Percept™ RC
GUDID 00763000564346
INS B35300 DBS PERCEPT RC US EMANUAL
MEDTRONIC, INC.
Deep brain electrical stimulation system| Primary Device ID | 00763000564346 |
| NIH Device Record Key | f67a393e-d16f-4f1a-a842-b875deb707c4 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | Percept™ RC |
| Version Model Number | B35300 |
| Company DUNS | 796986144 |
| Company Name | MEDTRONIC, INC. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | MR Conditional |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | false |
| Expiration Date | false |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Customer Support Contacts
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00763000564346 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Approval: P960009
FDA Product Code
| NHL | Stimulator, electrical, implanted, for parkinsonian symptoms |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2024-01-29 |
| Device Publish Date | 2024-01-21 |
Devices Manufactured by MEDTRONIC, INC.
| 00763000764234 - NA | 2026-04-01 CUSTOM PACK CB11Z66R 1/4 LOOP |
| 00763000764241 - NA | 2026-04-01 CUSTOM PACK CB11Z68R CDI PACK |
| 00763000764319 - NA | 2026-04-01 CUSTOM PACK HY2G71R9 ST. AGNES PK |
| 00763000764951 - NA | 2026-04-01 CUSTOM PACK BB12A73R AV LOOP |
| 00763000770419 - NA | 2026-04-01 CUSTOM PACK 11Z79R1 VALVE SK LN |
| 00763000770433 - NA | 2026-04-01 CUSTOM PACK BB12B18R HIIT FUS |
| 00763000774127 - NA | 2026-04-01 CUSTOM PACK BB11U49R3 ADULT QUANTUM |
| 00763000791421 - NA | 2026-04-01 CUSTOM PACK BB11Y57R5 HEART LUNG |
Trademark Results [Percept]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
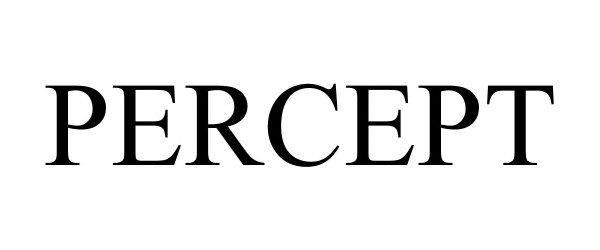 PERCEPT 90658294 not registered Live/Pending |
Microsoft Corporation 2021-04-20 |
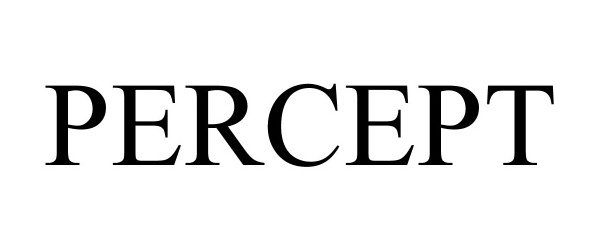 PERCEPT 90647170 not registered Live/Pending |
IONODES Inc. 2021-04-15 |
 PERCEPT 88182595 not registered Live/Pending |
Percept Corporation 2018-11-06 |
 PERCEPT 88182582 not registered Live/Pending |
Percept Corporation 2018-11-06 |
 PERCEPT 88048080 not registered Live/Pending |
Medtronic, Inc. 2018-07-23 |
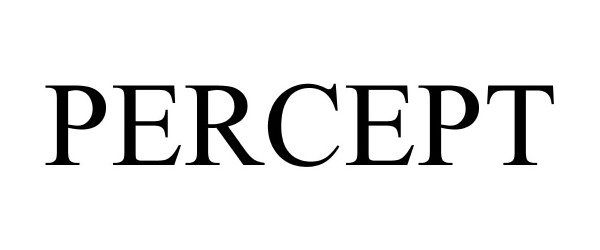 PERCEPT 87000843 not registered Live/Pending |
Tricerat, Inc. 2016-04-14 |
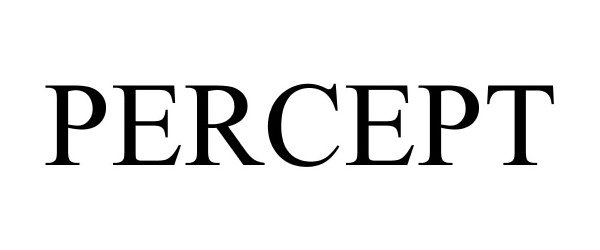 PERCEPT 86491197 4842820 Live/Registered |
Trustees of the University of Massachusetts 2014-12-29 |
 PERCEPT 86342680 4692434 Live/Registered |
Percept Eyewear 2014-07-21 |
 PERCEPT 86289474 4742223 Live/Registered |
Arbill Industries, Inc. 2014-05-22 |
 PERCEPT 85162969 4265029 Live/Registered |
PolyOne Corporation 2010-10-27 |
 PERCEPT 79367768 not registered Live/Pending |
Yonex Kabushiki Kaisha 2023-03-17 |
 PERCEPT 79350689 not registered Live/Pending |
Blickfeld GmbH 2022-04-26 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.