Phase 4 Gel® 509-P4-NSP
GUDID 00696588003627
Phase 4 Gel® Splint - Non Sterile (10/case)
Innovative Medical Products, Inc
Surgical linen/cushion set| Primary Device ID | 00696588003627 |
| NIH Device Record Key | a8d9f2aa-829d-4283-ad92-58f1cbc3ebb0 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | Phase 4 Gel® |
| Version Model Number | 509-P4-NSP |
| Catalog Number | 509-P4-NSP |
| Company DUNS | 147363154 |
| Company Name | Innovative Medical Products, Inc |
| Device Count | 1 |
| DM Exempt | true |
| Pre-market Exempt | true |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | true |
| Expiration Date | false |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | true |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00696588003627 [Primary] |
FDA Product Code
| FMP | Protector, Skin Pressure |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2022-08-10 |
| Device Publish Date | 2022-08-02 |
On-Brand Devices [Phase 4 Gel®]
| 00696588005621 | Sample Phase 4 Gel® Splint - Non Sterile (1 each) |
| 00696588003627 | Phase 4 Gel® Splint - Non Sterile (10/case) |
Trademark Results [Phase 4 Gel]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
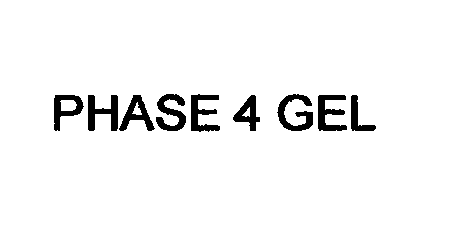 PHASE 4 GEL 76714911 4535142 Live/Registered |
Innovative Medical Products Inc. 2013-09-06 |
 PHASE 4 GEL 76697938 not registered Dead/Abandoned |
Innovative Medical Products Inc. 2009-06-17 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.