ACCOLADE™ MRI SR
GUDID 00802526559204
Pacemaker
BOSTON SCIENTIFIC CORPORATION
Single-chamber implantable pacemaker, rate-responsive| Primary Device ID | 00802526559204 |
| NIH Device Record Key | d91a980a-3099-4da8-883f-c0e09c1fac85 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | ACCOLADE™ MRI SR |
| Version Model Number | L310 |
| Company DUNS | 106295384 |
| Company Name | BOSTON SCIENTIFIC CORPORATION |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | MR Conditional |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00802526559204 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Approval: P150012
FDA Product Code
| LWP | Implantable pulse generator, pacemaker (non-CRT) |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 5 |
| Public Version Date | 2019-11-08 |
| Device Publish Date | 2016-05-10 |
Devices Manufactured by BOSTON SCIENTIFIC CORPORATION
| 08714729195573 - RotaWire™ and wireClip™ Torquer | 2026-03-31 Guidewire and Guidewire Manipulation Device |
| 08714729195566 - RotaWire™ and wireClip™ Torquer | 2026-03-31 Guidewire and Guidewire Manipulation Device |
| 08714729837527 - Stingray™ | 2026-03-31 Guidewire with Hydrophilic Coating |
| 08714729877523 - EP XT™ | 2026-03-20 Unidirectional Steerable Diagnostic Catheter |
| 08714729877714 - Dynamic XT™ | 2026-03-20 Unidirectional Steerable Diagnostic Catheter |
| 08714729879947 - EP XT™ | 2026-03-20 Unidirectional Steerable Diagnostic Catheter |
| 08714729181088 - Percuflex™ Biliary | 2026-03-19 Biliary Stent with Introducer Kit |
| 08714729181095 - Percuflex™ Biliary | 2026-03-19 Biliary Stent with Introducer Kit |
Trademark Results [ACCOLADE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ACCOLADE 98747980 not registered Live/Pending |
ALO, LLC 2024-09-12 |
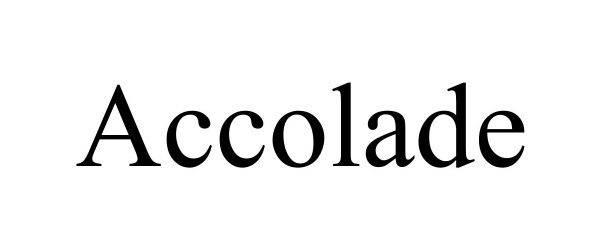 ACCOLADE 98676975 not registered Live/Pending |
Bradshaw, Lauren A. 2024-07-31 |
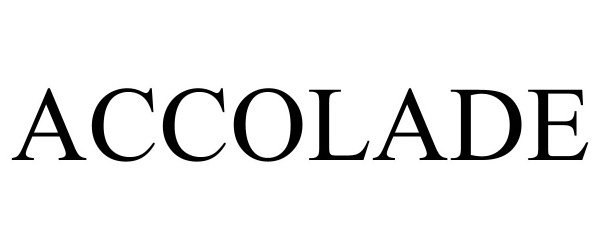 ACCOLADE 98475209 not registered Live/Pending |
ACH Food Companies, Inc. 2024-03-29 |
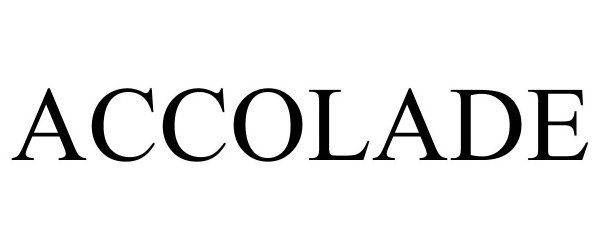 ACCOLADE 90715037 not registered Live/Pending |
CHS Inc. 2021-05-17 |
 ACCOLADE 90697215 not registered Live/Pending |
Allegheny Casualty Company 2021-05-07 |
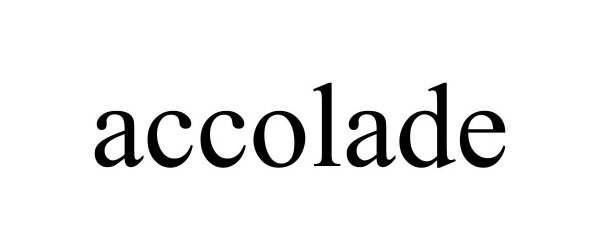 ACCOLADE 90697145 not registered Live/Pending |
Allegheny Casualty Company 2021-05-07 |
 ACCOLADE 88338615 5875981 Live/Registered |
Jayco, Inc. 2019-03-13 |
 ACCOLADE 88128665 not registered Dead/Abandoned |
AIREWINE, LLC 2018-09-24 |
 ACCOLADE 88117662 not registered Live/Pending |
Tacony Corporation 2018-09-14 |
 ACCOLADE 87684686 5613403 Live/Registered |
Meyer Manufacturing Company Limited 2017-11-14 |
 ACCOLADE 87014510 not registered Dead/Abandoned |
Triple E Canada Ltd. 2016-04-26 |
 ACCOLADE 86656171 4885589 Live/Registered |
BILLION SOFT (HONG KONG) LIMITED 2015-06-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.