EYE STREAM- purified water solution
Eye Stream by
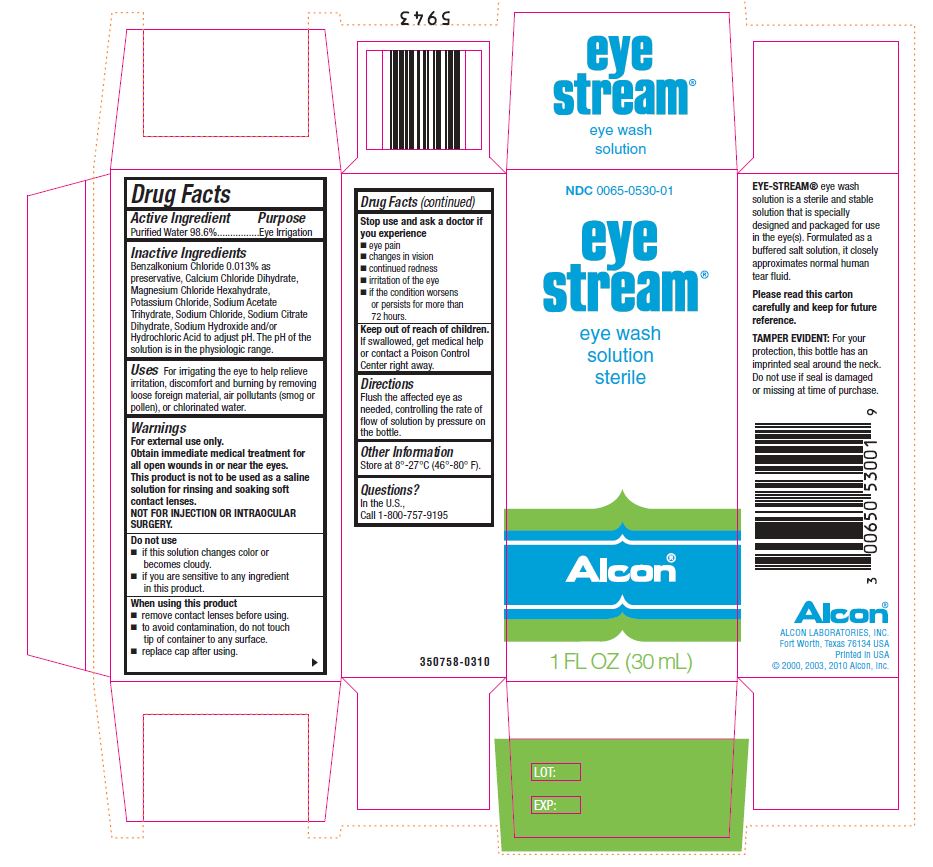
Drug Labeling and Warnings
Eye Stream by is a Otc medication manufactured, distributed, or labeled by Alcon Laboratories, Inc., Alcon Research LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
-
Inactive Ingredients
Benzalkonium Chloride 0.013% as preservative, Calcium Chloride Dihydrate, Magnesium Chloride Hexahydrate, Potassium Chloride, Sodium Acetate Trihydrate, Sodium Chloride, Sodium Citrate Dihydrate, Sodium Hydroxide and/or Hydrochloric Acid to adjust pH. The pH range of solution is in the physiologic range.
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if you experience
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other Information
- Questions?
- ACTIVE INGREDIENT
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if you experience
- Keep out of reach of children
- Directions
- Other Information
-
Inactive Ingredients
Benzalkonium Chloride 0.013% as a preservative, Calcium Chloride Dihydrate, Magnesium Chloride Hexahydrate, Potassium Chloride, Sodium Acetate Trihydrate, Sodium Citrate Dihydrate, Sodium Chloride and Sodium Hydroxide and/or Hydrochloric Acid to adjust pH. The pH of the solution is in the physiological range.
- Questions?
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EYE STREAM
purified water solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0065-0530 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Water (UNII: 059QF0KO0R) (Water - UNII:059QF0KO0R) Water 986 mg in 1 mL Inactive Ingredients Ingredient Name Strength Benzalkonium Chloride (UNII: F5UM2KM3W7) Calcium Chloride (UNII: M4I0D6VV5M) Magnesium Chloride (UNII: 02F3473H9O) Potassium Chloride (UNII: 660YQ98I10) Sodium Acetate (UNII: 4550K0SC9B) Sodium Chloride (UNII: 451W47IQ8X) Trisodium Citrate Dihydrate (UNII: B22547B95K) Sodium Hydroxide (UNII: 55X04QC32I) Hydrochloric Acid (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0065-0530-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/30/1990 2 NDC: 0065-0530-04 118 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/30/1990 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 09/30/1990 Labeler - Alcon Laboratories, Inc. (008018525) Registrant - Alcon Laboratories, Inc. (008018525) Establishment Name Address ID/FEI Business Operations Alcon Research LLC 007672236 manufacture(0065-0530)
Trademark Results [Eye Stream]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 EYE STREAM 75300945 not registered Dead/Abandoned |
QuadState Corporation 1997-05-30 |
 EYE STREAM 75300944 not registered Dead/Abandoned |
QuadState Corporation 1997-05-30 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.