RILPIVIRINE tablet, film coated
Rilpivirine by
Drug Labeling and Warnings
Rilpivirine by is a Prescription medication manufactured, distributed, or labeled by Somerset Therapeutics, LLC, Piramal Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use RILPIVIRINE TABLETS safely and effectively. See full prescribing information for RILPIVIRINE TABLETS
RILPIVIRINE tablets, for oral use
Initial U.S. Approval:2011RECENT MAJOR CHANGES
Warnings and Precautions 03/2024
Different Formulations Are Not Substitutable (5.6)
INDICATIONS AND USAGE
Rilpivirine tablets are a human immunodeficiency virus type 1 (HIV-1) specific, non-nucleoside reverse transcriptase inhibitor (NNRTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in treatment-naïve patients 12 years of age and older and weighing at least 35 kg with HIV-1 RNA less than or equal to 100,000 copies/mL (1.1)
Limitations of Use:
- More rilpivirine treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA≥50 copies/mL) compared to rilpivirine treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL. (1.1, 14)
Rilpivirine tablets are indicated in combination with VOCABRIA (cabotegravir), for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine. (1.2)
DOSAGE AND ADMINISTRATION
- One 25 mg rilpivirine tablet taken once daily with a meal for patients weighing at least 35 kg. (2.2)
- Do not substitute rilpivirine tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. (2.1,5.6)
- See full prescribing information for dosing information when used in combination with cabotegravir. (2.6)
- For pregnant patients who are already on a stable rilpivirine regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing more than 35 kg is one 25 mg tablet once daily taken orally with a meal. (2.5,12.3)
- Rifabutin coadministration: Take two 25 mg tablets of rilpivirine once daily with a meal for the duration of the rifabutin coadministration. (2.7)
DOSAGE FORMS AND STRENGTHS
- Rilpivirine: 25 mg tablets (3)
CONTRAINDICATIONS
Coadministration of rilpivirine tablets are contraindicated with drugs where significant decreases in rilpivirine plasma concentrations may occur, which may result in loss of virologic response and possible resistance and cross-resistance. (4)
WARNINGS AND PRECAUTIONS
- Skin and Hypersensitivity Reactions: Severe skin and hypersensitivity reactions have been reported during postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. Immediately discontinue treatment if hypersensitivity or rash with systemic symptoms or elevations in hepatic serum biochemistries develop and closely monitor clinical status, including hepatic serum biochemistries. (5.1)
- Hepatotoxicity: Hepatic adverse events have been reported in patients with underlying liver disease, including hepatitis B or C virus co-infection, or in patients with elevated baseline transaminases. A few cases of hepatotoxicity have occurred in virus co-infection, or marked elevations in transaminase. Also consider monitoring liver functions tests in patients without pre-existing hepatic dysfunction or other risk factors. (5.2)
- Depressive Disorders: Severe depressive disorders have been reported. Immediate medical evaluation is recommended for severe depressive disorders. (5.3)
- Patients may develop immune reconstitution syndrome. (5.5)
ADVERSE REACTIONS
The most common adverse reactions to rilpivirine tablets (incidence >2%) of at least moderate to severe intensity (≥ Grade 2) were depressive disorders, headache, insomnia and rash. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Somerset Therapeutics, LLC at 1-800-417-9175 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
DRUG INTERACTIONS
- Consider alternatives to rilpivirine tablets when coadministered with drugs with a known risk of torsade de pointes. (5.4)
- Rilpivirine tablets should not be used in combination with NNRTIs. (4, 7)
- Coadministration of rilpivirine tablets with drugs that induce or inhibit CYP3A may affect the plasma concentrations of rilpivirine. (4, 7)
- Coadministration of rilpivirine tablets with drugs that increase gastric pH may decrease plasma concentrations of rilpivirine. (4, 7)
- Refer to the Full Prescribing Information for other drugs that should not be coadministered with rilpivirine tablets and for other drugs that may require a change in dose or regimen. (7)
USE IN SPECIFIC POPULATIONS
- Pregnancy: Total rilpivirine exposures were generally lower during pregnancy compared to the postpartum period. (2.5, 8.1, 12.3)
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 2/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Treatment of HIV-1 in Treatment-Naïve Patients
1.2 Treatment of HIV-1 in Combination with Cabotegravir
2 DOSAGE AND ADMINISTRATION
2.1 Overview of Dosage Form
2.2 Recommended Dosage in Treatment-Naïve Adult Patients
2.3 Recommended Dosage in Treatment-Naïve Pediatric Patients 12 Years of Age and Older and Weighing at least 35 kg
2.5 Recommended Dosage During Pregnancy
2.6 Recommended Dosage in Combination with Cabotegravir in Adults and Adolescents 12 Years of Age and Older and Weighing at least 35 kg
2.7 Recommended Dosage with Rifabutin Coadministration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Skin and Hypersensitivity Reactions
5.2 Hepatotoxicity
5.3 Depressive Disorders
5.4 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
5.5 Immune Reconstitution Syndrome
5.6 Different Formulations Are Not Substitutable
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Treatment-Naïve Adult Subjects
14.2 Virologically-Suppressed Adults Treated in Combination with Cabotegravir
14.3 Treatment-Naïve Pediatric Subjects (≥12 to less than 18 Years of Age)
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Treatment of HIV-1 in Treatment-Naïve Patients
Rilpivirine tablets, in combination with other antiretroviral agents, are indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-naïve patients 12 years of age and older and weighing at least 35 kg with plasma HIV-1 RNA less than or equal to 100,000 copies/mL at the start of therapy.
Limitations of Use
- More rilpivirine treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to rilpivirine treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL [see Clinical Studies (14.1)].
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
1.2 Treatment of HIV-1 in Combination with Cabotegravir
Rilpivirine tablets are indicated in combination with VOCABRIA (cabotegravir) for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine, for use as [see Dosage and Administration (2.6)]:
- oral lead-in to assess the tolerability of rilpivirine prior to administration of rilpivirine extended-release injectable suspension, a component of CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension).
- oral therapy for patients who will miss planned injection dosing with CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension).
-
2 DOSAGE AND ADMINISTRATION
2.1 Overview of Dosage Form
Rilpivirine 25 mg film-coated tablets for adults and pediatric patients weighing at least 35 kg
Do not substitute rilpivirine tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. [see Warnings and Precautions (5.6)]. Take rilpivirine tablet once daily with a meal in combination with other antiretrovirals [see Clinical Pharmacology (12.3)].
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
2.2 Recommended Dosage in Treatment-Naïve Adult Patients
The recommended dosage of rilpivirine in adult patients is one 25 mg tablet taken orally once daily with a meal [see Use in Specific Populations (8.1) and Clinical Pharmacology (12.3)].
2.3 Recommended Dosage in Treatment-Naïve Pediatric Patients 12 Years of Age and Older and Weighing at least 35 kg
The recommended dosage of rilpivirine tablets in pediatric patients 12 years of age and older and weighing at least 35 kg is based on body weight (see Table 1). Rilpivirine tablets should be taken orally once daily with a meal [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.3)].
Table 1: Recommended Dosage of Rilpivirine Tablets for Pediatric Patients Body Weight (kg)
Rilpivirine 25 mg Tablets
Total Daily Dose
Greater than or equal to 35 kg
1 tablet once daily
25 mg rilpivirine tablet once daily
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
2.5 Recommended Dosage During Pregnancy
For pregnant patients who are already on a stable rilpivirine tablets regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing at least 35 kg is one 25 mg tablet once daily taken orally with a meal. Refer to Table 1 for dosing recommendations for pediatric patients [see Dosage and Administration (2.2, 2.3)]. Lower exposures of rilpivirine were observed during pregnancy, therefore viral load should be monitored closely [see Use in Specific Populations (8.1) and Clinical Pharmacology (12.3)].
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
2.6 Recommended Dosage in Combination with Cabotegravir in Adults and Adolescents 12 Years of Age and Older and Weighing at least 35 kg
Consult the prescribing information for CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) before initiating rilpivirine tablets to ensure therapy with CABENUVA is appropriate
Oral Lead-In Dosing to Assess Tolerability of Rilpivirine
Oral lead-in should be used for approximately 1 month (at least 28 days) to assess the tolerability of rilpivirine prior to the initiation of CABENUVA. The recommended oral daily dose is one 25 mg tablet of rilpivirine in combination with one 30 mg tablet of VOCABRIA (cabotegravir). Take rilpivirine tablets with VOCABRIA (cabotegravir) orally once daily at approximately the same time each day with a meal [see Clinical Pharmacology (12.3)]
Because rilpivirine tablets are indicated in combination with VOCABRIA (cabotegravir), the prescribing information for VOCABRIA (cabotegravir) tablets should also be consulted
The last oral dose should be taken on the same day injections with CABENUVA are started
Oral Dosing to Replace Planned Missed Injections of CABENUVA
Planned Missed Injections for Patients on Monthly Dosing Schedule
If a patient plans to miss a scheduled monthly injection of CABENUVA by more than 7 days, take daily oral therapy for up to 2 months to replace missed injection visits. The recommended oral daily dose is one 25 mg tablet of rilpivirine tablets and one 30 mg tablet of VOCABRIA (cabotegravir). Take rilpivirine tablets with VOCABRIA (cabotegravir) at approximately the same time each day with a meal. The first dose of oral therapy should be initiated at approximately the same time as the planned missed injection and continued until the day injection dosing is restarted. For oral therapy with rilpivirine tablets and VOCABRIA of durations greater than 2 months, an alternative oral regimen is recommended, which may include rilpivirine tablets. See full prescribing information for CABENUVA to resume monthly injection dosing.
Planned Missed Injections for Patients on Every-2-Month Dosing Schedule
If a patient plans to miss a scheduled every-2-month injection of CABENUVA by more than 7 days, take daily oral therapy for a duration of up to 2 months to replace 1 missed scheduled every-2-month injection. The recommended oral daily dose is one 25 mg tablet of rilpivirine and one 30 mg tablet of VOCABRIA (cabotegravir). Take rilpivirine tablets with VOCABRIA (cabotegravir) at approximately the same time each day with a meal. The first dose of oral therapy should be initiated at approximately the same time as the planned missed injection and continued until the day injection dosing is restarted. For oral therapy with rilpivirine tablets and VOCABRIA of durations greater than 2 months, an alternative oral regimen is recommended, which may include rilpivirine tablets. See full prescribing information for CABENUVA to resume every-2-month injection dosing.
2.7 Recommended Dosage with Rifabutin Coadministration
If rilpivirine tablets are coadministered with rifabutin, the rilpivirine tablets dose should be increased to 50 mg (two 25 mg tablets) once daily, taken with a meal. When rifabutin coadministration is stopped, the rilpivirine dose should be decreased to 25 mg once daily, taken with a meal [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
Note that use of CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) with rifabutin is contraindicated. Refer to CABENUVA labeling for additional detail.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Rilpivirine tablets are contraindicated for coadministration with the drugs in Table 2 for which significant decreases in rilpivirine plasma concentrations may occur due to CYP3A enzyme induction or gastric pH increase, which may result in loss of virologic response and possible resistance to rilpivirine tablets or to the class of NNRTIs [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
Table 2: Drugs That are Contraindicated with Rilpivirine Tablets
Drug Class
Contraindicated
Drugs in Class
Clinical Comment
Anticonvulsants
Carbamazepine
Oxcarbazepine
Phenobarbital
Phenytoin
Potential for significant decreases in rilpivirine plasma concentrations due to CYP3A enzyme induction, which may result in loss of virologic response.
Antimycobacterials
Rifampin
Rifapentine
Glucocorticoid
(systemic)
Dexamethasone
(more than a
single-dose treatment)
Herbal Products
St John's wort (Hypericum perforatum)
Proton Pump
Inhibitors
e.g.,Esomeprazole
Lansoprazole
Omeprazole
Pantoprazole
Rabeprazole
Potential for significant decreases in rilpivirine plasma concentrations due to gastric pH increase, which may result in loss of virologic response.
-
5 WARNINGS AND PRECAUTIONS
5.1 Skin and Hypersensitivity Reactions
Severe skin and hypersensitivity reactions have been reported during the postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. While some skin reactions were accompanied by constitutional symptoms such as fever, other skin reactions were associated with organ dysfunctions, including elevations in hepatic serum biochemistries. During the Phase 3 clinical trials, treatment-related rashes with at least Grade 2 severity were reported in 3% of subjects receiving rilpivirine. No Grade 4 rash was reported. Overall, most rashes were Grade 1 or 2 and occurred in the first four to six weeks of therapy [see Adverse Reactions (6.1 and 6.2)]. Discontinue rilpivirine tablets immediately if signs or symptoms of severe skin or hypersensitivity reactions develop, including but not limited to, severe rash or rash accompanied by fever, blisters, mucosal involvement, conjunctivitis, facial edema, angioedema, hepatitis or eosinophilia. Clinical status including laboratory parameters should be monitored and appropriate therapy should be initiated.
5.2 Hepatotoxicity
Hepatic adverse events have been reported in patients receiving a rilpivirine-containing regimen. Patients with underlying hepatitis B or C virus infection, or marked elevations in transaminases prior to treatment may be at increased risk for worsening or development of transaminase elevations with use of rilpivirine. A few cases of hepatic toxicity have been reported in adult patients receiving a rilpivirine-containing regimen who had no pre-existing hepatic disease or other identifiable risk factors. Appropriate laboratory testing prior to initiating therapy and monitoring for hepatotoxicity during therapy with rilpivirine tablets are recommended in patients with underlying hepatic disease such as hepatitis B or C virus infection, or in patients with marked elevations in transaminases prior to treatment initiation. Liver enzyme monitoring should also be considered for patients without pre-existing hepatic dysfunction or other risk factors.
5.3 Depressive Disorders
The adverse reaction depressive disorders (depressed mood, depression, dysphoria, major depression, mood altered, negative thoughts, suicide attempt, suicidal ideation) has been reported with rilpivirine tablets. Patients with severe depressive symptoms should seek immediate medical evaluation to assess the possibility that the symptoms are related to rilpivirine tablets, and if so, to determine whether the risks of continued therapy outweigh the benefits
During the Phase 3 trials in adults (N=1368) through 96 weeks, the incidence of depressive disorders (regardless of causality, severity) reported among rilpivirine (n=686) or efavirenz (n=682) was 9% and 8%, respectively. Most events were mild or moderate in severity. The incidence of Grade 3 and 4 depressive disorders (regardless of causality) was 1% for both rilpivirine and efavirenz. The incidence of discontinuation due to depressive disorders among rilpivirine or efavirenz was 1% in each arm. Suicidal ideation was reported in 4 subjects in each arm while suicide attempt was reported in 2 subjects in the rilpivirine arm
During the Phase 2 trial in pediatric subjects 12 to less than 18 years of age (N=36) receiving rilpivirine through 48 weeks, the incidence of depressive disorders (regardless of causality, severity) was 19.4% (7/36). Most events were mild or moderate in severity. The incidence of Grade 3 and 4 depressive disorders (regardless of causality) was 5.6% (2/36). None of the subjects discontinued due to depressive disorders. Suicidal ideation and suicide attempt were reported in 1 subject.
5.4 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
The concomitant use of rilpivirine and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7), Contraindications (4), and Drug Interactions (7)]:
- Loss of therapeutic effect of rilpivirine and possible development of resistance.
In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in rilpivirine tablets) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to rilpivirine when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2)].
See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during rilpivirine therapy and review concomitant medications during therapy.
5.5 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including rilpivirine. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia or tuberculosis), which may necessitate further evaluation and treatment
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barre syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
5.6 Different Formulations Are Not Substitutable
Rilpivirine tablets and EDURANT PED have differing pharmacokinetic profiles and are not substitutable on a milligram-per-milligram basis. Incorrect dosing of a given formulation may result in underdosing and loss of therapeutic effect and possible development of resistance or possible clinically significant adverse reactions from greater exposure to rilpivirine.
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed below and in other sections of the labeling:
- Skin and Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Depressive Disorders [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Clinical Trials Experience in Adults
The safety assessment is based on the Week 96 pooled data from 1368 patients in the Phase 3 controlled trials TMC278-C209 (ECHO) and TMC278-C215 (THRIVE) in antiretroviral treatment-naïve HIV-1 infected adult patients, 686 of whom received rilpivirine (25 mg once daily) [see Clinical Studies (14.1)]. The median duration of exposure for patients in the rilpivirine arm and efavirenz arm was 104.3 and 104.1 weeks, respectively. Most adverse reactions occurred in the first 48 weeks of treatment. The proportion of subjects who discontinued treatment with rilpivirine tablets or efavirenz due to adverse reaction, regardless of severity, was 2% and 4%, respectively. The most common adverse reactions leading to discontinuation were psychiatric disorders: 10 (1%) subjects in the rilpivirine arm and 11 (2%) subjects in the efavirenz arm. Rash led to discontinuation in 1 (<1%) subject in the rilpivirine arm and 10 (2%) subjects in the efavirenz arm
Common Adverse Reactions
Adverse reactions of at least moderate intensity (≥Grade 2) reported in at least 2% of adult subjects are presented in Table 3. Selected laboratory abnormalities are included in Table 4.
Table 3: Selected Adverse Reactions of at Least Moderate Intensity* (Grades 2–4) Occurring in at Least 2% of Antiretroviral Treatment-Naïve HIV-1 Infected Adult Subjects (Week 96 Analysis) N=total number of subjects per treatment group; BR=background regimen
* Intensities are defined as follows: Moderate (discomfort enough to cause interference with usual activity); Severe (incapacitating with inability to work or do usual activity).
† Includes adverse reactions reported as depressed mood, depression, dysphoria, major depression, mood altered, negative thoughts, suicide attempt, suicide ideation.
System Organ Class,
Preferred Term,
%
Pooled Data from the Phase 3 TMC278-C209 and
TMC278-C215 Trials
Rilpivirine + BR
N=686
Efavirenz + BR
N=682
Gastrointestinal Disorders
Abdominal pain
2%
2%
Nausea
1%
3%
Vomiting
1%
2%
General Disorders and Administration Site Conditions
Fatigue
2%
2%
Nervous System Disorders
Headache
3%
4%
Dizziness
1%
7%
Psychiatric Disorders
Depressive disorders†
5%
4%
Insomnia
3%
4%
Abnormal dreams
2%
4%
Skin and Subcutaneous Tissue Disorders
Rash
3%
11%
No new adverse reaction terms were identified in adult subjects in the Phase 3 TMC278-C209 and TMC278-C215 trials between 48 weeks and 96 weeks nor in the Phase 2b TMC278-C204 trial through 240 weeks. The incidence of adverse events in the Phase 2b TMC278-C204 trial was similar to the Phase 3 trials through 96 weeks.
Less Common Adverse Reactions
Adverse reactions of at least moderate intensity (≥Grade 2) occurring in less than 2% of antiretroviral treatment-naïve subjects receiving rilpivirine are listed below by System Organ Class. Some adverse events have been included because of investigator's assessment of potential causal relationship and were considered serious or have been reported in more than 1 subject treated with rilpivirine
Gastrointestinal Disorders: diarrhea, abdominal discomfort
Hepatobiliary Disorders: cholecystitis, cholelithiasis
Metabolism and Nutrition Disorders: decreased appetite
Nervous System Disorders: somnolence
Psychiatric Disorders: sleep disorders, anxiety
Renal and Urinary Disorders: glomerulonephritis membranous, glomerulonephritis mesangioproliferative, nephrolithiasis
Laboratory Abnormalities in Treatment-Naïve Subjects
The percentage of subjects treated with rilpivirine or efavirenz in the Phase 3 trials with selected laboratory abnormalities (Grades 1 to 4), representing worst Grade toxicity are shown in Table 4.
Table 4: Selected Changes in Laboratory Parameters (Grades 1 to 4) Observed in Antiretroviral Treatment-Naïve HIV-1-Infected Adult Subjects (Week 96 Analysis) BR=background regimen; ULN=upper limit of normal
N=number of subjects per treatment group
Note: Percentages were calculated versus the number of subjects in ITT.
Laboratory Parameter
Abnormality, (%)
DAIDS Toxicity
Range
Pooled Data from the Phase 3 TMC278-C209 and TMC278-C215 Trials
Rilpivirine + BR
N=686
Efavirenz + BR
N=682
BIOCHEMISTRY
Increased Creatinine
Grade 1
≥1.1-≤1.3 x ULN
6%
1%
Grade 2
>1.3-≤1.8 x ULN
1%
1%
Grade 3
>1.8-≤3.4 x ULN
<1%
0
Grade 4
>3.4 x ULN
0
<1%
Increased AST
Grade 1
≥1.25-≤2.5 x ULN
16%
19%
Grade 2
>2.5-≤5.0 x ULN
4%
7%
Grade 3
>5.0-≤10.0 x ULN
2%
2%
Grade 4
>10.0 x ULN
1%
1%
Increased ALT
Grade 1
≥1.25-≤2.5 x ULN
18%
20%
Grade 2
>2.5-≤5.0 x ULN
5%
7%
Grade 3
>5.0-≤10.0 x ULN
1%
2%
Grade 4
>10.0 x ULN
1%
1%
Increased Total Bilirubin
Grade 1
≥1.1-≤1.5 x ULN
5%
<1%
Grade 2
>1.5-≤2.5 x ULN
3%
1%
Grade 3
>2.5-≤5.0 x ULN
1%
<1%
Grade 4
>5.0 x ULN
0
0
Increased Total Cholesterol (fasted)
Grade 1
5.18-6.19 mmol/L
200-239 mg/dL
17%
31%
Grade 2
6.20-7.77 mmol/L
240-300 mg/dL
7%
19%
Grade 3
>7.77 mmol/L >300 mg/dL
<1%
3%
Increased LDL Cholesterol (fasted)
Grade 1
3.37-4.12 mmol/L
130-159 mg/dL
14%
26%
Grade 2
4.13-4.90 mmol/L
160-190 mg/dL
5%
13%
Grade 3
≥4.91 mmol/L
≥191 mg/dL
1%
5%
Increased Triglycerides
(fasted)
Grade 2
5.65-8.48 mmol/L
500-750 mg/dL
2%
2%
Grade 3
8.49-13.56 mmol/L
751-1,200 mg/dL
1%
3%
Grade 4
>13.56 mmol/L
>1,200 mg/dL
0
1%
In the pooled Phase 3 trials, at Week 96, there was an overall mean change from baseline in basal cortisol of -0.69 (-1.12, 0.27) micrograms/dL in the rilpivirine group and of -0.02 (-0.48, 0.44) micrograms/dL in the efavirenz group
In the rilpivirine group, 43/588 (7%) of subjects with a normal 250 micrograms ACTH stimulation test at baseline developed an abnormal 250 micrograms ACTH stimulation test (peak cortisol level <18.1 micrograms/dL) during the trial compared to 18/561 (3%) in the efavirenz group. Of the subjects who developed an abnormal 250 micrograms ACTH stimulation test during the trial, fourteen subjects in the rilpivirine group and nine subjects in the efavirenz group had an abnormal 250 micrograms ACTH stimulation test at Week 96. Overall, there were no serious adverse events, deaths, or treatment discontinuations that could clearly be attributed to adrenal insufficiency. The clinical significance of the higher abnormal rate of 250 micrograms ACTH stimulation tests in the rilpivirine group is not known.
Serum Creatinine
In the pooled Phase 3 trials, an increase in serum creatinine was observed over the 96 weeks of treatment with rilpivirine. Most of this increase occurred within the first four weeks of treatment, with a mean change of 0.1 mg/dL (range: -0.3 mg/dL to 0.6 mg/dL) observed after 96 weeks of treatment. In subjects who entered the trial with mild or moderate renal impairment, the serum creatinine increase observed was similar to that seen in subjects with normal renal function. These changes are not considered to be clinically relevant and no subject discontinued treatment due to increases in serum creatinine. Serum creatinine increases occurred regardless of the background N(t)RTI regimen.
Serum Lipids
Changes from baseline in total cholesterol, LDL-cholesterol, HDL-cholesterol and triglycerides are presented in Table 5. The clinical benefit of these findings has not been demonstrated.
Table 5: Lipid Values, Mean Change from Baseline* N=number of subjects per treatment group; BR=background regimen
*Excludes subjects who received lipid lowering agents during the treatment period
†The change from baseline is the mean of within-patient changes from baseline for patients with both baseline and Week 96 values
Pooled Data from the Week 96 Analysis of the Phase 3 TMC278-C209 and
TMC278-C215 Trials
Rilpivirine + BR
Efavirenz + BR
N
Baseline
Week 96
N
Baseline
Week 96
Mean
(95% CI)
Mean (mg/dL)
Mean (mg/dL)
Mean
Change† (mg/dL)
Mean (mg/dL)
Mean (mg/dL)
Mean
Change† (mg/dL)
Total Cholesterol
(fasted)
546
161
166
5
507
160
187
28
HDL-cholesterol
(fasted)
545
41
46
4
505
40
51
11
LDL-cholesterol (fasted)
543
96
98
1
503
95
109
14
Triglycerides
(fasted)
546
122
116
-6
507
130
141
11
Subjects Co-infected with Hepatitis B and/or Hepatitis C Virus
In subjects co-infected with hepatitis B or C virus receiving rilpivirine, the incidence of hepatic enzyme elevation was higher than in subjects receiving rilpivirine who were not co-infected. This observation was the same in the efavirenz arm. The pharmacokinetic exposure of rilpivirine in co-infected subjects was comparable to that in subjects without co-infection.
Use in Combination with Cabotegravir
Safety findings from Phase 3/3b trials in adults were similar when rilpivirine was administered in combination with VOCABRIA (cabotegravir) or other antiretrovirals. See full prescribing information for VOCABRIA and CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) for additional information.
Clinical Trials Experience in Pediatric Patients
Pediatric Population (≥12 to less than 18 years of age)
Trial TMC278-C213 Cohort 1
The safety assessment is based on the Week 48 analysis of the single-arm, open-label, Phase 2 trial, TMC278-C213 Cohort 1, in which 36 antiretroviral treatment-naïve HIV-1 infected patients 12 to less than 18 years of age and weighing at least 32 kg received rilpivirine tablet (25 mg once daily) in combination with other antiretroviral agents [see Clinical Studies (14.3)]. The median duration of exposure was 63.5 weeks. There were no patients who discontinued treatment due to adverse reactions. No new adverse reactions were identified compared to those seen in adults.
Adverse reactions were reported in nineteen pediatric subjects (53%). Most adverse reactions were Grade 1 or 2. The most common adverse reactions reported in at least 2 subjects (regardless of severity) include headache (19%), depression (19%), somnolence (14%), nausea (11%), dizziness (8%), abdominal pain (8%), vomiting (6%) and rash (6%).
Observed laboratory abnormalities were comparable to those in adults.
Adrenal Function
In trial TMC278-C213 Cohort 1, at Week 48, the overall mean change from baseline in basal cortisol showed an increase of 1.59 (0.24, 2.93) micrograms/dL.
Six of 30 (20%) subjects with a normal 250 micrograms ACTH stimulation test at baseline developed an abnormal 250 micrograms ACTH stimulation test (peak cortisol level <18.1 micrograms/dL) during the trial. Three of these subjects had an abnormal 250 micrograms ACTH stimulation test at Week 48. Overall, there were no serious adverse events, deaths, or treatment discontinuations that could clearly be attributed to adrenal insufficiency. The clinical significance of the abnormal 250 micrograms ACTH stimulation tests is not known.
Trial 208580 [MOCHA]
Based on data from the Week 16 analysis of the MOCHA trial in 15 adolescents (12 to less than 18 years of age and weighing ≥35 kg) receiving rilpivirine tablets (25 mg once daily) in addition to continuing background antiretroviral therapy, the safety profile during the oral lead-in period in adolescents was consistent with the safety profile established with rilpivirine in adults.
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
6.2 Postmarketing Experience
Adverse reactions have been identified during postmarketing experience in patients receiving a rilpivirine containing regimen. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Renal and Genitourinary Disorders: nephrotic syndrome
Skin and Subcutaneous Tissue Disorders: Severe skin and hypersensitivity reactions including DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms)
-
7 DRUG INTERACTIONS
Rilpivirine is primarily metabolized by cytochrome P450 (CYP)3A, and drugs that induce or inhibit CYP3A may thus affect the clearance of rilpivirine. Coadministration of rilpivirine tablets and drugs that induce CYP3A may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine or to the class of NNRTIs. Coadministration of rilpivirine tablets and drugs that inhibit CYP3A may result in increased plasma concentrations of rilpivirine. Coadministration of rilpivirine tablets with drugs that increase gastric pH may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine or to the class of NNRTIs.
Rilpivirine tablets at the recommended doses are not likely to have a clinically relevant effect on the exposure of drugs metabolized by CYP enzymes.
Table 6 shows the established and other potentially significant drug interactions based on which alterations in dose or regimen of rilpivirine tablets and/or coadministered drug may be recommended. Drugs that are not recommended for coadministration with rilpivirine tablets are also included in Table 6. [see Dosage and Administration (2), Contraindications (4), and Clinical Pharmacology (12.3)].
Table 6: Established and Other Potentially Significant Drug Interactions: Alterations in Dose or Regimen May Be Recommended Based on Drug Interaction Studies or Predicted Interaction [see Clinical Pharmacology (12.3)] ↑=increase, ↓=decrease, ↔=no change
*The interaction between rilpivirine tablets and the drug was evaluated in a clinical study. All other drug-drug interactions shown are predicted.
†This interaction study has been performed with a dose higher than the recommended dose for rilpivirine tablets assessing the maximal effect on the coadministered drug. The dosing recommendation is applicable to the recommended doses of rilpivirine once daily.
Concomitant Drug Class: Drug Name
Effect on
Concentration of
Rilpivirine or
Concomitant Drug
Clinical Comment
Antacids:
antacids (e.g., aluminum or magnesium hydroxide, calcium carbonate)
↔ rilpivirine (antacids taken at least 2 hours before or at least 4 hours after rilpivirine)
↓ rilpivirine
(concomitant intake)
The combination of rilpivirine tablets and antacids should be used with caution as coadministration may cause significant decreases in rilpivirine plasma concentrations (increase in gastric pH). Antacids should only be administered either at least 2 hours before or at least 4 hours after rilpivirine tablets.
Anticonvulsants:
carbamazepine
oxcarbazepine
phenobarbital
phenytoin
↓ rilpivirine
Coadministration is contraindicated with rilpivirine tablets [see Contraindications (4)]
Antimycobacterials:
rifampin
rifapentine
↓ rilpivirine
Coadministration is contraindicated with rilpivirine tablets [see Contraindications (4)]
Antimycobacterials:
rifabutin*
↓ rilpivirine
Concomitant use of rilpivirine tablets with rifabutin may cause a decrease in the plasma concentrations of rilpivirine (induction of CYP3A enzymes). Throughout coadministration of rilpivirine tablets with rifabutin, the rilpivirine tablets dose should be increased from 25 mg once daily to 50 mg once daily. When rifabutin coadministration is stopped, the rilpivirine tablets dose should be decreased to 25 mg once daily.
Azole Antifungal Agents:
fluconazole
itraconazole
ketoconazole*†
posaconazole
voriconazole
↑ rilpivirine
↓ ketoconazole
Concomitant use of rilpivirine tablets with azole antifungal agents may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No rilpivirine dose adjustment is required when rilpivirine tablets are coadministered with azole antifungal agents. Clinically monitor for breakthrough fungal infections when azole antifungals are coadministered with rilpivirine tablets.
Glucocorticoid (systemic):
dexamethasone
(more than a single-dose treatment)
↓ rilpivirine
Coadministration is contraindicated with rilpivirine tablets [see Contraindications (4)]
H2-Receptor Antagonists:
cimetidine
famotidine*†
nizatidine
ranitidine
↔ rilpivirine
(famotidine taken 12 hours before rilpivirine or 4 hours after rilpivirine)
↓ rilpivirine
(famotidine taken 2 hours before rilpivirine)
The combination of rilpivirine tablets and H2-receptor antagonists should be used with caution as coadministration may cause significant decreases in rilpivirine plasma concentrations (increase in gastric pH). H2-receptor antagonists should only be administered at least 12 hours before or at least 4 hours after rilpivirine tablets.
Herbal Products:
St. John's wort
(Hypericum
perforatum)
↓ rilpivirine
Coadministration is contraindicated with rilpivirine tablets [see Contraindications (4)]
HIV-Antiviral Agents: Non-nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
NNRTI
(delavirdine)
Other NNRTIs
(efavirenz, etravirine, nevirapine)
↑ rilpivirine
↔ delavirdine
↓ rilpivirine
↔ other NNRTIs
It is not recommended to coadminister rilpivirine tablets with delavirdine and other NNRTIs.
HIV-Antiviral Agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs)
didanosine*†
↔ rilpivirine
↔ didanosine
No dose adjustment is required when rilpivirine tablets are coadministered with didanosine. Didanosine is to be administered on an empty stomach and at least two hours before or at least four hours after rilpivirine tablets (which should be administered with a meal).
HIV-Antiviral Agents: Protease Inhibitors (PIs)-Boosted (i.e., with coadministration of low-dose ritonavir) or Unboosted (i.e., without coadministration of low-dose ritonavir)
darunavir/ritonavir*†
↑ rilpivirine
↔ boosted darunavir
Concomitant use of rilpivirine tablets with darunavir/ritonavir may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No dose adjustment is required when rilpivirine tablets are coadministered with darunavir/ritonavir.
Lopinavir/ritonavir*†
↑ rilpivirine
↔ boosted lopinavir
Concomitant use of rilpivirine tablets with lopinavir/ritonavir may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No dose adjustment is required when rilpivirine tablets are coadministered with lopinavir/ritonavir.
Other boosted PIs (atazanavir/ritonavir, fosamprenavir/ritonavir, saquinavir/ritonavir, tipranavir/ritonavir)
↑ rilpivirine
↔ boosted PI
Concomitant use of rilpivirine tablets with boosted PIs may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). Rilpivirine tablets are not expected to affect the plasma concentrations of coadministered PIs.
Unboosted PIs
(atazanavir,
fosamprenavir,
indinavir, nelfinavir)
↑ rilpivirine
↔ unboosted PI
Concomitant use of rilpivirine tablets with unboosted PIs may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). Rilpivirine tablets are not expected to affect the plasma concentrations of coadministered PIs.
Macrolide or ketolide antibiotics:
azithromycin
clarithromycin
erythromycin
↑ rilpivirine
↔ azithromycin
↔ clarithromycin
↔ erythromycin
Macrolides are expected to increase concentrations of rilpivirine and are associated with a risk of Torsade de Pointes [Warnings and Precautions (5.4)] . Where possible, consider alternatives, such as azithromycin, which increases rilpivirine concentrations less than other macrolides
Narcotic Analgesics:
methadone*
↓ R(-) methadone
↓ S(+) methadone
No dose adjustments are required when initiating coadministration of methadone with rilpivirine tablets. However, clinical monitoring is recommended as methadone maintenance therapy may need to be adjusted in some patients.
Proton Pump Inhibitors: e.g., esomeprazole lansoprazole
omeprazole
pantoprazole
rabeprazole
↓ rilpivirine
Coadministration is contraindicated with rilpivirine tablets [see Contraindications (4)]
In addition to the drugs included in Table 6, the interaction between rilpivirine tablets and the following drugs was evaluated in clinical studies and no dose adjustment is needed for either drug [see Clinical Pharmacology (12.3)]: acetaminophen, atorvastatin, chlorzoxazone, cabotegravir, ethinylestradiol, norethindrone, raltegravir, sildenafil, simeprevir and tenofovir disoproxil fumarate. Rilpivirine did not have a clinically significant effect on the pharmacokinetics of digoxin or metformin. No clinically relevant drug-drug interaction is expected when rilpivirine tablets are coadministered with maraviroc, ribavirin or the NRTIs abacavir, emtricitabine, lamivudine, stavudine and zidovudine.
QT Prolonging Drugs
There is limited information available on the potential for a pharmacodynamic interaction between rilpivirine and drugs that prolong the QTc interval of the electrocardiogram. In a study of healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in rilpivirine tablets) have been shown to prolong the QTc interval of the electrocardiogram [see Clinical Pharmacology (12.2)]. Consider alternatives to rilpivirine tablets when coadministered with a drug with a known risk of torsade de pointes.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to rilpivirine tablets during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) 1-800-258-4263.
Risk Summary
Available data from the APR show no difference in the overall risk of birth defects for rilpivirine compared with the background rate for major birth defects of 2.7% in the Metropolitan Atlanta Congenital Defects Program (MACDP) reference population (see Data). The rate of miscarriage is not reported in the APR. The estimated background rate of miscarriage in clinically recognized pregnancies in the U.S. general population is 15% to 20%. The background risk for major birth defects and miscarriage for the indicated population is unknown. Methodologic limitations of the APR include the use of MACDP as the external comparator group. The MACDP population is not disease-specific, evaluates women and infants from a limited geographic area, and does not include outcomes for births that occurred at <20 weeks gestation. In a clinical trial, total rilpivirine exposures were generally lower during pregnancy compared to the postpartum period (see Data).
In animal reproduction studies, no adverse developmental outcomes were observed when rilpivirine was administered orally at exposures up to 15 (rats) and 70 (rabbits) times the exposure in humans (≥12 years of age and weighing at least 32 kg) at the recommended dose of 25 mg once daily (see Data).
Clinical Considerations
Dosing During Pregnancy and the Postpartum Period
Based on the experience of HIV-1-infected pregnant women who completed a clinical trial through the postpartum period with a rilpivirine-based regimen, no dose adjustments are required for pregnant patients who are already on a stable rilpivirine tablets regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL). [see Dosage and Administration (2.5)]. Lower exposures of rilpivirine were observed during pregnancy, therefore viral load should be monitored closely [see Clinical Pharmacology (12.3)].
Data
Human Data
Based on prospective reports to the APR of over 550 exposures to rilpivirine during the first trimester of pregnancy resulting in live births, there was no significant difference between the overall risk of birth defects with rilpivirine compared to the background birth defect rate of 2.7% in the U.S. reference population of the MACDP. The prevalence of birth defects in live births was 1.4% (95% CI: 0.6% to 2.8%) and 1.5% (95% CI: 0.3% to 4.3%) following first and second/third trimester exposure, respectively, to rilpivirine-containing regimens.
Rilpivirine in combination with a background regimen was evaluated in a clinical trial of 19 HIV-1 infected pregnant women during the second and third trimesters and postpartum. Each of the women were on a rilpivirine-based regimen at the time of enrollment. Twelve subjects completed the trial through the postpartum period (6 -12 weeks after delivery) and pregnancy outcomes are missing for six subjects. The exposure (C0h and AUC) of total rilpivirine was approximately 30 to 40% lower during pregnancy compared with postpartum (6 -12 weeks). The protein binding of rilpivirine was similar (>99%) during second trimester, third trimester, and postpartum period. One subject discontinued the trial following spontaneous termination of the pregnancy at 25 weeks gestation due to suspected premature rupture of membranes. Among the 12 subjects who were virologically suppressed at baseline (less than 50 copies/mL), virologic response was preserved in 10 subjects (83.3%) through the third trimester visit and in 9 subjects (75%) through the 6 to12 week postpartum visit. Virologic outcomes during the third trimester visit were missing for two subjects who were withdrawn (one subject was nonadherent to the study drug and one subject withdrew consent). Among the 10 infants with HIV test results available, born to 10 HIV-infected pregnant women, all had test results that were negative for HIV-1 at the time of delivery and up to 16 weeks postpartum. All 10 infants received antiretroviral prophylactic treatment with zidovudine. Rilpivirine was well tolerated during pregnancy and postpartum. There were no new safety findings compared with the known safety profile of rilpivirine in HIV-1-infected adults.
Animal Data
Rilpivirine was administered orally to pregnant rats (40, 120, or 400 mg per kg per day) and rabbits (5, 10, or 20 mg per kg per day) through organogenesis (on gestation Days 6 through 17, and 6 through 19, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with rilpivirine in rats and rabbits at exposures 15 (rats) and 70 (rabbits) times higher than the exposure in humans (≥12 years of age and weighing >32 kg) at the recommended dose of 25 mg once daily. In a pre- and postnatal development study, rilpivirine was administered orally up to 400 mg/kg/day through lactation. No adverse effects were noted in the offspring at maternal exposures up to 63 times the exposure in humans (≥12 years of age and weighing >32 kg) at the recommended dose of 25 mg daily.
8.2 Lactation
Based on limited data after oral administration, rilpivirine is present in human breast milk. The data do not allow determination of the amount of rilpivirine that is transferred to milk. There are no data on the effects on a breastfed infant, or the effects on milk production. Rilpivirine is present in rat milk (see Data). Potential risks of breastfeeding include: ((1) HIV transmission (in HIV-negative infants), (2) developing viral resistance (in HIV-positive infants), and (3) adverse reactions in a breastfed infant similar to those seen in adults
Data
Animal Data
Animal lactation studies with rilpivirine have not been conducted. However, rilpivirine was detected in the plasma of nursing pups on lactation day 7 in the rat pre- and postnatal development study.
8.4 Pediatric Use
The safety and effectiveness of rilpivirine tablets has been established for the treatment of HIV-1 infection in treatment-naïve pediatric patients 12 years of age and older and weighing at least 35 kg. Use of rilpivirine tablets in this population is supported by two trials: TMC278-C213 and MOCHA.
Trial TMC278-C213
TMC278-C213 was a single arm, open-label, Phase 2 trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects, and was divided into two Cohorts.
- Cohort 1 evaluated the safety, efficacy and pharmacokinetics of rilpivirine and enrolled 36 children aged 12 to less than 18 years of age and weighing at least 32 kg [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.3)].
The safety, tolerability, and pharmacokinetics of oral and injectable cabotegravir and oral and injectable rilpivirine are being assessed in an ongoing Phase 1/2 multicenter, open-label, non comparative study, MOCHA (IMPAACT 2017) [see Adverse Reactions (6.1)]. Refer to the VOCABRIA and CABENUVA prescribing information for additional information when rilpivirine is used in combination with cabotegravir.
The safety and effectivness of rilpivirine in these pediatric subjects were similar to that seen in adults, and there were no significant changes on rilpivirine exposures [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients less than 2 years of age or weighing less than 14 kg have not been established.
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
8.5 Geriatric Use
Clinical studies of rilpivirine tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, caution should be exercised in the administration and monitoring of rilpivirine tablets in elderly patients reflecting the greater frequency of decreased renal and hepatic function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
No dose adjustment of rilpivirine tablets are required in patients with mild or moderate renal impairment. However, in patients with severe renal impairment or end-stage renal disease, rilpivirine should be used with caution and with increased monitoring for adverse effects, as rilpivirine concentrations may be increased due to alteration of drug absorption, distribution, and metabolism secondary to renal dysfunction. As rilpivirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dosage adjustment of rilpivirine tablets are required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. Rilpivirine has not been studied in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
There is no specific antidote for overdose with rilpivirine tablets. Human experience of overdose with rilpivirine tablets are limited. Treatment of overdose with rilpivirine tablets consists of general supportive measures including monitoring of vital signs and ECG (QT interval) as well as observation of the clinical status of the patient. It is advisable to contact a poison control center to obtain the latest recommendations for the management of an overdose. Since rilpivirine is highly bound to plasma protein, dialysis is unlikely to result in significant removal of the active substance.
-
11 DESCRIPTION
Rilpivirine tablets are a non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1).
The chemical name for rilpivirine hydrochloride is 4-[[4-[[4-[(E)-2-cyanoethenyl]-2,6-dimethylphenyl]amino]-2-pyrimidinyl]amino]benzonitrile monohydrochloride. Its molecular formula is C22H18N6 HCl and its molecular weight is 402.88. Rilpivirine hydrochloride has the following structural formula:
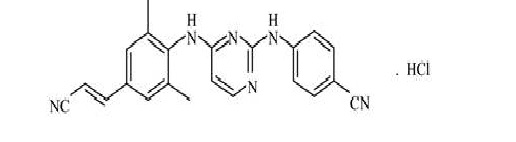
Rilpivirine hydrochloride is a white to almost white powder. Rilpivirine hydrochloride is practically insoluble in water over a wide pH range.
Rilpivirine 25 mg tablets are available as a white to off-white, film-coated, round, biconvex, 6.5 mm tablet for oral administration. Each tablet contains 27.5 mg of rilpivirine hydrochloride, which is equivalent to 25 mg of rilpivirine. Each rilpivirine 25 mg tablet also contains the inactive ingredients croscarmellose sodium, lactose monohydrate, magnesium stearate, polysorbate 20, povidone K30 and silicified microcrystalline cellulose. The tablet coating contains hypromellose 2910 6 mPa.s, lactose monohydrate, polyethylene glycol 3000, titanium dioxide and triacetin.
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
The effect of rilpivirine tablets at the recommended dose of 25 mg once daily on the QTcF interval was evaluated in a randomized, placebo and active (moxifloxacin 400 mg once daily) controlled crossover study in 60 healthy adults, with 13 measurements over 24 hours at steady state. The maximum mean time-matched (95% upper confidence bound) differences in QTcF interval from placebo after baseline-correction was 2.0 (5.0) milliseconds (i.e., below the threshold of clinical concern).
When doses of 75 mg once daily and 300 mg once daily of rilpivirine tablets (3 times and 12 times the dose in rilpivirine tablets) were studied in healthy adults, the maximum mean time matched (95% upper confidence bound) differences in QTcF interval from placebo after baseline-correction were 10.7 (15.3) and 23.3 (28.4) milliseconds, respectively. Steady-state administration of rilpivirine tablet 75 mg once daily and 300 mg once daily resulted in a mean steady-state Cmax approximately 2.6-fold and 6.7-fold, respectively, higher than the mean Cmax observed with the recommended 25 mg once daily dose of rilpivirine tablets [see Warnings and Precautions (5.4)].
12.3 Pharmacokinetics
The pharmacokinetic properties of rilpivirine have been evaluated in adult healthy subjects and in adult antiretroviral treatment-naïve HIV-1-infected subjects. Exposure to rilpivirine was generally lower in HIV-1 infected subjects than in healthy subjects.
Table 7: Pharmacokinetic Estimates of Rilpivirine 25 mg Once Daily in Antiretroviral Treatment-Naïve HIV-1-Infected Adult Subjects (Pooled Data from Phase 3 Trials through Week 96) Parameter
Rilpivirine 25 mg once daily
N=679
AUC24h (ng.h/mL)
Mean±Standard Deviation
2235±851
Median (Range)
2096 (198 - 7307)
C0h (ng/mL)
Mean±Standard Deviation
79±35
Median (Range)
73 (2 - 288)
Absorption and Bioavailability
After oral administration, the maximum plasma concentration of rilpivirine is generally achieved within 4 - 5 hours. The absolute bioavailability of rilpivirine is unknown.
Effects of Food on Oral Absorption
The exposure to rilpivirine was approximately 40% lower when rilpivirine tablets was taken in a fasted condition as compared to a normal caloric meal (533 kcal) or high-fat high-caloric meal (928 kcal). When rilpivirine tablets were taken with only a protein-rich nutritional drink, exposures were 50% lower than when taken with a meal.
Distribution
Rilpivirine is approximately 99.7% bound to plasma proteins in vitro, primarily to albumin. The distribution of rilpivirine into compartments other than plasma (e.g., cerebrospinal fluid, genital tract secretions) has not been evaluated in humans.
Metabolism
In vitro experiments indicate that rilpivirine primarily undergoes oxidative metabolism mediated by the cytochrome P450 (CYP) 3A system.
Elimination
The terminal elimination half-life of rilpivirine is approximately 50 hours. After single dose oral administration of 14C-rilpivirine, on average 85% and 6.1% of the radioactivity could be retrieved in feces and urine, respectively. In feces, unchanged rilpivirine accounted for on average 25% of the administered dose. Only trace amounts of unchanged rilpivirine (<1% of dose) were detected in urine.
Specific Populations
Pregnancy and Postpartum
The exposure (C0h and AUC24h) to total rilpivirine after intake of rilpivirine 25 mg once daily as part of an antiretroviral regimen was 30 to 40% lower during pregnancy (similar for the second and third trimester), compared with postpartum (see Table 8). However, the exposure during pregnancy was not significantly different from exposures obtained in Phase 3 trials. Based on the exposure-response relationship for rilpivirine, this decrease is not considered clinically relevant in patients who are virollogically suppressed. The protein binding of rilpivirine was similar (>99%) during the second trimester, third trimester, and postpartum.
Table 8: Pharmacokinetic Results of Total Rilpivirine After Administration of Rilpivirine 25 mg Once Daily as Part of an Antiretroviral Regimen, During the 2nd Trimester of Pregnancy, the 3rd Trimester of Pregnancy and Postpartum Pharmacokinetics of total rilpivirine (mean ± SD, tmax:
median [range])
Postpartum
(6 - 12 Weeks)
(n=11)
2nd Trimester of pregnancy
(n=15)
3rd Trimester of pregnancy
(n=13)
C0h, ng/mL
111±69.2
65.0±23.9
63.5±26.2
Cmin, ng/mL
84.0±58.8
54.3±25.8
52.9±24.4
Cmax, ng/mL
167±101
121±45.9
123±47.5
tmax, h
4.00 (2.03-25.08)
4.00 (1.00-9.00)
4.00 (2.00-24.93)
AUC24h, ng.h/mL
2714±1535
1792±711
1762±662
The pharmacokinetics of rilpivirine in HIV-1 infected pediatric patients 12 to less than 18 years of age receiving the recommended dosing regimen of rilpivirine tablets were comparable than those obtained in treatment-naïve HIV-1 infected adult patients (see Table 9).
Table 9: Pharmacokinetic Estimates of Rilpivirine After Administration of the Recommended Daily Oral Dosing Regimen in Pediatric Patients ≥12 to <18 Years (Trial TMC278-C213)a a The 25 mg dose was administered as one 25 mg tablet.
Pharmacokinetics of
rilpivirine
Mean±SD
Median (range)
25 mg once daily
N
34
AUC24h (ng.h/mL)
2424±1024
2269 (417 - 5166)
C0h (ng/mL)
85±40
79 (7 - 202)
Pharmacokinetic analysis indicated that rilpivirine exposure was similar in HIV-1 infected subjects with mild renal impairment relative to HIV-1 infected subjects with normal renal function. No dose adjustment is required in patients with mild renal impairment. There is limited or no information regarding the pharmacokinetics of rilpivirine in patients with moderate or severe renal impairment or in patients with end-stage renal disease, and rilpivirine concentrations may be increased due to alteration of drug absorption, distribution, and metabolism secondary to renal dysfunction. The potential impact is not expected to be of clinical relevance for HIV-1-infected subjects with moderate renal impairment, and no dose adjustment is required in these patients. Rilpivirine should be used with caution and with increased monitoring for adverse effects in patients with severe renal impairment or end-stage renal disease. As rilpivirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Use in Specific Populations (8.6)].
Hepatic Impairment
Rilpivirine is primarily metabolized and eliminated by the liver. In a study comparing 8 subjects with mild hepatic impairment (Child-Pugh score A) to 8 matched controls, and 8 subjects with moderate hepatic impairment (Child-Pugh score B) to 8 matched controls, the multiple dose exposure of rilpivirine was 47% higher in subjects with mild hepatic impairment and 5% higher in subjects with moderate hepatic impairment. Rilpivirine tablets has not been studied in subjects with severe hepatic impairment (Child-Pugh score C) [see Use in Specific Populations (8.7)].
Sex, Race, Hepatitis B and/or Hepatitis C Virus Co-infection
No clinically relevant differences in the pharmacokinetics of rilpivirine have been observed between sex, race and patients with hepatitis B and/or C-virus co-infection.
Drug Interactions
[see Contraindications (4) and Drug Interactions (7)].
Rilpivirine is primarily metabolized by cytochrome P450 (CYP)3A, and drugs that induce or inhibit CYP3A may thus affect the clearance of rilpivirine. Coadministration of rilpivirine and drugs that induce CYP3A may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance. Coadministration of rilpivirine and drugs that inhibit CYP3A may result in increased plasma concentrations of rilpivirine. Coadministration of rilpivirine with drugs that increase gastric pH may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine and to the class of NNRTIs.
Rilpivirine tablets at the recommended doses are not likely to have a clinically relevant effect on the exposure of medicinal products metabolized by CYP enzymes.
Drug interaction studies were performed with rilpivirine and other drugs likely to be coadministered or commonly used as probes for pharmacokinetic interactions. The effects of coadministration of other drugs on the Cmax, AUC, and Cmin values of rilpivirine are summarized in Table 11 (effect of other drugs on rilpivirine). The effect of coadministration of rilpivirine on the Cmax, AUC, and Cmin values of other drugs are summarized in Table 12 (effect of rilpivirine tablets on other drugs). [For information regarding clinical recommendations, see Drug Interactions (7)].
Table 11: Drug Interactions: Pharmacokinetic Parameters for Rilpivirine in the Presence of Coadministered Drugs CI=Confidence Interval; N=maximum number of subjects with data; N.A.=not available; ↑=increase; ↓=decrease; ↔=no change; q.d.=once daily; b.i.d.=twice daily
* Comparison based on historic controls
† This interaction study has been performed with a dose higher than the recommended dose for rilpivirine tablets assessing the maximal effect on the coadministered drug.
Coadministered Drug
Dose/Schedule
Mean Ratio of Rilpivirine Pharmacokinetic Parameters
With/Without Coadministered Drug (90% CI); No Effect=1.00
Coadministered Drug
Rilpivirine
N
Cmax
AUC
Cmin
Coadministration With HIV Protease Inhibitors (PIs)
Darunavir/ritonavir
800/100 mg q.d.
150 mg q.d.†
14
1.79
(1.56-2.06)
2.30
(1.98-2.67)
2.78
(2.39-3.24)
Lopinavir/ritonavir
(soft gel capsule)
400/100 mg b.i.d.
150 mg q.d.†
15
1.29
(1.18-1.40)
1.52
(1.36-1.70)
1.74
(1.46-2.08)
Coadministration With HIV Nucleoside or Nucleotide Reverse Transcriptase Inhibitors (NRTIs/N[t]RTIs)
Didanosine
400 mg q.d. delayed release capsules taken 2 hours before rilpivirine
150 mg
q.d.†
21
1.00
(0.90-1.10)
1.00
(0.95-1.06)
1.00
(0.92-1.09)
Tenofovir disoproxil fumarate
300 mg q.d.
150 mg
q.d.†
16
0.96
(0.81-1.13)
1.01
(0.87-1.18)
0.99
(0.83-1.16)
Coadministration With HIV Integrase Strand Transfer Inhibitors
Cabotegravir
30 mg q.d.
25 mg q.d.
11
0.96
(0.85-1.09)
0.99
(0.89-1.09)
0.92
(0.79-1.07)
Raltegravir
400 mg b.i.d.
25 mg q.d.
23
1.12
(1.04-1.20)
1.12
(1.05-1.19)
1.03
(0.96-1.12)
Coadministration With other Antivirals
Simeprevir
150 mg q.d.
25 mg q.d.
23
1.04
(0.95-1.13)
1.12
(1.05-1.19)
1.25
(1.16-1.35)
Coadministration With Drugs other than Antiretrovirals
Acetaminophen
500 mg single dose
150 mg
q.d.†
16
1.09
(1.01-1.18)
1.16
(1.10-1.22)
1.26
(1.16-1.38)
Atorvastatin
40 mg q.d.
150 mg
q.d.†
16
0.91
(0.79-1.06)
0.90
(0.81-0.99)
0.90
(0.84-0.96)
Chlorzoxazone
500 mg single dose taken 2 hours
after rilpivirine
150 mg
q.d.†
16
1.17
(1.08-1.27)
1.25
(1.16-1.35)
1.18
(1.09-1.28)
Ethinylestradiol/
Norethindrone
0.035 mg q.d./
1 mg q.d.
25 mg
q.d.
15
↔*
↔*
↔*
Famotidine
40 mg single dose taken 12 hours before rilpivirine
150 mg single
dose†
24
0.99
(0.84-1.16)
0.91
(0.78-1.07)
N.A.
Famotidine
40 mg single dose taken 2 hours before rilpivirine
150 mg single
dose†
23
0.15
(0.12-0.19)
0.24
(0.20-0.28)
N.A.
Famotidine
40 mg single dose taken 4 hours
after rilpivirine
150 mg single
dose†
24
1.21
(1.06-1.39)
1.13
(1.01-1.27)
N.A.
Ketoconazole
400 mg q.d.
150 mg
q.d.†
15
1.30
(1.13-1.48)
1.49
(1.31-1.70)
1.76
(1.57-1.97)
Methadone
60-100 mg q.d., individualized dose
25 mg
q.d.
12
↔*
↔*
↔*
Omeprazole
20 mg q.d.
150 mg q.d.†
16
0.60
(0.48-0.73)
0.60
(0.51-0.71)
0.67
(0.58-0.78)
Rifabutin
300 mg q.d.
25 mg q.d.
18
0.69
(0.62-0.76)
0.58
(0.52-0.65)
0.52
(0.46-0.59)
Rifabutin
300 mg q.d.
50 mg q.d.
18
1.43
(1.30-1.56)
1.16
(1.06-1.26)
0.93
(0.85-1.01)
(reference arm for comparison was 25 mg q.d. rilpivirine administered alone)
Rifampin
600 mg q.d.
150 mg
q.d.†
16
0.31
(0.27-0.36)
0.20
(0.18-0.23)
0.11
(0.10-0.13)
Sildenafil
50 mg single dose
75 mg
q.d.†
16
0.92
(0.85-0.99)
0.98
(0.92-1.05)
1.04
(0.98-1.09)
Table 12: Drug Interactions: Pharmacokinetic Parameters for Coadministered Drugs in the Presence of Rilpivirine CI=Confidence Interval; N=maximum number of subjects with data; N.A.=not available; ↑=increase; ↓=decrease; ↔=no change; q.d.=once daily; b.i.d.=twice daily
† This interaction study has been performed with a dose higher than the recommended dose for rilpivirine tablets (25 mg once daily) assessing the maximal effect on the coadministered drug.
# AUC(0-last)
^ N (maximum number of subjects with data) for AUC(0-∞)=15
Dose/Schedule
Mean Ratio of Coadministered Drug
Pharmacokinetic Parameters
With/Without Rilpivirine
(90% CI); No Effect=1.00
Coadministered
Drug
Coadministered Drug
Rilpivirine
N
Cmax
AUC
Cmin
Coadministration With HIV Protease Inhibitors (PIs)
Darunavir/ritonavir
800/100 mg q.d.
150 mg q.d.†
15
0.90
(0.81-1.00)
0.89
(0.81-0.99)
0.89
(0.68-1.16)
Lopinavir/ritonavir
(soft gel capsule)
400/100 mg b.i.d.
150 mg q.d.†
15
0.96
(0.88-1.05)
0.99
(0.89-1.10)
0.89
(0.73-1.08)
Coadministration With HIV Nucleoside or Nucleotide Reverse Transcriptase Inhibitors (NRTIs/N[t]RTIs)
Didanosine
400 mg q.d. delayed release capsules taken
2 hours before rilpivirine
150 mg q.d.†
13
0.96
(0.80-1.14)
1.12
(0.99-1.27)
N.A.
Tenofovir disoproxil fumarate
300 mg q.d.
150 mg q.d.†
16
1.19
(1.06-1.34)
1.23
(1.16-1.31)
1.24
(1.10-1.38)
Coadministration With HIV Integrase Strand Transfer Inhibitors
Cabotegravir
30 mg q.d.
25 mg q.d.
11
1.05
(0.96-1.15)
1.12
(1.05-1.19)
1.14
(1.04-1.24)
Raltegravir
400 mg b.i.d.
25 mg q.d.
23
1.10
(0.77-1.58)
1.09
(0.81-1.47)
1.27
(1.01-1.60)
Coadministration With other Antivirals
Simeprevir
150 mg q.d.
25 mg q.d.
21
1.10
(0.97-1.26)
1.06
(0.94-1.19)
0.96
(0.83-1.11)
Coadministration With Drugs other than Antiretrovirals
Acetaminophen
500 mg single dose
150 mg q.d.†
16
0.97
(0.86-1.10)
0.91
(0.86-0.97)
N.A.
Atorvastatin
40 mg q.d.
150 mg q.d.†
16
1.35
(1.08-1.68)
1.04
(0.97-1.12)
0.85
(0.69-1.03)
2-hydroxy-atorvastatin
16
1.58 (1.33-1.87)
1.39
(1.29-1.50)
1.32
(1.10-1.58)
4-hydroxy-atorvastatin
16
1.28 (1.15-1.43)
1.23 (1.13-1.33)
N.A.
Chlorzoxazone
500 mg single dose taken 2 hours after rilpivirine
150 mg q.d.†
16
0.98
(0.85-1.13)
1.03
(0.95-1.13)
N.A.
Digoxin
0.5 mg single dose
25 mg q.d.
22
1.06 (0.97-1.17)
0.98 (0.93-1.04)#
N.A.
Ethinylestradiol
0.035 mg q.d.
25 mg q.d.
17
1.17 (1.06-1.30)
1.14 (1.10-1.19)
1.09 (1.03-1.16)
Norethindrone
1 mg q.d.
17
0.94 (0.83-1.06)
0.89 (0.84-0.94)
0.99 (0.90-1.08)
Ketoconazole
400 mg q.d.
150 mg q.d.†
14
0.85 (0.80-0.90)
0.76 (0.70-0.82)
0.34 (0.25-0.46)
R(-) methadone
60-100 mg q.d., individualized dose
25 mg q.d.
13
0.86 (0.78-0.95)
0.84 (0.74-0.95)
0.78 (0.67-0.91)
S(+) methadone
13
0.87 (0.78-0.97)
0.84 (0.74-0.96)
0.79 (0.67-0.92)
Metformin
850 mg single dose
25 mg q.d.
20
1.02
(0.95-1.10)
0.97
(0.90-1.06)^
N.A.
Omeprazole
20 mg q.d.
150 mg q.d.†
15
0.86 (0.68-1.09)
0.86 (0.76-0.97)
N.A.
Rifampin
25-desacetylrifampin
600 mg q.d.
150 mg q.d.†
16
16
1.02 (0.93-1.12)
1.00 (0.87-1.15)
0.99 (0.92-1.07)
0.91 (0.77-1.07)
N.A.
N.A.
Sildenafil
N-desmethyl-sildenafil
50 mg single dose
75 mg q.d.†
16
16
0.93 (0.80-1.08)
0.90 (0.80-1.02)
0.97 (0.87-1.08)
0.92 (0.85-0.99) #
N.A.
N.A.
12.4 Microbiology
Rilpivirine is a diarylpyrimidine non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1) and inhibits HIV-1 replication by non-competitive inhibition of HIV-1 reverse transcriptase (RT). Rilpivirine does not inhibit the human cellular DNA polymerases α, β and γ.
Antiviral Activity in Cell Culture
Rilpivirine exhibited activity against laboratory strains of wild-type HIV-1 in an acutely infected T-cell line with a median EC50 value for HIV-1IIIB of 0.73 nM (0.27 ng/mL). Rilpivirine demonstrated limited activity in cell culture against HIV-2 with a median EC50 value of 5220 nM (range 2510 to 10830 nM) (920 to 3970 ng/mL).
Rilpivirine demonstrated antiviral activity against a broad panel of HIV-1 group M (subtype A, B, C, D, F, G, H) primary isolates with EC50 values ranging from 0.07 to 1.01 nM (0.03 to 0.37 ng/mL) and was less active against group O primary isolates with EC50 values ranging from 2.88 to 8.45 nM (1.06 to 3.10 ng/mL).
The antiviral activity of rilpivirine was not antagonistic when combined with the NNRTIs efavirenz, etravirine or nevirapine; the N(t)RTIs abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir or zidovudine; the PIs amprenavir, atazanavir, darunavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir or tipranavir; the fusion inhibitor enfuvirtide; the CCR5 co-receptor antagonist maraviroc, or the integrase strand transfer inhibitor raltegravir.
Resistance
In Cell Culture
Rilpivirine-resistant strains were selected in cell culture starting from wild-type HIV-1 of different origins and subtypes as well as NNRTI resistant HIV-1. The frequently observed amino acid substitutions that emerged and conferred decreased phenotypic susceptibility to rilpivirine included: L100I, K101E, V106I and A, V108I, E138K and G, Q, R, V179F and I, Y181C and I, V189I, G190E, H221Y, F227C and M230I and L.
In Treatment-Naïve Adult Subjects
In the Week 96 pooled resistance analysis of the Phase 3 trials TMC278-C209 and TMC278-C215, the emergence of resistance was greater among subjects' viruses in the rilpivirine arm compared to the efavirenz arm, and was dependent on baseline viral load. In the pooled resistance analysis, 58% (57/98) of the subjects who qualified for resistance analysis (resistance analysis subjects) in the rilpivirine arm had virus with genotypic and/or phenotypic resistance to rilpivirine compared to 45% (25/56) of the resistance analysis subjects in the efavirenz arm who had genotypic and/or phenotypic resistance to efavirenz. Moreover, genotypic and/or phenotypic resistance to a background drug (emtricitabine, lamivudine, tenofovir, abacavir or zidovudine) emerged in viruses from 52% (51/98) of the resistance analysis subjects in the rilpivirine arm compared to 23% (13/56) in the efavirenz arm.
Emerging NNRTI substitutions in the rilpivirine resistance analysis of subjects viruses included V90I, K101E/P/T, E138K/A/Q/G, V179I/L, Y181C/I, V189I, H221Y, F227C/L and M230L, which were associated with a rilpivirine phenotypic fold change range of 2.6 - 621. The E138K substitution emerged most frequently during rilpivirine treatment commonly in combination with the M184I substitution. The emtricitabine and lamivudine resistance-associated substitutions M184I or V and NRTI resistance-associated substitutions (K65R/N, A62V, D67N/G, K70E, Y115F, T215S/T, or K219E/R) emerged more frequently in rilpivirine resistance analysis subjects compared to efavirenz resistance analysis subjects (see Table 13).
NNRTI- and NRTI-resistance substitutions emerged less frequently in resistance analysis of viruses from subjects with baseline viral load of ≤100,000 copies/mL compared to viruses from subjects with baseline viral load of >100,000 copies/mL: 26% (14/54) compared to 74% (40/54) of NNRTI-resistance substitutions and 22% (11/50) compared to 78% (39/50) of NRTI-resistance substitutions. This difference was also observed for the individual emtricitabine/lamivudine and tenofovir resistance substitutions: 23% (11/47) compared to 77% (36/47) for M184I/V and 0% (0/8) compared to 100% (8/8) for K65R/N. Additionally, NNRTI- and NRTI-resistance substitutions emerged less frequently in the resistance analysis of viruses from subjects with baseline CD4+ cell counts ≥200 cells/mm3 compared to viruses from subjects with baseline CD4+ cell counts <200 cells/mm3: 37% (20/54) compared to 63% (34/54) of NNRTI-resistance substitutions and 28% (14/50) compared to 72% (36/50) of NRTI-resistance substitutions.
Table 13: Proportion of Resistance Analysis Subjects* with Frequently Emerging Reverse Transcriptase Substitutions from the Pooled Phase 3 TMC278-C209 and TMC278-C215 Trials in the Week 96 Analysis BR=background regimen
* Subjects who qualified for resistance analysis.
† V90, L100, K101, K103, V106, V108, E138, V179, Y181, Y188, V189, G190, H221, P225, F227 or M230
‡ This combination of NNRTI and NRTI substitutions is a subset of those with the E138K.
§ A62V, K65R/N, D67N/G, K70E, L74I, V75I, Y115F, M184I/V, L210F, T215S/T, K219E/R
¶ These substitutions emerged in addition to the primary substitutions M184V/I or K65R/N; A62V (n=3), D67N/G (n=3), K70E (n=4), Y115F (n=2), T215S/T (n=1),
K219E/R (n=8) in rilpivirine resistance analysis subjects.
TMC278-C209 and TMC278-C215
N=1368
Rilpivirine + BR
N=686
Efavirenz + BR
N=682
Subjects who Qualified for Resistance Analysis
15% (98/652)
9% (56/604)
Subjects with Evaluable Post-Baseline Resistance Data
87
43
Emerging NNRTI Substitutions†
Any
62% (54/87)
53% (23/43)
V90I
13% (11/87)
2% (1/43)
K101E/P/T/Q
20% (17/87)
9% (4/43)
K103N
1% (1/87)
40% (17/43)
E138K/A/Q/G
40% (35/87)
2% (1/43)
E138K + M184I‡
25% (22/87)
0
V179I/L/D
6% (5/87)
7% (3/43)
Y181C/I/S
10% (9/87)
2% (1/43)
V189I
8% (7/87)
2% (1/43)
H221Y
9% (8/87)
0
Emerging NRTI Substitutions§
Any
57% (50/87)
30% (13/43)
M184I/V
54% (47/87)
26% (11/43)
K65R/N
9% (8/87)
5% (2/43)
A62V, D67N/G, K70E, Y115F, T215S/T or K219E/R¶
21% (18/87)
2% (1/43)
Treatment--Naïve HIV-1-Infected Pediatric Subjects
In trial TMC278-C213 Cohort 1, a single-arm, open-label Phase 2 trial in antiretroviral treatment-naïve HIV-1-infected pediatric subjects ≥12 to less than 18 years [see Clinical Studies (14.3)], rilpivirine resistance associated substitutions were observed in 62.5% (5/8) of subjects with virologic failure and post-baseline genotypic data at 48 weeks with 4 of 5 having ≥2.5-fold decrease in susceptibility to rilpivirine. In addition, 4 of the 5 subjects with rilpivirine resistance substitutions also had at least 1 treatment-emergent resistance substitution to nucleos(t)ide reverse transcriptase inhibitors.
The emergent rilpivirine resistance-associated substitutions in pediatric patients are consistent with those seen in adults failing on a rilpivirine-containing regimen (see Table 13).
Cross-Resistance
Site-Directed NNRTI Mutant Virus
Cross-resistance has been observed among NNRTIs. The single NNRTI substitutions K101P, Y181I and Y181V conferred 52-fold, 15-fold and 12-fold decreased susceptibility to rilpivirine, respectively. The combination of E138K and M184I showed 6.7-fold reduced susceptibility to rilpivirine compared to 2.8-fold for E138K alone. The K103N substitution did not result in reduced susceptibility to rilpivirine by itself. However, the combination of K103N and L100I resulted in a 7-fold reduced susceptibility to rilpivirine. Combinations of 2 or 3 NNRTI resistance-associated substitutions had decreased susceptibility to rilpivirine (fold change range of 3.7 to 554) in 38% and 66% of mutants analyzed, respectively.
Treatment-Naïve HIV-1-Infected Adult Subjects
Considering all available cell culture and clinical data, any of the following amino acid substitutions, when present at baseline, are likely to decrease the antiviral activity of rilpivirine: K101E, K101P, E138A, E138G, E138K, E138R, E138Q, V179L, Y181C, Y181I, Y181V, Y188L, H221Y, F227C, M230I or M230L
Cross-resistance to efavirenz, etravirine and/or nevirapine is likely after virologic failure and development of rilpivirine resistance. In the Week 96 pooled analyses of the Phase 3 TMC278-C209 and TMC278-C215 clinical trials, 50 of the 87 (57%) rilpivirine resistance analysis subjects with post-baseline resistance data had virus with decreased susceptibility to rilpivirine (≥2.5-fold change). Of these, 86% (n=43/50) were resistant to efavirenz (≥3.3-fold change), 90% (n= 45/50) were resistant to etravirine (≥3.2-fold change) and 62% (n=31/50) were resistant to nevirapine (≥6-fold change). In the efavirenz arm, 3 of the 21 (14%) efavirenz resistance analysis subjects' viruses were resistant to etravirine and rilpivirine, and 95% (n=20/21) were resistant to nevirapine. Virus from subjects experiencing virologic failure on rilpivirine developed more NNRTI resistance-associated substitutions conferring more cross-resistance to the NNRTI class and had a higher likelihood of cross-resistance to all NNRTIs in the class compared to virus from subjects who failed on efavirenz.
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
Two-year carcinogenicity studies in mice and rats were conducted with rilpivirine. In rats, there were no drug related neoplasms at exposures 3 times those observed in humans (12 years of age and older and weighing greater than 32 kg) and ≥ 1.4 times relative to the predicted exposures in children (2 to less than 12 years of age, weighing at least 14 kg) at the recommended daily dose. In mice, rilpivirine was positive for hepatocellular neoplasms in both males and females. The observed hepatocellular findings in mice may be rodent-specific. At the lowest tested dose in the mouse carcinogenicity study, the systemic exposure to rilpivirine was 21 times that observed in humans (12 years of age and older and weighing greater than 32 kg) and ≥ 12 times relative to the predicted exposures in children (2 to less than 12 years of age, weighing at least 14 kg) at the recommended daily dose.
Rilpivirine was not genotoxic in the bacterial reverse mutation assay, mouse lymphoma assay, or in the in vivo rodent micronucleus assay.
Impairment of Fertility
In rat fertility and early embryonic development studies with rilpivirine, no effects on fertility were observed at rilpivirine exposures (AUC) greater than 36 times (male) and 40 times (female) the exposure in humans (12 years of age and older and weighing at least 32 kg) at the recommended daily dose of 25 mg.
-
14 CLINICAL STUDIES
14.1 Treatment-Naïve Adult Subjects
The evidence of efficacy of rilpivirine tablets are based on the analyses of 48- and 96-week data from 2 randomized, double-blinded, active controlled, Phase 3 trials TMC278-C209 (ECHO) and TMC278-C215 (THRIVE) in antiretroviral treatment-naïve adults. Antiretroviral treatment-naïve HIV-1 infected subjects enrolled in the Phase 3 trials had a plasma HIV-1 RNA ≥5000 copies/mL and were screened for susceptibility to N(t)RTIs and for absence of specific NNRTI resistance-associated substitutions (RASs). The Phase 3 trials were identical in design, apart from the background regimen (BR). In TMC278-C209, the BR was fixed to the N(t)RTIs, tenofovir disoproxil fumarate plus emtricitabine. In TMC278-C215, the BR consisted of 2 investigator-selected N(t)RTIs: tenofovir disoproxil fumarate plus emtricitabine or zidovudine plus lamivudine or abacavir plus lamivudine. In both trials, randomization was stratified by screening viral load. In TMC278-C215, randomization was also stratified by N(t)RTI BR.
In the pooled analysis for TMC278-C209 and TMC278-C215, demographics and baseline characteristics were balanced between the rilpivirine arm and the efavirenz arm. Table 14 displays selected demographic and baseline disease characteristics of the subjects in the rilpivirine and efavirenz arms.
Table 14: Demographic and Baseline Disease Characteristics of Antiretroviral Treatment-Naïve HIV-1-Infected Adult Subjects in the TMC278-C209 and TMC278-C215 Trials (Pooled Analysis) BR=background regimen
Pooled Data from the Phase 3 TMC278-C209 and TMC278-C215 Trials
Rilpivirine + BR
N=686
Efavirenz + BR
N=682
Demographic Characteristics
Median Age, years (range)
36 (18-78)
36 (19-69)
Sex
Male
76%
76%
Female
24%
24%
Race
White
61%
60%
Black/African American
24%
23%
Asian
11%
14%
Other
2%
2%
Not allowed to ask per local regulations
1%
1%
Baseline Disease Characteristics
Median Baseline Plasma HIV-1 RNA (range), log10 copies/mL
5.0 (2-7)
5.0 (3-7)
Percentage of Patients with Baseline Plasma Viral Load:
≤100,000
54%
48%
>100,000 to ≤500,000
36%
40%
>500,000
10%
12%
Median Baseline CD4+ Cell Count (range), cells/mm3
249 (1-888)
260 (1-1137)
Percentage of Subjects with:
Hepatitis B/C Virus Co-infection
7%
10%
Percentage of Patients with the Following Background
Regimens:
tenofovir disoproxil fumarate plus emtricitabine
80%
80%
zidovudine plus lamivudine
15%
15%
abacavir plus lamivudine
5%
5%
Week 96 efficacy outcomes for subjects treated with rilpivirine tablet 25 mg once daily from the pooled analysis are shown in Table 15. The incidence of virologic failure was higher in the rilpivirine arm than the efavirenz arm at Week 96. Virologic failures and discontinuations due to adverse events mostly occurred in the first 48 weeks of treatment. Regardless of HIV-1 RNA at the start of therapy, more rilpivirine treated subjects with CD4+ cell count less than 200 cells/mm3 experienced virologic failure compared to rilpivirine tablets treated subjects with CD4+ cell count greater than or equal to 200 cells/mm3.
Table 15: Virologic Outcome of Randomized Treatment of Studies TMC278- C209 and TMC278-C215 (Pooled Data) at Week 96 N=total number of subjects per treatment group; BR=background regimen.
* CI=Predicted difference (95% CI) of response rate is -0.2 (-4.7; 4.3) at Week 96.
† Includes subjects who had ≥50 copies/mL in the Week 96 window, subjects who discontinued early due to lack or loss of efficacy, subjects who
discontinued for reasons other than an adverse event, death or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥50 copies/mL, and subjects who had a switch in background regimen that was not permitted by the protocol.
‡ Includes subjects who discontinued due to an adverse event or death if this resulted in no on-treatment virologic data in the Week 96 window.
§ Includes subjects who discontinued for reasons other than an adverse event, death or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up,
etc.
Note: Analysis was based on the last observed viral load data within the Week 96 window (Week 90 -103), respectively.
Rilpivirine + BR
N=686
Efavirenz + BR
N=682
HIV-1 RNA <50 copies/mL*
76%
77%
HIV-1 RNA ≥50 copies/mL†
16%
10%
No virologic data at Week 96 window
Reasons
Discontinued study due to adverse event or death‡
4%
8%
Discontinued study for other reasons and last available HIV-1 RNA <50 copies/mL (or missing)§
4%
5%
Missing data during window but on study
<1%
<1%
HIV-1 RNA <50 copies/mL by Baseline HIV-1 RNA (copies/mL)
≤100,000
82%
78%
>100,000
70%
75%
HIV-1 RNA ≥50 copies/mL† by Baseline HIV-1 RNA (copies/mL)
≤100,000
9%
8%
>100,000
24%
11%
HIV-1 RNA <50 copies/mL by CD4+ cell count (cells/mm3)
<200
68%
74%
≥200
81%
77%
HIV-1 RNA ≥50 copies/mL† by CD4+ cell count (cells/mm3)
<200
27%
10%
≥200
10%
9%
At Week 96, the mean CD4+ cell count increase from baseline was 228 cells/mm3 for rilpivirine treated subjects and 219 cells/mm3 for efavirenz-treated subjects in the pooled analysis of the TMC278-C209 and TMC278-C215 trials.
Study TMC278-C204 was a randomized, active-controlled, Phase 2b trial in antiretroviral treatment-naïve HIV-1-infected adult subjects consisting of 2 parts: an initial 96 weeks, partially-blinded dose-finding part [rilpivirine tablet doses blinded] followed by a long-term, open-label part. After Week 96, subjects randomized to one of the 3 doses of rilpivirine tablets were switched to rilpivirine tablet 25 mg once daily. Subjects in the control arm received efavirenz 600 mg once daily in addition to a BR in both parts of the study. The BR consisted of 2 investigator-selected N(t)RTIs: zidovudine plus lamivudine or tenofovir disoproxil fumarate plus emtricitabine
Study TMC278-C204 enrolled 368 HIV-1-infected treatment-naïve adult subjects who had a plasma HIV-1 RNA ≥5000 copies/mL, previously received ≤2 weeks of treatment with an N(t)RTI or protease inhibitor, had no prior use of NNRTIs, and were screened for susceptibility to N(t)RTI and for absence of specific NNRTI RASs.
At 96 weeks, the proportion of subjects with <50 HIV-1 RNA copies/mL receiving rilpivirine tablets 25 mg (N=93) compared to subjects receiving efavirenz (N=89) was 76% and 71%, respectively. The mean increase from baseline in CD4+ counts was 146 cells/mm3 in subjects receiving rilpivirine tablets 25 mg and 160 cells/mm3 in subjects receiving efavirenz
At 240 weeks, 60% (56/93) of subjects who originally received 25 mg once daily achieved HIV RNA <50 copies/mL compared to 57% (51/89) of subjects in the control group.
14.2 Virologically-Suppressed Adults Treated in Combination with Cabotegravir
The use of rilpivirine tablets in combination with VOCABRIA (cabotegravir) as an oral lead-in and in patients who miss planned injections with CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) was evaluated in two Phase 3 randomized, multicenter, active-controlled, parallel-arm, open-label, non-inferiority trials (Trial 201584: FLAIR [NCT02938520], Trial 201585: ATLAS [NCT2951052]), and one Phase 3b randomized, multicenter, parallel-group, open-label, non-inferiority trial (Trial 207966: ATLAS-2M [NCT03299049]) in subjects who were virologically suppressed (HIV-1 RNA <50 copies/mL). See full prescribing information for VOCABRIA and CABENUVA for additional information.
14.3 Treatment-Naïve Pediatric Subjects (≥12 to less than 18 Years of Age)
The pharmacokinetics, safety, tolerability and efficacy of rilpivirine tablet 25 mg once daily, in combination with an investigator-selected background regimen (BR) containing two NRTIs, was evaluated in trial TMC278-C213 Cohort 1, a single-arm, open-label Phase 2 trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects 12 to less than 18 years of age and weighing at least 32 kg. Thirty six (36) subjects were enrolled in the trial to complete at least 48 weeks of treatment. The 36 subjects had a median age of 14.5 years (range: 12 to 17 years), and were 56% female, 89% Black and 11% Asian.
In the efficacy analysis, most subjects (75%; 28/36) had baseline HIV RNA <100,000 copies/mL. For these 28 subjects the median baseline plasma HIV-1 RNA was 44,250 (range: 2,060-92,600 copies/mL) and the median baseline CD4+ cell count was 445.5 cells/mm3 (range: 123 to 983 cells/mm3)
Among the subjects who had baseline HIV RNA ≤100,000, the proportion with HIV-1 RNA <50 copies/mL at Week 48 was 79% (22/28), versus 50% (4/8) in those with >100,000 copies/mL. The proportion of virologic failures among subjects with a baseline viral load ≤100,000 copies/mL was 21% (6/28), versus 38% (3/8) in those with >100,000 copies/mL. At Week 48, the mean increase in CD4+ cell count from baseline was 201.2 cells/mm3.
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Rilpivirine 25 mg tablets are supplied as white to off-white, film-coated, round, biconvex, 6.5 mm tablets. Each tablet contains 27.5 mg of rilpivirine hydrochloride, which is equivalent to 25 mg of rilpivirine. Each tablet is debossed with "RP" on one side and "25" on the other side.
Rilpivirine 25 mg tablets are packaged in bottles in the following configuration:
25 mg tablets-bottles of 30 (NDC: 70069-848-30)
Store rilpivirine tablets in the original bottle in order to protect from light. Store rilpivirine tablets at 20° to 25°C (68° to 77°F); with excursions permitted to 15° to 30°C (59° to 86°F) [see USP controlled room temperature].
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Severe Skin and Hypersensitivity Reactions
Advise patients to immediately contact their healthcare provider if they develop a rash. Instruct patients to immediately stop taking rilpivirine tablets and seek medical attention if they develop a rash associated with any of the following symptoms as it may be a sign of more serious reactions such as DRESS severe hypersensitivity: fever, blisters, mucosal involvement, eye inflammation (conjunctivitis), severe allergic reaction causing a swelling of the face, eyes, lips, mouth, tongue or throat, which may lead to difficulty swallowing or breathing, and any signs and symptoms of liver problems as it may be a sign of a more serious reaction. Advise patients that if severe rash occurs, they will be closely monitored, laboratory tests will be performed and appropriate therapy will be initiated [see Warnings and Precautions (5.1)].
Hepatotoxicity
Inform patients that hepatotoxicity has been reported with rilpivirine tablets. Inform patients that laboratory monitoring for hepatotoxicity during therapy with rilpivirine tablets are recommended, especially for patients with underlying liver disease such as hepatitis B or C virus infection [see Warnings and Precautions (5.2)].
Depressive Disorders
Inform patients that depressive disorders (depressed mood, depression, dysphoria, major depression, mood altered, negative thoughts, suicide attempt, suicidal ideation) have been reported with rilpivirine tablets. Advise patients to seek immediate medical evaluation if they experience depressive symptoms [see Warnings and Precautions (5.3)].
Drug Interactions
Rilpivirine tablets may interact with many drugs; therefore, advise patients to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products, including St. John's wort [see Contraindications (4), Warnings and Precautions (5.4), and Drug Interactions (7)].
For patients concomitantly receiving rifabutin, the rilpivirine tablets dose should be increased to 50 mg once daily, taken with a meal. When rifabutin coadministration is stopped, the rilpivirine tablets dose should be decreased to 25 mg once daily, taken with a meal [see Dosage and Administration (2.7)].
Immune Reconstitution Syndrome
Advise patients to inform their healthcare provider immediately of any signs or symptoms of infection as inflammation from previous infection may occur soon after combination antiretroviral therapy, including when rilpivirine tablets are started [see Warnings and Precautions (5.5)].
Rilpivirine Tablets And EDURANT PED Tablets for Oral Suspension Are Not Substitutable
Advise patients that rilpivirine tablets and EDURANT PED have differing pharmacokinetic profiles and are not substitutable on a milligram-per-milligram basis. Advise patients or their care providers that patients switching from EDURANT PED tablets for oral suspension to rilpivirine tablets must adjust the dose [see Dosage and Administration (2.1) and Warnings and Precautions (5.6)]
To avoid a dosing error from using the wrong formulation of rilpivirine tablets, strongly advise patients and caregivers to visually inspect the tablets to verify the correct formulation each time the prescription is filled [see Dosage and Administration (2), Warnings and Precautions (5.6), and How Supplied/Storage and Handling (16)].
Administration Instructions
Advise patients to take rilpivirine tablet with a meal once a day as prescribed. A protein drink or yogurt alone does not replace a meal [see Clinical Pharmacology (12.3)]. Rilpivirine tablets must always be used in combination with other antiretroviral drugs. Advise patients not to alter the dose of rilpivirine tablets or discontinue therapy without consulting their physician.
If the patient misses a dose of rilpivirine tablets within 12 hours of the time it is usually taken, advise the patient to take rilpivirine tablet with a meal as soon as possible and then take the next dose of rilpivirine tablet at the regularly scheduled time. If a patient misses a dose of rilpivirine tablet by more than 12 hours, advise the patient to not take the missed dose, but resume the usual dosing schedule. Inform the patient that he or she should not take more or less than the prescribed dose of rilpivirine tablet at any one time.
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to rilpivirine tablets during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Inform individuals with HIV-1 infection that the potential risks of breastfeeding include: (1) HIV-1 transmission (in HIV-1-negative infants), (2) developing viral resistance (in HIV-1-positive infants), and (3) adverse reactions in a breastfed infant similar to those seen in adults [see Use in Specific Populations (8.2)].
Manufactured for:
Somerset Therapeutics, LLC.
Somerset, NJ 08873
Made in India
EDURANT® is a registered trademark of Johnson & Johnson and is used herein for reference purposes only. All other trademarks referenced herein are the property of their respective owners.
-
SPL MEDGUIDE SECTION
Rilpivirine (ril" pi vir' een)
tablets, for oral use
What are rilpivirine tablets?
- Rilpivirine tablets are prescription medicine that are used with other human immunodeficiency virus-1 (HIV-1) medicines to treat HIV-1 infection in people 12 years of age and older and who weigh at least 77 pounds (lbs) or 35 kilograms (kg) who:
- have nevertaken HIV-1 medicines before, and
- have an amount of HIV-1 in their blood (this is called 'viral load') that is no more than 100,000 copies/mL.
- Rilpivirine tablets are also used with oral VOCABRIA (cabotegravir) for short term treatment of HIV-1 infection in people 12 years of age and older and who weigh at least 77 lbs (35 kg) when their healthcare provider determines that they meet certain requirements.
HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome).
If you take rilpivirine tablets in combination with oral VOCABRIA (cabotegravir), you should also read the Patient Information that comes with oral VOCABRIA (cabotegravir).
It is not known if rilpivirine tablets are safe and effective in children less than 2 years of age or who weigh less than 31 lbs (14 kg).
Do not take rilpivirine tablets if you are taking any of the following medicines:
- carbamazepine
- oxcarbazepine
- phenobarbital
- phenytoin
- rifampin
- rifapentine
- dexamethasone (more than a single dose treatment)
- St. John's wort (Hypericum perforatum )
- esomeprazole
- lansoprazole
- ·omeprazole
- pantoprazole
- rabeprazole
Before taking rilpivirine tablets, tell your healthcare provider about all your medical conditions, including if you:
- have ever had a severe skin rash or an allergic reaction to medicines that contain rilpivirine
- have or had liver problems, including hepatitis B or C virus infection.
- have kidney problems
- have ever had a mental health problem.
- are pregnant or plan to become pregnant. It is not known if rilpivirine tablets will harm your unborn baby. Tell your healthcare provider if you become pregnant during treatment with rilpivirine tablets.
- Pregnancy Registry: There is a pregnancy registry for women who take rilpivirine tablets during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk with your healthcare provider about how you can take part in this registry.
- are breastfeeding or plan to breastfeed. Rilpivirine can pass into your breast milk. Talk with your healthcare provider about the following risks of breastfeeding during treatment with rilpivirine:
- The HIV-1 virus may pass to your baby if your baby does not have the HIV-1 infection.
- The HIV-1 virus may become harder to treat if your baby has HIV-1 infection.
- Your baby may get side effects from rilpivirine.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines interact with rilpivirine tablets. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine.
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with rilpivirine tablets.
- Do not start taking a new medicine without telling your healthcare provider.Your healthcare provider can tell you if it is safe to take rilpivirine tablets with other medicines.
How should I take rilpivirine tablets?
- Take rilpivirine tablets exactly as your healthcare provider tells you to.
- Take rilpivirine tablet 1 time each day with a meal. A protein drink or yogurt alone does not replace a meal.
- Rilpivirine tablets must be used with other HIV-1 medicines.
- Do not change your dose or stop taking rilpivirine tablets without first talking with your healthcare provider.
- Stay under the care of your healthcare provider during treatment with rilpivirine tablets.
- EDURANT PED tablets for oral suspension provided in a blister package are not the same as rilpivirine tablets provided in a bottle and cannot be substituted for each other. Contact your pharmacist or healthcare provider if you did not receive the correct dosage form.
- If you take an H2-receptor antagonist (such as famotidine, cimetidine, nizatidine, or ranitidine), you should take these medicines at least 12 hours before or at least 4 hours after you take rilpivirine tablets.
- If you take antacids, or other products that contain aluminum, calcium carbonate, or magnesium hydroxide, you should take these medicines at least 2 hours before or at least 4 hours after you take rilpivirine tablets.
- Do not miss a dose of rilpivirine tablets.
- If you miss a dose of rilpivirine tablet within 12 hours of the time you usually take it, take your dose of rilpivirine tablet with a meal as soon as possible. Then, take your next dose of rilpivirine tablet at the regularly scheduled time. If you miss a dose of rilpivirine tablet by more than 12 hours of the time you usually take it, wait and then take the next dose of rilpivirine tablet at the regularly scheduled time.
- Do not take more than your prescribed dose to make up for a missed dose or take less than your prescribed dose.
- If you take too much rilpivirine tablets, call your healthcare provider or go to the nearest hospital emergency room right away.
- When your supply of rilpivirine tablets starts to run low, get more from your healthcare provider or pharmacy. It is important not to run out of rilpivirine tablets. The amount of HIV in your blood may increase if the medicine is stopped even for a short time.
- When your healthcare provider prescribes use of rilpivirine tablets with oral VOCABRIA (cabotegravir):
- Take rilpivirine tablet and oral VOCABRIA (cabotegravir) 1 time a day at about the same time each day with a meal.
- You will receive treatment with rilpivirine tablets in combination with VOCABRIA tablets for one month (at least 28 days) before you receive the long-acting medicine called CABENUVA (cabotegravir; rilpivirine extended-release injectable suspensions) for the first time. This will allow your healthcare provider to assess how well you tolerate these medicines.
- Your final dose of rilpivirine tablets and VOCABRIA tablets should be taken on the same day you receive your first CABENUVA injections.
- If you miss or plan to miss a scheduled monthly or every 2 months injection of CABENUVA by more than 7 days, call your healthcare provider right away to discuss your treatment options.
What are the possible side effects of rilpivirine tablets?
Rilpivirine tablets can cause serious side effects including:
- Severe skin rash and allergic reactions.Call your healthcare provider right away if you develop a rash with rilpivirine tablets. In some cases, rash and allergic reaction may need to be treated in a hospital.
Stop taking rilpivirine tablets and get medical help right away if you develop a rash with any of the following signs or symptoms:
o fever
o tiredness
o difficulty breathing or swallowing
o skin blisters
o swelling of the face, lips, mouth, tongue, or throat
o generally ill feeling
o muscle or joint aches
o blisters or mouth sores
o redness or swelling of the eyes (conjunctivitis)
- Liver problems. People with a history of hepatitis B or C virus infection or who have certain liver function test changes may have an increased risk of developing new or worsening changes in certain liver tests during treatment with rilpivirine tablets. Liver problems have also happened in people without a history of problems or other risk factors. Your healthcare provider may need to do tests to check your liver function before and during treatment with rilpivirine tablets. Call your healthcare provider right away if you develop any of the following signs or symptoms of liver problems:
o your skin or the white part of your eyes turns yellow (jaundice)
o light colored stools (bowel movements)
o pain, aching, or tenderness on the right side of the stomach area
o loss of appetite
o dark or "tea colored" urine
o nausea or vomiting
- Depression or mood changes. Call your healthcare provider right away if you have any of the following symptoms:
- feeling sad or hopeless
- feeling anxious or restless
- have thoughts of hurting yourself (suicide) or have tried to hurt yourself
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you start having any new symptoms after starting your HIV-1 medicine.
The most common side effects of rilpivirine tablets include depression, headache, trouble sleeping (insomnia) and rash.
These are not all the possible side effects of rilpivirine tablets.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store rilpivirine tablets?
- Store rilpivirine tablets at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep rilpivirine tablets in the original bottle to protect from light.
Keep rilpivirine tablets and all medicines out of the reach of children.
General information about the safe and effective use of rilpivirine tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use rilpivirine tablets for a condition for which it was not prescribed. Do not give rilpivirine tablets to other people even if they have the same condition you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about rilpivirine tablets that is written for health professionals.
What are the ingredients in rilpivirine tablets?
Active ingredient: rilpivirine.
Inactive ingredients:
Rilpivirine 25 mg tablets: croscarmellose sodium, lactose monohydrate, magnesium stearate, polysorbate 20, povidone K30, and silicified microcrystalline cellulose. The tablet coating contains hypromellose 2910 6 mPa.s, lactose monohydrate, polyethylene glycol 3000, titanium dioxide and triacetin.
Additional pediatric use information is approved for Janssen Products LP's Edurant (Rilpivirine) tablets. However, due to Janssen Products LP's marketing exclusivity rights, this drug product is not labeled with that information.
Manufactured for:
Somerset Therapeutics, LLC.
Somerset, NJ 08873
Made in India
EDURANT® is a registered trademark of Johnson & Johnson and is used herein for reference purposes only. All other trademarks referenced herein are the property of their respective owners.
For more information contact Somerset Therapeutics, LLC at 1-800-417-9175
This Patient Information has been approved by the U.S. Food and Drug Administration
Revised 01/2026
- PATIENT PACKAGE INSERT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RILPIVIRINE
rilpivirine tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70069-848 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RILPIVIRINE HYDROCHLORIDE (UNII: 212WAX8KDD) (RILPIVIRINE - UNII:FI96A8X663) RILPIVIRINE 25 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) POVIDONE K30 (UNII: U725QWY32X) POLYSORBATE 20 (UNII: 7T1F30V5YH) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) CI 77891 (UNII: 15FIX9V2JP) PEG-60 (UNII: SA1B764746) TRIACETIN (UNII: XHX3C3X673) SILICIFIED MICROCRYSTALLINE CELLULOSE (125 .MICRO.M, HIGH-DENSITY) (UNII: GRV5BG8C5N) Product Characteristics Color WHITE (White to off-white) Score no score Shape ROUND (Biconvex) Size 6mm Flavor Imprint Code RP;25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70069-848-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 01/30/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA218798 01/30/2026 Labeler - Somerset Therapeutics, LLC (079947873) Registrant - Somerset Therapeutics, LLC (079947873) Establishment Name Address ID/FEI Business Operations Piramal Pharma Limited 862202793 ANALYSIS(70069-848) , MANUFACTURE(70069-848) , PACK(70069-848)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
