FlutiCare Nasal Allergy Spray
FlutiCare by
Drug Labeling and Warnings
FlutiCare by is a Otc medication manufactured, distributed, or labeled by Innovus Pharmaceuticals Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
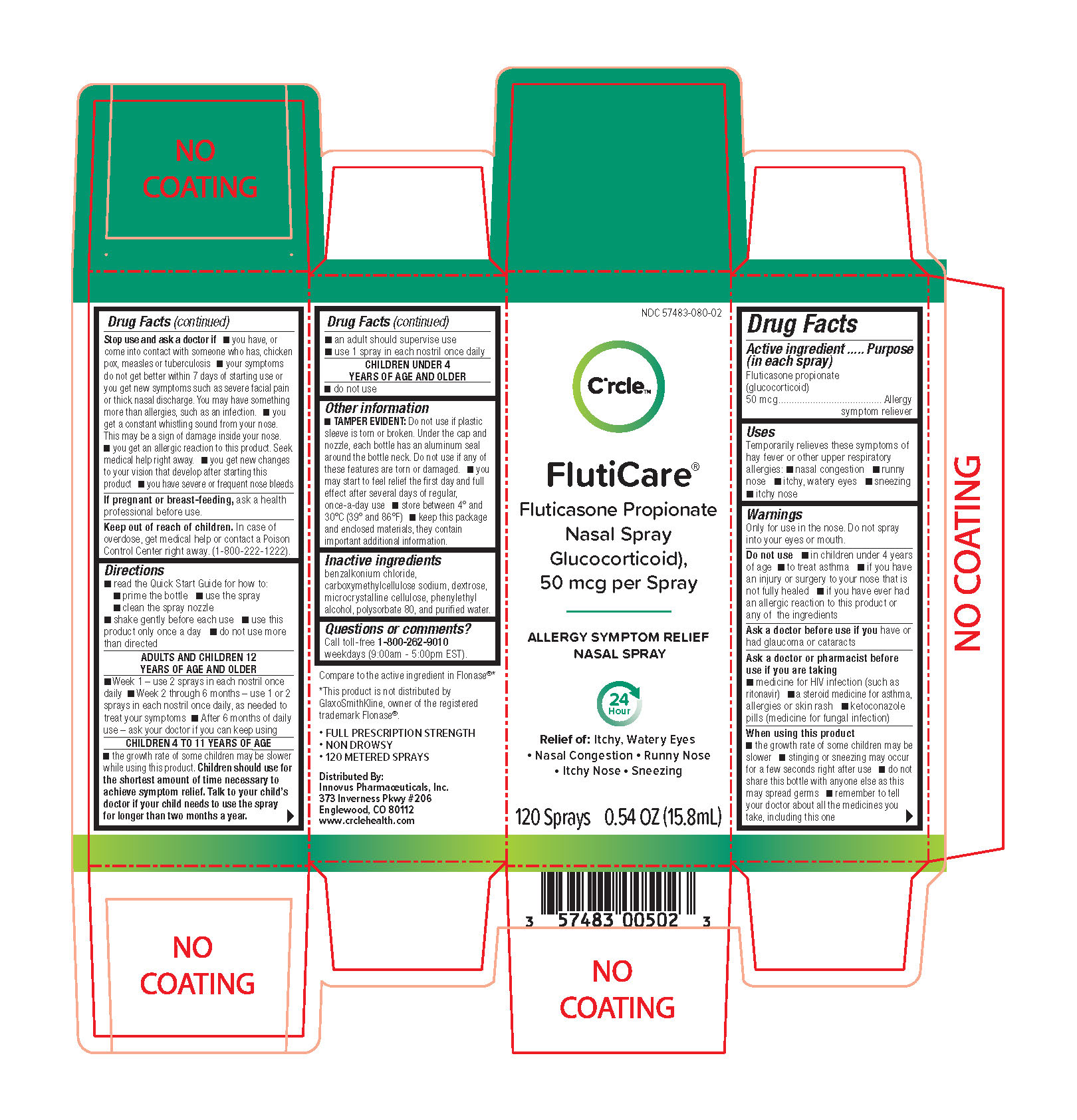
FLUTICARE NASAL ALLERGY- fluticasone propionate (glucocorticoid) spray, metered
Innovus Pharmaceuticals Inc.
----------
FlutiCare Nasal Allergy Spray
Uses
Temporarily relieves these symptoms of hay fever or other upper respiratory allergies:
- nasal congestion
- runny nose
- itchy watery, eyes
- sneezing
- itchy nose
Warnings
Only for use in the nose. Do not spray into your eyes or mouth.
Do not use
- in children under 4 years of age
- to treat asthma
- if you have an injury or surgery to your nose that is not fully healed
- if you have ever had an allergic reaction to this product or any of the ingredients
Ask a doctor before use if you have or had
glaucoma or cataracts
Ask a doctor or pharmacist before use if you are taking
- medicine for HIV infection (such as ritonavir)
- a steroid medicine for asthma, allergies or skin rash
- ketoconazole pills (medicine for fungal infection)
When using this product
- the growth rate of some children may be slower
- stinging or sneezing may occur for a few seconds right after use
- do not share this bottle with anyone else as this may spread germs
- remember to tell your doctor about all the medicines you take, including this one
Stop use and ask a doctor if
- you have, or come into contact with someone who has, chicken pox, measles or tuberculosis
- your symptoms do not get better within 7 days of starting use or you get new symptoms such as severe facial pain or thick nasal discharge. You may have something more than allergies, such as an infection.
- you get a constant whistling sound from your nose. This may be a sign of damage inside your nose.
- you get an allergic reaction to this product. Seek medical help right away.
- you get new changes to your vision that develop after starting this product
- you have severe or frequent nosebleeds
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Directions
read the Quick Start Guide for how to:
- prime the bottle
- use the spray
- clean the spray nozzle
- shake gently before each use
- use this product only once a day
- do not use more than directed
ADULTS AND CHILDREN 12 YEARS OF AGE AND OLDER
- Week 1 – use 2 sprays in each nostril once daily
- Week 2 through 6 months – use 1 or 2 sprays in each nostril once daily, as needed to treat your symptoms
- After 6 months of daily use – ask your doctor if you can keep using
CHILDREN 4 TO 11 YEARS OF AGE
- the growth rate of some children may be slower while using this product. Children should use for the shortest amount of time necessary to achieve symptom relief. Talk to your child’s doctor if your child needs to use the spray for longer than two monthsa year.
- an adult should supervise use
- use 1 spray in each nostril once daily
CHILDREN UNDER 4 YEARS OF AGE
- do not use
Other information
- TAMPER EVIDENT: Do not use if plastic sleeve is torn or broken. Under the cap and nozzle, each bottle has an aluminum seal around the bottle neck. Do not use if any of these featurees are torn or damaged.
- you may start to feel relief the first day and full effect after several days of regular, once-a-day use
- store between 4° and 30°C (39° and 86°F)
- keep this package and enclosed materials. They contain important additional information.
| FLUTICARE
NASAL ALLERGY
fluticasone propionate (glucocorticoid) spray, metered |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Innovus Pharmaceuticals Inc. (962507187) |
| Registrant - Innovus Pharmaceuticals Inc. (962507187) |
Trademark Results [FlutiCare]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FLUTICARE 98371336 not registered Live/Pending |
Yanggen, Steven Douglas 2024-01-23 |
 FLUTICARE 87601188 5471512 Live/Registered |
Innovus Pharmaceuticals, Inc. 2017-09-08 |
 FLUTICARE 87262502 5331309 Live/Registered |
Innovus Pharmaceuticals, Inc. 2016-12-08 |
 FLUTICARE 85971232 not registered Dead/Abandoned |
Novalere FP, Inc. 2013-06-27 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.