EMBLEM™ S-ICD
GUDID 00802526575600
Subcutaneous Electrode
BOSTON SCIENTIFIC CORPORATION
Endocardial defibrillation lead| Primary Device ID | 00802526575600 |
| NIH Device Record Key | a78e1923-9bd0-4986-90d7-4fcee7a6c0b6 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | EMBLEM™ S-ICD |
| Version Model Number | 3401 |
| Company DUNS | 106295384 |
| Company Name | BOSTON SCIENTIFIC CORPORATION |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | MR Conditional |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Device Dimensions
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00802526575600 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Approval: P110042
FDA Product Code
| LWS | Implantable cardioverter defibrillator (non-CRT) |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 3 |
| Public Version Date | 2018-07-06 |
| Device Publish Date | 2015-06-12 |
On-Brand Devices [EMBLEM™ S-ICD]
| 00802526599200 | Subcutaneous Electrode |
| 00802526585968 | Subcutaneous Electrode |
| 00802526575600 | Subcutaneous Electrode |
| 00802526544101 | Subcutaneous Implantable Cardioverter Defibrillator |
Trademark Results [EMBLEM]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
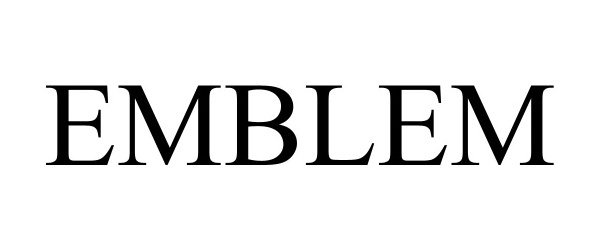 EMBLEM 98010040 not registered Live/Pending |
Topgolf Callaway Brands Corp. 2023-05-23 |
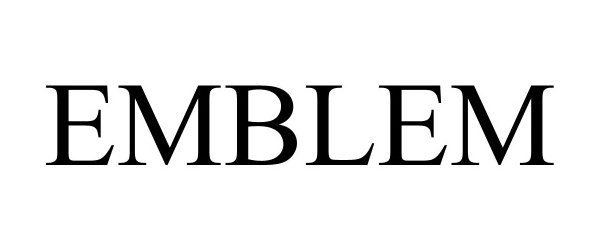 EMBLEM 97775916 not registered Live/Pending |
Towers Watson Software Limited 2023-02-01 |
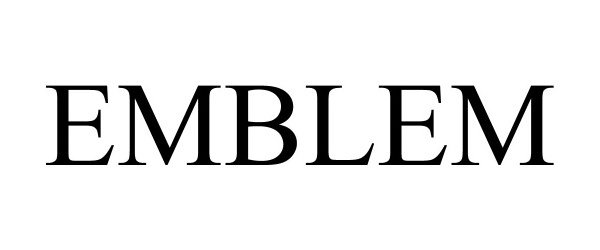 EMBLEM 88875507 not registered Live/Pending |
Chasity Pritchett, LLC 2020-04-16 |
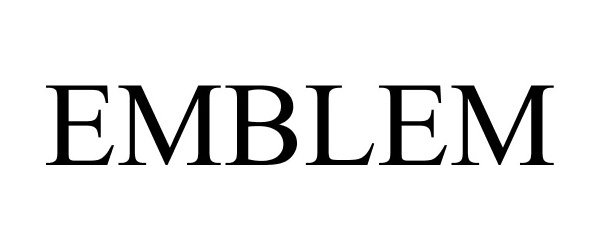 EMBLEM 88090120 not registered Live/Pending |
Emblem LLC 2018-08-23 |
 EMBLEM 87833881 not registered Dead/Abandoned |
Ava Food Labs, Inc. 2018-03-14 |
 EMBLEM 87600878 5571919 Live/Registered |
Jayco, Inc. 2017-09-08 |
 EMBLEM 87498141 5515212 Live/Registered |
Britax Child Safety, Inc. 2017-06-20 |
 EMBLEM 86549852 not registered Dead/Abandoned |
Jayco, Inc. 2015-03-02 |
 EMBLEM 86461556 5064837 Live/Registered |
Enseo, Inc. 2014-11-21 |
 EMBLEM 86354094 4740953 Live/Registered |
ZTE (USA), INC. 2014-07-31 |
 EMBLEM 86190094 4732801 Live/Registered |
SWIMC LLC 2014-02-11 |
 EMBLEM 86078512 4827622 Live/Registered |
Cardiac Pacemakers, Inc. 2013-09-30 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.