5% Dextrose Injection, USP
Dextrose by
Drug Labeling and Warnings
Dextrose by is a Prescription medication manufactured, distributed, or labeled by Baxter Healthcare Corporation, Baxter Corporation. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DEXTROSE- dextrose monohydrate injection
Baxter Healthcare Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
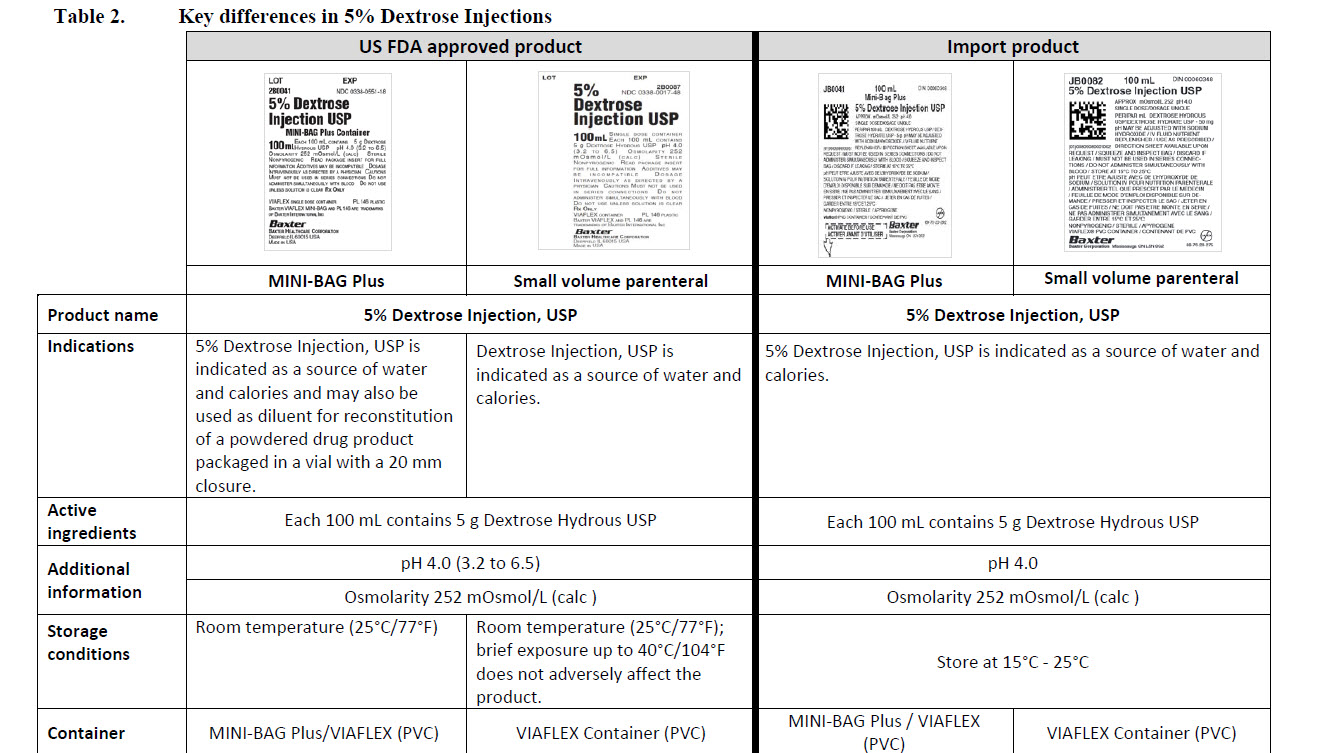
5% Dextrose Injection, USP
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
JB0081
50 mL
DIN 00060348
5% Dextrose Injection USP
Bar Code
(01)00809080000999
APPROX mOsmol/L 252 pH 4.0
SINGLE DOSE / DOSAGE UNIQUE
PER/PAR mL DEXTROSE HYDROUS USP /
DEXTROSE HYDRATE USP – 50 mg pH MAY
BE ADJUSTED WITH SODIUM HYDROXIDE /
IV FLUID NUTRIENT REPLENISHER / USE AS
PRESCRIBED / DIRECTION SHEET AVAILABLE
UPON REQUEST / SQUEEZE AND INSPECT
BAG / DISCARD IF LEAKING / MUST NOT BE
USED IN SERIES CONNECTIONS / DO NOT
ADMINISTER SIMULTANEOUSLY WITH BLOOD /
STORE AT 15ºC TO 25º C
Ph PEUT ETRE ADJUSTE AVEC DE L’HYDROXYDE DE SODIUM / SO-
LUTION IV POUR NUTRITION PARENTERALE / ADMINISTRER TEL
QUE PRESCRIT PAR LE MEDECIN / FEUILLE DE MODE D’EMPLOI
DISPONIBLE SUR DEMANDE / PRESSER ET INSPECTER LE SAC /
JETER EN CAS DE FUITES / NE DOIT PAS ETRE MONTE EN SERIE /
NE PAS ADMINISTRER SIMULTANEMENT AVEC LE SANG /
GARDER ENTRE 15ºC ET 25 ºC
NONPYROGENIC / STERILE / APYROGENE
VIAFLEX® PVC CONTAINER / CONTENANT DE PVC
Baxter Logo
Baxter Corporation Mississauga ON L5N 0C2
No Latex Symbol
88-70-20-274
JB0082
100 mL
DIN 00060348
5% Dextrose Injection USP
Bar Code
(01)00809080001002
APPROX mOsmol/L 252 pH 4.0
SINGLE DOSE/DOSAGE UNIQUE
PER/PAR mL DEXTROSE HYDROUS
USP /DEXTROSE HYDRATE USP – 50 mg
pH MAY BE ADJUSTED WITH SODIUM
HYDROXIDE /IV FLUID NUTRIENT
REPLENISHER / USE AS PRESCRIBED /
DIRECTION SHEET AVAILABLE UPON
REQUEST / SQUEEZE AND INSPECT BAG / DISCARD IF
LEAKING / MUST NOT BE USED IN SERIES CONNEC-
TIONS / DO NOT ADMINISTER SIMULTANEOUSLY WITH
BLOOD / STORE AT 15ºC TO 25º C
pH PEUT ETRE AJUSTE AVEC DE L’HYDROXYDE DE
SODIUM / SOLUTION IV POUR NUTRITION PARENTERALE
/ ADMINISTRER TEL QUE PRESCRIT PAR LE MEDECIN
/ FEUILLE DE MODE D’EMPLOI DISPONIBLE SUR DE-
MANDE / PRESSER ET INSPECTER LE SAC / JETER EN
CAS DE FUITES / NE DOIT PAS ETRE MONTE EN SERIE /
NE PAS ADMINISTRER SIMULTANEMENT AVEC LE SANG /
GARDER ENTRE 15ºC ET 25 ºC
NONPYROGENIC / STERILE / APYROGENE
VIAFLEX® PVC CONTAINER / CONTENANT DE PVC
Baxter Logo
Baxter Corporation Mississauga ON L5N 0C2
No Latex Symbol
88-70-20-275
| DEXTROSE
dextrose monohydrate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEXTROSE
dextrose monohydrate injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Baxter Healthcare Corporation (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Corporation | 205087968 | ANALYSIS(0338-9533, 0338-9530) , MANUFACTURE(0338-9533, 0338-9530) , LABEL(0338-9533, 0338-9530) , PACK(0338-9533, 0338-9530) , STERILIZE(0338-9533, 0338-9530) | |