EternaTear by Eternatear, Inc. / TUBILUX PHARMA SPA Drug Facts
EternaTear by
Drug Labeling and Warnings
EternaTear by is a Otc medication manufactured, distributed, or labeled by Eternatear, Inc., TUBILUX PHARMA SPA. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ETERNATEAR- mineral oil solution
Eternatear, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
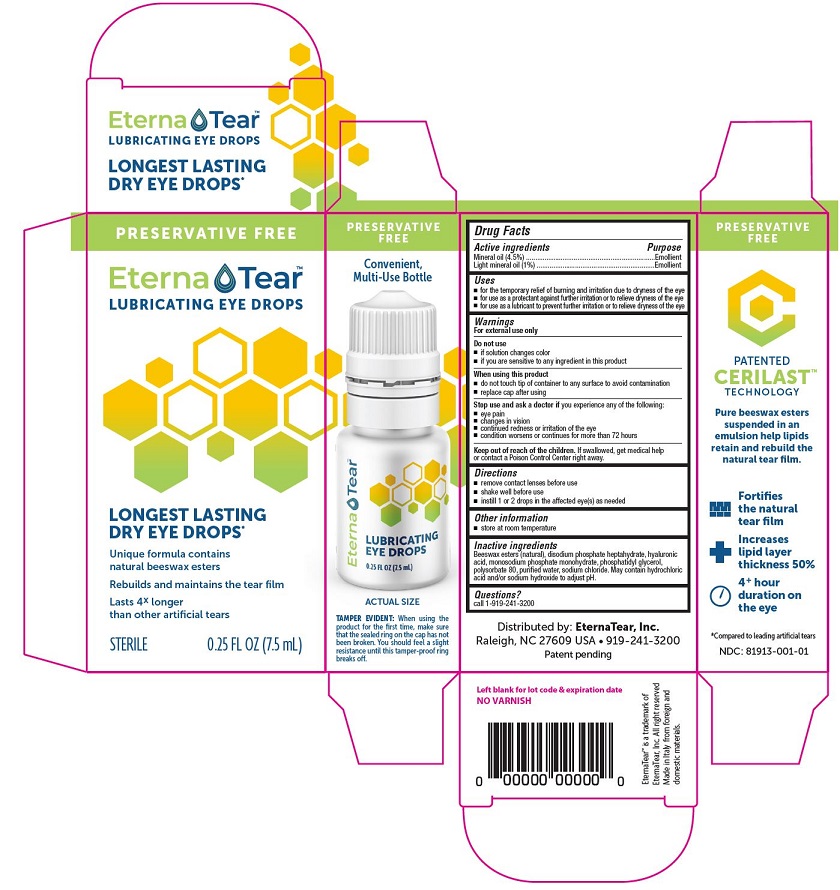
Drug Facts
Uses
- for the temporary relief of burning and irritation due to dryness of the eye
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye
When using this product
- do not touch tip of container to any surface to avoid contamination
- replace cap after using
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or continues for more than 72 hours
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- remove contact lenses before use
- shake well before use
- instill 1 or 2 drops in the affected eye(s) as needed
Inactive Ingredients
Beeswax esters (natural), disodium phosphate heptahydrate, hyaluronic acid, monosodium phosphate monohydrate, phosphatidyl glycerol, polysorbate 80, purified water, sodium chloride. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
PRINCIPAL DISPLAY PANEL
PRESERVATIVE FREE
EternaTear™
LUBRICATING EYE DROPS
LONGEST LASTING
DRY EYE DROPS*
Unique formula contains
natural beeswax esters
Rebuilds and maintains the tear film
Lasts 4x longer
than other artificial tears
STERILE 0.25 FL OZ (7.5 mL)
PRESERVATIVE FREE
Convenient,
Multi-Use Bottle
ACTUAL SIZE
TAMPER EVIDENT: When using the
product for the first time, make sure
that the sealed ring on the cap has not
been broken. You should feel a slight
resistance until this tamper-proof ring
breaks off.
PRESERVATIVE FREE
PATENTED
CERILAST™
TECHNOLOGY
Pure beeswax esters
suspended in an
emulsion help lipids
retain and rebuild the
natural tear film.
Fortifies the natural tear film
Increases lipid layer thickness 50%
4+ hour duration on the eye
*Compared to leading artificial tears
NDC: 81913-001-01
Distributed by: EternaTear, Inc.
Raleigh, NC 27609 USA 919-241-3200
Patent pending
EternaTear™ is a trademark of
EternaTear, Inc. All right reserved
Made in Italy from foreign and domestic materials.
| ETERNATEAR
mineral oil solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Eternatear, Inc. (118029357) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| TUBILUX PHARMA SPA | 338981251 | manufacture(81913-001) | |
Trademark Results [EternaTear]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ETERNATEAR 90197575 not registered Live/Pending |
EternaTear, Inc. 2020-09-21 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.