c).HbA1c (%) Baseline (mean) 8.7 8.7 8.8 8.7 8.6 8.9 8.7 8.6 Change from baseline (adjusted mean) -2.0 -2.1 -1.9 -2.1 -1.4 -1.4 -1.2 -1.8 Comparison vs JARDIANCE (adjusted mean) (95% CI) -0.6b
(-0.9, -0.4)-0.7b
(-1.0, -0.5)-0.6c
(-0.8, -0.3)-0.7c
(-1.0, -0.5)-- -- -- -- Comparison vs metformin (adjusted mean) (95% CI) -0.8b
(-1.0, -0.6)-0.3b
(-0.6, -0.1)-0.8c
(-1.0, -0.5)-0.3c
(-0.6, -0.1)-- -- -- -- Add-On Combination Therapy with Metformin and Sulfonylurea
A total of 666 patients with type 2 diabetes participated in a double-blind, placebo-controlled study to evaluate the efficacy and safety of JARDIANCE in combination with metformin plus a sulfonylurea.
Patients with inadequately controlled type 2 diabetes on at least 1500 mg per day of metformin and on a sulfonylurea, entered a 2 week open-label placebo run-in. At the end of the run-in, patients who remained inadequately controlled and had an HbA1c between 7% and 10% were randomized to placebo, JARDIANCE 10 mg, or JARDIANCE 25 mg.
Treatment with JARDIANCE 10 mg or 25 mg daily provided statistically significant reductions in HbA1c (p-value <0.0001), FPG, and body weight compared with placebo (see Table 7).
Table 7 Results at Week 24 from a Placebo-Controlled Study for JARDIANCE in Combination with Metformin and Sulfonylurea JARDIANCE
10 mg + Metformin
+ SU
N=225JARDIANCE
25 mg + Metformin
+ SU
N=216Placebo +
Metformin + SU
N=225aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 17.8%, 16.7%, and 25.3% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively.
bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region. Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.)
cFPG (mg/dL); for JARDIANCE 10 mg, n=225, for JARDIANCE 25 mg, n=215, for placebo, n=224HbA1c (%)a Baseline (mean) 8.1 8.1 8.2 Change from baseline (adjusted mean) -0.8 -0.8 -0.2 Difference from placebo (adjusted mean) (95% CI) -0.6b (-0.8, -0.5) -0.6b (-0.7, -0.4) -- Patients [n (%)] achieving HbA1c <7% 55 (26%) 65 (32%) 20 (9%) FPG (mg/dL)c Baseline (mean) 151 156 152 Change from baseline (adjusted mean) -23 -23 6 Difference from placebo (adjusted mean) -29 -29 -- Body Weight Baseline mean in kg 77 78 76 % change from baseline (adjusted mean) -2.9 -3.2 -0.5 Difference from placebo (adjusted mean) (95% CI) -2.4b (-3.0, -1.8) -2.7b (-3.3, -2.1) -- In Combination with Linagliptin as Add-On to Metformin Therapy
A total of 686 patients with type 2 diabetes participated in a double-blind, active-controlled study to evaluate the efficacy and safety of JARDIANCE 10 mg or 25 mg in combination with linagliptin 5 mg compared to the individual components.
Patients with type 2 diabetes inadequately controlled on at least 1500 mg of metformin per day entered a single-blind placebo run-in period for 2 weeks. At the end of the run-in period, patients who remained inadequately controlled and had an HbA1c between 7 and 10.5% were randomized 1:1:1:1:1 to one of 5 active-treatment arms of JARDIANCE 10 mg or 25 mg, linagliptin 5 mg, or linagliptin 5 mg in combination with 10 mg or 25 mg JARDIANCE as a fixed dose combination tablet.
At Week 24, JARDIANCE 10 mg or 25 mg used in combination with linagliptin 5 mg provided statistically significant improvement in HbA1c (p-value <0.0001) and FPG (p-value <0.001) compared to the individual components in patients who had been inadequately controlled on metformin. Treatment with JARDIANCE/linagliptin 25 mg/5 mg or JARDIANCE/linagliptin 10 mg/5 mg daily also resulted in a statistically significant reduction in body weight compared to linagliptin 5 mg (p-value <0.0001). There was no statistically significant difference in body weight compared to JARDIANCE alone.
Active-Controlled Study versus Glimepiride in Combination with Metformin
The efficacy of JARDIANCE was evaluated in a double-blind, glimepiride-controlled, study in 1545 patients with type 2 diabetes with insufficient glycemic control despite metformin therapy.
Patients with inadequate glycemic control and an HbA1c between 7% and 10% after a 2-week run-in period were randomized to glimepiride or JARDIANCE 25 mg.
At Week 52, JARDIANCE 25 mg and glimepiride lowered HbA1c and FPG (see Table 8, Figure 4). The difference in observed effect size between JARDIANCE 25 mg and glimepiride excluded the pre-specified non-inferiority margin of 0.3%. The mean daily dose of glimepiride was 2.7 mg and the maximal approved dose in the United States is 8 mg per day.
Table 8 Results at Week 52 from an Active-Controlled Study Comparing JARDIANCE to Glimepiride as Add-On Therapy in Patients Inadequately Controlled on Metformin JARDIANCE 25 mg + Metformin
N=765Glimepiride + Metformin
N=780aModified intent to treat population. Last observation on study (LOCF) was used to impute data missing at Week 52. At Week 52, data was imputed for 15.3% and 21.9% of patients randomized to JARDIANCE 25 mg and glimepiride, respectively.
bNon-inferior, ANCOVA model p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region)
cANCOVA p-value <0.0001 (Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.)
dFPG (mg/dL); for JARDIANCE 25 mg, n=764, for glimepiride, n=779HbA1c (%)a Baseline (mean) 7.9 7.9 Change from baseline (adjusted mean) -0.7 -0.7 Difference from glimepiride (adjusted mean) (97.5% CI) -0.07b (-0.15, 0.01) -- FPG (mg/dL)d Baseline (mean) 150 150 Change from baseline (adjusted mean) -19 -9 Difference from glimepiride (adjusted mean) -11 -- Body Weight Baseline mean in kg 82.5 83 % change from baseline (adjusted mean) -3.9 2.0 Difference from glimepiride (adjusted mean) (95% CI) -5.9c (-6.3, -5.5) -- Figure 4 Adjusted mean HbA1c Change at Each Time Point (Completers) and at Week 52 (mITT Population) - LOCF 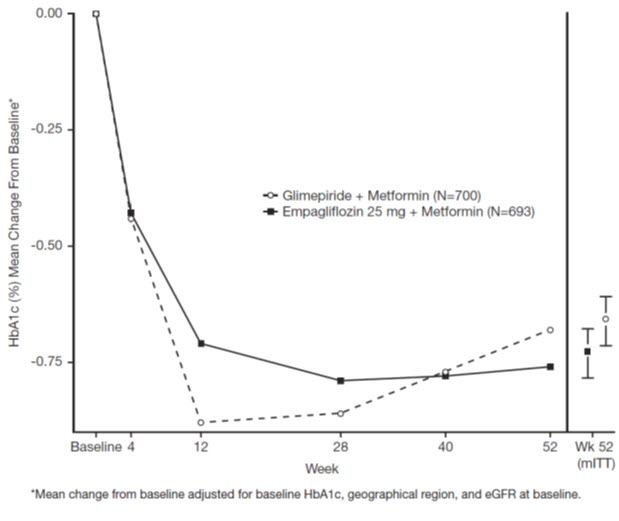
At Week 52, the adjusted mean change from baseline in systolic blood pressure was -3.6 mmHg, compared to 2.2 mmHg for glimepiride. The differences between treatment groups for systolic blood pressure was statistically significant (p-value <0.0001).
At Week 104, the adjusted mean change from baseline in HbA1c was -0.75% for JARDIANCE 25 mg and -0.66% for glimepiride. The adjusted mean treatment difference was -0.09% with a 97.5% confidence interval of (-0.32%, 0.15%), excluding the pre-specified non-inferiority margin of 0.3%. The mean daily dose of glimepiride was 2.7 mg and the maximal approved dose in the United States is 8 mg per day. The Week 104 analysis included data with and without concomitant glycemic rescue medication, as well as off-treatment data. Missing data for patients not providing any information at the visit were imputed based on the observed off-treatment data. In this multiple imputation analysis, 13.9% of the data were imputed for JARDIANCE 25 mg and 12.9% for glimepiride.
At Week 104, JARDIANCE 25 mg daily resulted in a statistically significant difference in change from baseline for body weight compared to glimepiride (-3.1 kg for JARDIANCE 25 mg vs. +1.3 kg for glimepiride; ANCOVA-LOCF, p-value <0.0001).
Add-On Combination Therapy with Pioglitazone with or without Metformin
A total of 498 patients with type 2 diabetes participated in a double-blind, placebo-controlled study to evaluate the efficacy and safety of JARDIANCE in combination with pioglitazone, with or without metformin.
Patients with inadequately controlled type 2 diabetes on metformin at a dose of at least 1500 mg per day and pioglitazone at a dose of at least 30 mg per day were placed into an open-label placebo run-in for 2 weeks. Patients with inadequate glycemic control and an HbA1c between 7% and 10% after the run-in period were randomized to placebo, JARDIANCE 10 mg, or JARDIANCE 25 mg.
Treatment with JARDIANCE 10 mg or 25 mg daily resulted in statistically significant reductions in HbA1c (p-value <0.0001), FPG, and body weight compared with placebo (see Table 9).
Table 9 Results of Placebo-Controlled Study for JARDIANCE in Combination Therapy with Pioglitazone JARDIANCE
10 mg + Pioglitazone
N=165JARDIANCE
25 mg + Pioglitazone
N=168Placebo + Pioglitazone
N=165aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 10.9%, 8.3%, and 20.6% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively.
bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and background medication. Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.)
cFPG (mg/dL); for JARDIANCE 10 mg, n=163HbA1c (%)a Baseline (mean) 8.1 8.1 8.2 Change from baseline (adjusted mean) -0.6 -0.7 -0.1 Difference from placebo + pioglitazone (adjusted mean) (95% CI) -0.5b (-0.7, -0.3) -0.6b (-0.8, -0.4) -- Patients [n (%)] achieving HbA1c <7% 36 (24%) 48 (30%) 12 (8%) FPG (mg/dL)c Baseline (mean) 152 152 152 Change from baseline (adjusted mean) -17 -22 7 Difference from placebo + pioglitazone (adjusted mean) (97.5% CI) -23b (-31.8, -15.2) -28b (-36.7, -20.2) -- Body Weight Baseline mean in kg 78 79 78 % change from baseline (adjusted mean) -2.0 -1.8 0.6 Difference from placebo (adjusted mean) (95% CI) -2.6b (-3.4, -1.8) -2.4b (-3.2, -1.6) -- Add-On Combination with Insulin with or without Metformin and/or Sulfonylureas
A total of 494 patients with type 2 diabetes inadequately controlled on insulin, or insulin in combination with oral drugs participated in a double-blind, placebo-controlled study to evaluate the efficacy of JARDIANCE as add-on therapy to insulin over 78 weeks.
Patients entered a 2-week placebo run-in period on basal insulin (e.g., insulin glargine, insulin detemir, or NPH insulin) with or without metformin and/or sulfonylurea background therapy. Following the run-in period, patients with inadequate glycemic control were randomized to the addition of JARDIANCE 10 mg, JARDIANCE 25 mg, or placebo. Patients were maintained on a stable dose of insulin prior to enrollment, during the run-in period, and during the first 18 weeks of treatment. For the remaining 60 weeks, insulin could be adjusted. The mean total daily insulin dose at baseline for JARDIANCE 10 mg, 25 mg, and placebo was 45 IU, 48 IU, and 48 IU, respectively.
JARDIANCE used in combination with insulin (with or without metformin and/or sulfonylurea) provided statistically significant reductions in HbA1c and FPG compared to placebo after both 18 and 78 weeks of treatment (see Table 10). JARDIANCE 10 mg or 25 mg daily also resulted in statistically significantly greater percent body weight reduction compared to placebo.
Table 10 Results at Week 18 and 78 for a Placebo-Controlled Study for JARDIANCE in Combination with Insulin 18 weeks (no insulin adjustment) 78 weeks (adjustable insulin dose after 18 weeks) JARDIANCE
10 mg + Insulin
N=169JARDIANCE
25 mg + Insulin
N=155Placebo + Insulin
N=170JARDIANCE
10 mg + Insulin
N=169JARDIANCE
25 mg + Insulin
N=155Placebo + Insulin
N=170aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 18 and 78. At Week 18, 21.3%, 30.3%, and 21.8% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively. At Week 78, 32.5%, 38.1% and 42.4% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively.
bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, and region; FPG: MMRM model includes baseline FPG, baseline HbA1c, treatment, region, visit and visit by treatment interaction. Body weight: MMRM model includes baseline body weight, baseline HbA1c, treatment, region, visit and visit by treatment interaction.
cp-value=0.0049
dp-value=0.0052
ep-value=0.0463HbA1c (%)a Baseline (mean) 8.3 8.3 8.2 8.3 8.3 8.2 Change from baseline (adjusted mean) -0.6 -0.7 0 -0.4 -0.6 0.1 Difference from placebo (adjusted mean) (97.5% CI) -0.6b
(-0.8, -0.4)-0.7b
(-0.9, -0.5)-- -0.5b
(-0.7, -0.3)-0.7b
(-0.9, -0.5)-- Patients (%) achieving HbA1c <7% 18.0 19.5 5.5 12.0 17.5 6.7 FPG (mg/dL) Baseline (mean) 138 146 142 138 146 142 Change from baseline (adjusted mean, SE) -17.9 (3.2) -19.1 (3.3) 10.4 (3.1) -10.1 (3.2) -15.2 (3.4) 2.8 (3.2) Difference from placebo (adjusted mean) (95% CI) -28.2b
(-37.0, -19.5)-29.5b
(-38.4, -20.6)-- -12.9c
(-21.9, 3.9)-17.9b
(-27.0, -8.8)-- Body Weight Baseline mean in kg 92 95 90 92 95 90 % change from baseline (adjusted mean) -1.8 -1.4 -0.1 -2.4 -2.4 0.7 Difference from placebo (adjusted mean) (95% CI) -1.7d
(-3.0, -0.5)-1.3e
(-2.5, -0.0)-- -3.0b
(-4.4, -1.7)-3.0b
(-4.4, -1.6)-- Add-on Combination with MDI Insulin with or without Metformin
A total of 563 patients with type 2 diabetes inadequately controlled on multiple daily injections (MDI) of insulin (total daily dose >60 IU), alone or in combination with metformin, participated in a double-blind, placebo-controlled study to evaluate the efficacy of JARDIANCE as add-on therapy to MDI insulin over 18 weeks.
Patients entered a 2-week placebo run-in period on MDI insulin with or without metformin background therapy. Following the run-in period, patients with inadequate glycemic control were randomized to the addition of JARDIANCE 10 mg, JARDIANCE 25 mg, or placebo. Patients were maintained on a stable dose of insulin prior to enrollment, during the run-in period, and during the first 18 weeks of treatment. The mean total daily insulin dose at baseline for JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo was 88.6 IU, 90.4 IU, and 89.9 IU, respectively.
JARDIANCE 10 mg or 25 mg daily used in combination with MDI insulin (with or without metformin) provided statistically significant reductions in HbA1c compared to placebo after 18 weeks of treatment (see Table 11).
Table 11 Results at Week 18 for a Placebo-Controlled Study for JARDIANCE in Combination with Insulin and with or without Metformin JARDIANCE 10 mg + Insulin +/- Metformin
N=186JARDIANCE 25 mg + Insulin +/- Metformin
N=189Placebo + Insulin +/- Metformin
N=188aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 18. At Week 18, 23.7%, 22.8% and 23.4% was imputed for patients randomized to JARDIANCE 10 mg, JARDIANCE 25 mg, and placebo, respectively.
bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, geographical region, and background medication).HbA1c (%)a Baseline (mean) 8.4 8.3 8.3 Change from baseline (adjusted mean) -0.9 -1.0 -0.5 Difference from placebo (adjusted mean) (95% CI) -0.4b (-0.6, -0.3) -0.5b (-0.7, -0.4) -- During an extension period with treatment for up to 52 weeks, insulin could be adjusted to achieve defined glucose target levels. The change from baseline in HbA1c was maintained from 18 to 52 weeks with both JARDIANCE 10 mg and 25 mg. After 52 weeks, JARDIANCE 10 mg or 25 mg daily resulted in statistically greater percent body weight reduction compared to placebo (p-value <0.0001). The mean change in body weight from baseline was -1.95 kg for JARDIANCE 10 mg, and -2.04 kg for JARDIANCE 25 mg.
Renal Impairment
A total of 738 patients with type 2 diabetes and a baseline eGFR less than 90 mL/min/1.73 m2 participated in a randomized, double-blind, placebo-controlled, parallel-group to evaluate the efficacy and safety of JARDIANCE in patients with type 2 diabetes and renal impairment. The trial population comprised of 290 patients with mild renal impairment (eGFR 60 to less than 90 mL/min/1.73 m2), 374 patients with moderate renal impairment (eGFR 30 to less than 60 mL/min/1.73 m2), and 74 with severe renal impairment (eGFR less than 30 mL/min/1.73 m2). A total of 194 patients with moderate renal impairment had a baseline eGFR of 30 to less than 45 mL/min/1.73 m2 and 180 patients a baseline eGFR of 45 to less than 60 mL/min/1.73 m2.
At Week 24, JARDIANCE 25 mg provided statistically significant reduction in HbA1c relative to placebo in patients with mild to moderate renal impairment (see Table 12). A statistically significant reduction relative to placebo was also observed with JARDIANCE 25 mg in patients with either mild [-0.7 (95% CI: -0.9, -0.5)] or moderate [-0.4 (95% CI: -0.6, -0.3)] renal impairment and with JARDIANCE 10 mg in patients with mild [-0.5 (95% CI: -0.7, -0.3)] renal impairment.
The glucose lowering efficacy of JARDIANCE 25 mg decreased with decreasing level of renal function in the mild to moderate range. Least square mean Hb1Ac changes at 24 weeks were -0.6%, -0.5%, and -0.2% for those with a baseline eGFR of 60 to less than 90 mL/min/1.73 m2, 45 to less than 60 mL/min/1.73 m2, and 30 to less than 45 mL/min/1.73 m2, respectively [see Dosage and Administration (2) and Use in Specific Populations (8.6)]. For placebo, least square mean HbA1c changes at 24 weeks were 0.1%, -0.1%, and 0.2% for patients with a baseline eGFR of 60 to less than 90 mL/min/1.73 m2, 45 to less than 60 mL/min/1.73 m2, and 30 to less than 45 mL/min/1.73 m2, respectively.
Table 12 Results at Week 24 (LOCF) of Placebo-Controlled Study for JARDIANCE in Patients with Type 2 Diabetes and Renal Impairment Mild and Moderate Impairmentb JARDIANCE 25 mg ap-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and background medication)
beGFR 30 to less than 90 mL/min/1.73 m2- Modified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 24.6% and 26.2% was imputed for patients randomized to JARDIANCE 25 mg and placebo, respectively.HbA1c Number of patients n=284 Comparison vs placebo (adjusted mean) (95% CI) -0.5a (-0.6, -0.4) For patients with severe renal impairment, the analyses of changes in HbA1c and FPG showed no discernible treatment effect of JARDIANCE 25 mg compared to placebo [see Dosage and Administration (2.2) and Use in Specific Populations (8.6)].
14.2 Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
The effect of JARDIANCE on cardiovascular risk in adult patients with type 2 diabetes and established, stable, atherosclerotic cardiovascular disease was evaluated in the EMPA-REG OUTCOME study, a multicenter, multi-national, randomized, double-blind parallel group trial. The study compared the risk of experiencing a major adverse cardiovascular event (MACE) between JARDIANCE and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease. Coadministered antidiabetic medications were to be kept stable for the first 12 weeks of the trial. Thereafter, antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 7020 patients were treated (JARDIANCE 10 mg = 2345; JARDIANCE 25 mg = 2342; placebo = 2333) and followed for a median of 3.1 years. Approximately 72% of the study population was Caucasian, 22% was Asian, and 5% was Black. The mean age was 63 years and approximately 72% were male.
All patients in the study had inadequately controlled type 2 diabetes mellitus at baseline (HbA1c greater than or equal to 7%). The mean HbA1c at baseline was 8.1% and 57% of participants had diabetes for more than 10 years. Approximately 31%, 22% and 20% reported a past history of neuropathy, retinopathy and nephropathy to investigators respectively and the mean eGFR was 74 mL/min/1.73 m2. At baseline, patients were treated with one (~30%) or more (~70%) antidiabetic medications including metformin (74%), insulin (48%), and sulfonylurea (43%).
All patients had established atherosclerotic cardiovascular disease at baseline including one (82%) or more (18%) of the following; a documented history of coronary artery disease (76%), stroke (23%) or peripheral artery disease (21%). At baseline, the mean systolic blood pressure was 136 mmHg, the mean diastolic blood pressure was 76 mmHg, the mean LDL was 86 mg/dL, the mean HDL was 44 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 175 mg/g. At baseline, approximately 81% of patients were treated with renin angiotensin system inhibitors, 65% with beta-blockers, 43% with diuretics, 77% with statins, and 86% with antiplatelet agents (mostly aspirin).
The primary endpoint in EMPA-REG OUTCOME was the time to first occurrence of a Major Adverse Cardiac Event (MACE). A major adverse cardiac event was defined as occurrence of either a cardiovascular death or a nonfatal myocardial infarction (MI) or a nonfatal stroke. The statistical analysis plan had pre-specified that the 10 and 25 mg doses would be combined. A Cox proportional hazards model was used to test for non-inferiority against the pre-specified risk margin of 1.3 for the hazard ratio of MACE and superiority on MACE if non-inferiority was demonstrated. Type-1 error was controlled across multiples tests using a hierarchical testing strategy.
JARDIANCE significantly reduced the risk of first occurrence of primary composite endpoint of cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke (HR: 0.86; 95% CI 0.74, 0.99). The treatment effect was due to a significant reduction in the risk of cardiovascular death in subjects randomized to empagliflozin (HR: 0.62; 95% CI 0.49, 0.77), with no change in the risk of non-fatal myocardial infarction or non-fatal stroke (see Table 13 and Figure 5 and 6). Results for the 10 mg and 25 mg empagliflozin doses were consistent with results for the combined dose groups.
Table 13 Treatment Effect for the Primary Composite Endpoint, and its Componentsa Placebo
N=2333JARDIANCE
N=4687Hazard ratio vs placebo
(95% CI)aTreated set (patients who had received at least one dose of study drug)
bp−value for superiority (2−sided) 0.04
cTotal number of eventsComposite of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke (time to first occurrence)b 282 (12.1%) 490 (10.5%) 0.86 (0.74, 0.99) Non-fatal myocardial infarctionc 121 (5.2%) 213 (4.5%) 0.87 (0.70, 1.09) Non-fatal strokec 60 (2.6%) 150 (3.2%) 1.24 (0.92, 1.67) Cardiovascular deathc 137 (5.9%) 172 (3.7%) 0.62 (0.49, 0.77) Figure 5 Estimated Cumulative Incidence of First MACE 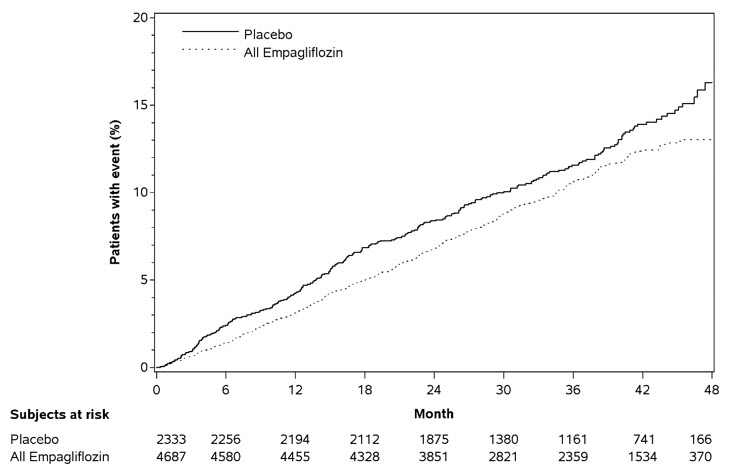
Figure 6 Estimated Cumulative Incidence of Cardiovascular Death 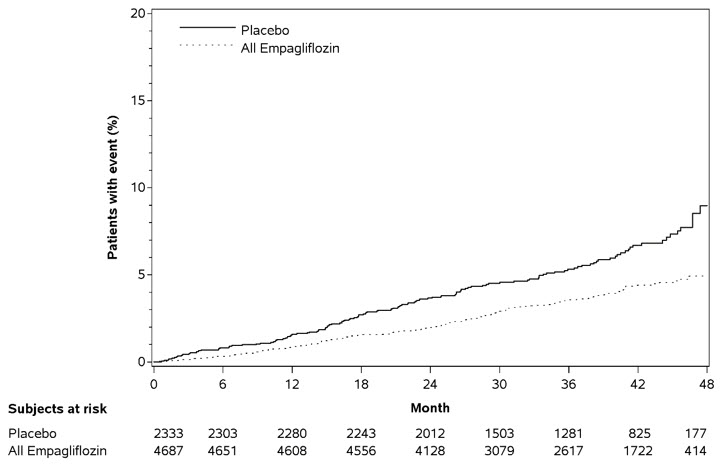
The efficacy of JARDIANCE on cardiovascular death was generally consistent across major demographic and disease subgroups.
Vital status was obtained for 99.2% of subjects in the trial. A total of 463 deaths were recorded during the EMPA-REG OUTCOME trial. Most of these deaths were categorized as cardiovascular deaths. The non-cardiovascular deaths were only a small proportion of deaths, and were balanced between the treatment groups (2.1% in patients treated with JARDIANCE, and 2.4% of patients treated with placebo).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
JARDIANCE tablets are available in 10 mg and 25 mg strengths as follows:
10 mg tablets: pale yellow, round, biconvex and bevel-edged, film-coated tablets debossed with "S 10" on one side and the Boehringer Ingelheim company symbol on the other side.
Bottles of 30 (NDC: 0597-0152-30)
Bottles of 90 (NDC: 0597-0152-90)
Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC: 0597-0152-37), institutional pack.25 mg tablets: pale yellow, oval, biconvex film-coated tablets, debossed with "S 25" on one side and the Boehringer Ingelheim company symbol on the other side.
Bottles of 30 (NDC: 0597-0153-30)
Bottles of 90 (NDC: 0597-0153-90)
Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC: 0597-0153-37), institutional pack.Dispense in a well-closed container as defined in the USP.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Instructions
Instruct patients to read the Medication Guide before starting JARDIANCE therapy and to reread it each time the prescription is renewed. Instruct patients to inform their doctor or pharmacist if they develop any unusual symptom, or if any known symptom persists or worsens.
Inform patients of the potential risks and benefits of JARDIANCE and of alternative modes of therapy. Also inform patients about the importance of adherence to dietary instructions, regular physical activity, periodic blood glucose monitoring and HbA1c testing, recognition and management of hypoglycemia and hyperglycemia, and assessment for diabetes complications. Advise patients to seek medical advice promptly during periods of stress such as fever, trauma, infection, or surgery, as medication requirements may change.
Instruct patients to take JARDIANCE only as prescribed. If a dose is missed, it should be taken as soon as the patient remembers. Advise patients not to double their next dose.
Inform patients that the most common adverse reactions associated with the use of JARDIANCE are urinary tract infections and mycotic genital infections.
Advise pregnant women, and females of reproductive potential of the potential risk to a fetus with treatment with JARDIANCE [see Use in Specific Populations (8.1)]. Instruct females of reproductive potential to report pregnancies to their physicians as soon as possible.
Advise women that breastfeeding is not recommended during treatment with JARDIANCE [see Use in Specific Populations (8.2)].
Hypotension
Inform patients that hypotension may occur with JARDIANCE and advise them to contact their healthcare provider if they experience such symptoms [see Warnings and Precautions (5.1)]. Inform patients that dehydration may increase the risk for hypotension, and to have adequate fluid intake.
Ketoacidosis
Inform patients that ketoacidosis is a serious life-threatening condition and that cases of ketoacidosis have been reported during use of JARDIANCE, sometimes associated with illness or surgery among other risk factors. Instruct patients to check ketones (when possible) if symptoms consistent with ketoacidosis occur even if blood glucose is not elevated. If symptoms of ketoacidosis (including nausea, vomiting, abdominal pain, tiredness, and labored breathing) occur, instruct patients to discontinue JARDIANCE and seek medical attention immediately [see Warnings and Precautions (5.2)].
Acute Kidney Injury
Inform patients that acute kidney injury has been reported during use of JARDIANCE. Advise patients to seek medical advice immediately if they have reduced oral intake (such as due to acute illness or fasting) or increased fluid losses (such as due to vomiting, diarrhea, or excessive heat exposure), as it may be appropriate to temporarily discontinue JARDIANCE use in those settings [see Warnings and Precautions (5.3)].
Serious Urinary Tract Infections
Inform patients of the potential for urinary tract infections, which may be serious. Provide them with information on the symptoms of urinary tract infections. Advise them to seek medical advice if such symptoms occur [see Warnings and Precautions (5.4)].
Necrotizing Fasciitis of the Perineum (Fournier's Gangrene)
Inform patients that necrotizing infections of the perineum (Fournier's gangrene) have occurred with JARDIANCE. Counsel patients to promptly seek medical attention if they develop pain or tenderness, redness, or swelling of the genitals or the area from the genitals back to the rectum, along with a fever above 100.4°F or malaise [see Warnings and Precautions (5.6)].
Genital Mycotic Infections in Females (e.g., Vulvovaginitis)
Inform female patients that vaginal yeast infections may occur and provide them with information on the signs and symptoms of vaginal yeast infections. Advise them of treatment options and when to seek medical advice [see Warnings and Precautions (5.7)].
Genital Mycotic Infections in Males (e.g., Balanitis or Balanoposthitis)
Inform male patients that yeast infection of penis (e.g., balanitis or balanoposthitis) may occur, especially in uncircumcised males and patients with chronic and recurrent infections. Provide them with information on the signs and symptoms of balanitis and balanoposthitis (rash or redness of the glans or foreskin of the penis). Advise them of treatment options and when to seek medical advice [see Warnings and Precautions (5.7)].
Hypersensitivity Reactions
Inform patients that serious hypersensitivity reactions, such as urticaria and angioedema, have been reported with JARDIANCE. Advise patients to report immediately any skin reaction or angioedema, and to discontinue drug until they have consulted prescribing physician [see Warnings and Precautions (5.8)].
Laboratory Tests
Inform patients that renal function should be assessed prior to initiation of JARDIANCE and monitored periodically thereafter.
Inform patients that elevated glucose in urinalysis is expected when taking JARDIANCE.
Inform patients that response to all diabetic therapies should be monitored by periodic measurements of blood glucose and HbA1c levels, with a goal of decreasing these levels toward the normal range. Hemoglobin A1c monitoring is especially useful for evaluating long-term glycemic control.
-
SPL UNCLASSIFIED SECTION
Distributed by:
Boehringer Ingelheim Pharmaceuticals, Inc.
Ridgefield, CT 06877 USAMarketed by:
Boehringer Ingelheim Pharmaceuticals, Inc.
Ridgefield, CT 06877 USA
and
Eli Lilly and Company
Indianapolis, IN 46285 USALicensed from:
Boehringer Ingelheim International GmbH, Ingelheim, GermanyBoehringer Ingelheim Pharmaceuticals, Inc. either owns or uses the Jardiance® and EMPA-REG OUTCOME® trademarks under license.
The other brands listed are trademarks of their respective owners and are not trademarks of Boehringer Ingelheim Pharmaceuticals, Inc.
Copyright © 2020 Boehringer Ingelheim International GmbH
ALL RIGHTS RESERVEDIT5728VA152020
-
MEDICATION GUIDE
This MEDICATION GUIDE has been approved by the U.S. Food and Drug Administration. Revised: January 2020 MEDICATION GUIDE
JARDIANCE® (jar DEE ans)
(empagliflozin)
TabletsWhat is the most important information I should know about JARDIANCE?
JARDIANCE can cause serious side effects, including:-
Dehydration. JARDIANCE can cause some people to have dehydration (the loss of body water and salt). Dehydration may cause you to feel dizzy, faint, light-headed, or weak, especially when you stand up (orthostatic hypotension).
You may be at higher risk of dehydration if you:- have low blood pressure
- take medicines to lower your blood pressure, including diuretics (water pills)
- are on low sodium (salt) diet
- have kidney problems
- are 65 years of age or older
-
Vaginal yeast infection. Women who take JARDIANCE may get vaginal yeast infections. Symptoms of a vaginal yeast infection include:
- vaginal odor
- white or yellowish vaginal discharge (discharge may be lumpy or look like cottage cheese)
- vaginal itching
-
Yeast infection of the penis (balanitis or balanoposthitis). Men who take JARDIANCE may get a yeast infection of the skin around the penis. Certain men who are not circumcised may have swelling of the penis that makes it difficult to pull back the skin around the tip of the penis. Other symptoms of yeast infection of the penis include:
- redness, itching, or swelling of the penis
- rash of the penis
- foul smelling discharge from the penis
- pain in the skin around penis
Talk to your doctor about what to do if you get symptoms of a yeast infection of the vagina or penis. Your doctor may suggest you use an over-the-counter antifungal medicine. Talk to your doctor right away if you use an over-the-counter antifungal medication and your symptoms do not go away. What is JARDIANCE? - JARDIANCE is a prescription medicine used:
- along with diet and exercise to lower blood sugar in adults with type 2 diabetes.
- to reduce the risk of cardiovascular death in adults with type 2 diabetes who have known cardiovascular disease.
- JARDIANCE is not for people with type 1 diabetes.
- JARDIANCE is not for people with diabetic ketoacidosis (increased ketones in the blood or urine).
- It is not known if JARDIANCE is safe and effective in children under 18 years of age.
Who should not take JARDIANCE?
Do not take JARDIANCE if you:- are allergic to empagliflozin or any of the ingredients in JARDIANCE. See the end of this Medication Guide for a list of ingredients in JARDIANCE.
- have severe kidney problems or are on dialysis
What should I tell my doctor before using JARDIANCE?
Before you take JARDIANCE, tell your doctor if you:- have kidney problems
- have liver problems
- have a history of urinary tract infections or problems with urination
- are going to have surgery. Your doctor may stop your JARDIANCE before you have surgery. Talk to your doctor if you are having surgery about when to stop taking JARDIANCE and when to start it again.
- are eating less, or there is a change in your diet
- have or have had problems with your pancreas, including pancreatitis or surgery on your pancreas
- drink alcohol very often, or drink a lot of alcohol in the short term ("binge" drinking)
- have any other medical conditions
- are pregnant or plan to become pregnant. JARDIANCE may harm your unborn baby. If you become pregnant while taking JARDIANCE, tell your doctor as soon as possible. Talk with your doctor about the best way to control your blood sugar while you are pregnant.
- are breastfeeding or plan to breastfeed. JARDIANCE may pass into your breast milk and may harm your baby. Talk with your doctor about the best way to feed your baby if you are taking JARDIANCE. Do not breastfeed while taking JARDIANCE.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. JARDIANCE may affect the way other medicines work, and other medicines may affect how JARDIANCE works. Especially tell your doctor if you take: - diuretics (water pills)
- insulin or other medicines that can lower your blood sugar
Ask your doctor or pharmacist for a list of these medicines if you are not sure if your medicine is listed above. How should I take JARDIANCE? - Take JARDIANCE exactly as your doctor tells you to take it.
- Take JARDIANCE by mouth 1 time in the morning each day, with or without food.
- Your doctor may change your dose if needed.
- If you miss a dose, take it as soon as you remember. If you do not remember until it is time for your next dose, skip the missed dose and go back to your regular schedule. Do not take two doses of JARDIANCE at the same time. Talk with your doctor if you have questions about a missed dose.
- Your doctor may tell you to take JARDIANCE along with other diabetes medicines. Low blood sugar can happen more often when JARDIANCE is taken with certain other diabetes medicines. See "What are the possible side effects of JARDIANCE?"
- If you take too much JARDIANCE, call your doctor or go to the nearest hospital emergency room right away.
- When your body is under some types of stress, such as fever, trauma (such as a car accident), infection, or surgery, the amount of diabetes medicine that you need may change. Tell your doctor right away if you have any of these conditions and follow your doctor's instructions.
- Check your blood sugar as your doctor tells you to.
- Stay on your prescribed diet and exercise program while taking JARDIANCE.
- Talk to your doctor about how to prevent, recognize and manage low blood sugar (hypoglycemia), high blood sugar (hyperglycemia), and complications of diabetes.
- Your doctor will check your diabetes with regular blood tests, including your blood sugar levels and your hemoglobin HbA1c.
- When taking JARDIANCE, you may have sugar in your urine, which will show up on a urine test.
What are the possible side effects of JARDIANCE? JARDIANCE may cause serious side effects, including: - See "What is the most important information I should know about JARDIANCE?"
- Ketoacidosis (increased ketones in your blood or urine). Ketoacidosis has happened in people who have type 1 diabetes or type 2 diabetes, during treatment with JARDIANCE. Ketoacidosis has also happened in people with diabetes who were sick or who had surgery during treatment with JARDIANCE. Ketoacidosis is a serious condition, which may need to be treated in a hospital. Ketoacidosis may lead to death. Ketoacidosis can happen with JARDIANCE even if your blood sugar is less than 250 mg/dL. Stop taking JARDIANCE and call your doctor right away if you get any of the following symptoms:
- nausea
- vomiting
- stomach-area (abdominal) pain
- tiredness
- trouble breathing
If you get any of these symptoms during treatment with JARDIANCE, if possible, check for ketones in your urine, even if your blood sugar is less than 250 mg/dL. -
Kidney problems. Sudden kidney injury has happened to people taking JARDIANCE. Talk to your doctor right away if you:
- reduce the amount of food or liquid you drink for example, if you are sick or cannot eat or
- start to lose liquids from your body for example, from vomiting, diarrhea or being in the sun too long
- Serious urinary tract infections. Serious urinary tract infections that may lead to hospitalization have happened in people who are taking JARDIANCE. Tell your doctor if you have any signs or symptoms of a urinary tract infection such as a burning feeling when passing urine, a need to urinate often, the need to urinate right away, pain in the lower part of your stomach (pelvis), or blood in the urine. Sometimes people also may have a fever, back pain, nausea or vomiting.
- Low blood sugar (hypoglycemia). If you take JARDIANCE with another medicine that can cause low blood sugar, such as a sulfonylurea or insulin, your risk of getting low blood sugar is higher. The dose of your sulfonylurea medicine or insulin may need to be lowered while you take JARDIANCE. Signs and symptoms of low blood sugar may include:
- headache
- drowsiness
- weakness
- irritability
- hunger
- fast heartbeat
- confusion
- shaking or feeling jittery
- dizziness
- sweating
- A rare but serious bacterial infection that causes damage to the tissue under the skin (necrotizing fasciitis) in the area between and around the anus and genitals (perineum). Necrotizing fasciitis of the perineum has happened in women and men who take JARDIANCE. Necrotizing fasciitis of the perineum may lead to hospitalization, may require multiple surgeries, and may lead to death. Seek medical attention immediately if you have a fever or you are feeling very weak, tired or uncomfortable (malaise), and you develop any of the following symptoms in the area between and around your anus and genitals:
- pain or tenderness
- swelling
- redness of skin (erythema)
-
Allergic (hypersensitivity) reactions. Serious allergic reactions have happened in people who are taking JARDIANCE. Symptoms may include
- swelling of your face, lips, throat and other areas of your skin
- difficulty with swallowing or breathing.
- raised, red areas on your skin (hives)
- Increased fats in your blood (cholesterol)
These are not all the possible side effects of JARDIANCE. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store JARDIANCE?
Store JARDIANCE at room temperature 68°F to 77°F (20°C to 25°C).General information about the safe and effective use of JARDIANCE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use JARDIANCE for a condition for which it is not prescribed. Do not give JARDIANCE to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about JARDIANCE. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about JARDIANCE that is written for health professionals.
For more information about JARDIANCE including current prescribing information and Medication Guide, go to www.jardiance.com, scan the code below, or call Boehringer Ingelheim Pharmaceuticals, Inc. at 1-800-542-6257 or (TTY) 1-800-459-9906.

What are the ingredients in JARDIANCE? Active Ingredient: empagliflozin Inactive Ingredients: lactose monohydrate, microcrystalline cellulose, hydroxypropyl cellulose, croscarmellose sodium, colloidal silicon dioxide and magnesium stearate. In addition, the film coating contains the following inactive ingredients: hypromellose, titanium dioxide, talc, polyethylene glycol, and yellow ferric oxide. Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc.; Ridgefield, CT 06877 USA
Marketed by: Boehringer Ingelheim Pharmaceuticals, Inc.; Ridgefield, CT 06877 USA and Eli Lilly and Company, Indianapolis, IN 46285 USA
Licensed from: Boehringer Ingelheim International GmbH, Ingelheim, GermanyBoehringer Ingelheim Pharmaceuticals, Inc. either owns or uses the Jardiance® and EMPA-REG OUTCOME® trademarks under license. The other brands listed are trademarks of their respective owners and are not trademarks of Boehringer Ingelheim Pharmaceuticals, Inc. Copyright © 2020 Boehringer Ingelheim International GmbH
ALL RIGHTS RESERVEDIT5728VA152020 -
Dehydration. JARDIANCE can cause some people to have dehydration (the loss of body water and salt). Dehydration may cause you to feel dizzy, faint, light-headed, or weak, especially when you stand up (orthostatic hypotension).
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC 0597-0152-37
DISPENSE WITH ENCLOSED MEDICATION GUIDE
Jardiance®
(empagliflozin)
Tablets
10 mg
This unit does package is not child-resistant. If dispensed for outpatient use, a child-resistant container should be utilized.
Rx only
30 tablets
3 blister cards. Each card contains 10 tablets.
Boehringer Ingelheim
Lilly
NDC 0597-0153-37
DISPENSE WITH ENCLOSED MEDICATION GUIDE
Jardiance®
(empagliflozin)
Tablets
25 mg
This unit does package is not child-resistant. If dispensed for outpatient use, a child-resistant container should be utilized.
Rx only
30 tablets
3 blister cards. Each card contains 10 tablets.
Boehringer Ingelheim
Lilly
-
INGREDIENTS AND APPEARANCE
JARDIANCE
empagliflozin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0597-0152 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength EMPAGLIFLOZIN (UNII: HDC1R2M35U) (EMPAGLIFLOZIN - UNII:HDC1R2M35U) EMPAGLIFLOZIN 10 mg Product Characteristics Color YELLOW (pale yellow) Score no score Shape ROUND Size 9mm Flavor Imprint Code S;10 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0597-0152-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 08/01/2014 2 NDC: 0597-0152-07 1 in 1 CARTON 08/01/2014 2 7 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC: 0597-0152-70 7 in 1 CARTON 08/01/2014 3 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC: 0597-0152-37 30 in 1 CARTON 08/01/2014 4 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 5 NDC: 0597-0152-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 08/01/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204629 08/01/2014 JARDIANCE
empagliflozin tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0597-0153 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength EMPAGLIFLOZIN (UNII: HDC1R2M35U) (EMPAGLIFLOZIN - UNII:HDC1R2M35U) EMPAGLIFLOZIN 25 mg Product Characteristics Color YELLOW (pale yellow) Score no score Shape OVAL Size 11mm Flavor Imprint Code S;25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0597-0153-37 30 in 1 CARTON 08/01/2014 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 0597-0153-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 08/01/2014 3 NDC: 0597-0153-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 08/01/2014 4 NDC: 0597-0153-07 1 in 1 CARTON 08/01/2014 4 7 in 1 BOTTLE; Type 0: Not a Combination Product 5 NDC: 0597-0153-70 7 in 1 CARTON 08/01/2014 5 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204629 08/01/2014 Labeler - Boehringer Ingelheim Pharmaceuticals, Inc. (603175944) Registrant - Boehringer Ingelheim Pharmaceuticals, Inc. (603175944) Establishment Name Address ID/FEI Business Operations Boehringer Ingelheim Pharma GmbH and Co. KG 551147440 API MANUFACTURE(0597-0152, 0597-0153) , LABEL(0597-0152, 0597-0153) , MANUFACTURE(0597-0152, 0597-0153) , PACK(0597-0152, 0597-0153) Establishment Name Address ID/FEI Business Operations West-Ward Columbus Inc. 058839929 LABEL(0597-0152, 0597-0153) Establishment Name Address ID/FEI Business Operations Sixarp, LLC 016329513 LABEL(0597-0152, 0597-0153) Establishment Name Address ID/FEI Business Operations Boehringer Ingelheim Promeco, S.A. de C.V. 812579472 LABEL(0597-0152, 0597-0153) , PACK(0597-0152, 0597-0153) Establishment Name Address ID/FEI Business Operations Bidachem S.p.a. 429232812 API MANUFACTURE(0597-0152, 0597-0153) Establishment Name Address ID/FEI Business Operations Rottendorf Pharma GmbH 315974691 LABEL(0597-0152, 0597-0153) , PACK(0597-0152, 0597-0153)
Trademark Results [Jardiance]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 JARDIANCE 88902811 not registered Live/Pending |
Boehringer Ingelheim International GmbH 2020-05-06 |
 JARDIANCE 79060588 3595152 Live/Registered |
Boehringer Ingelheim International GmbH 2008-09-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.