Bard-Parker
GUDID 00840113231867
Scalpel Holder Red; Aspen Leaf with Bard-Parker; Serile (100/box)
ASPEN SURGICAL PRODUCTS, INC.
Surgical instrument interim-placement holder, single-use| Primary Device ID | 00840113231867 |
| NIH Device Record Key | d600cd5e-5c18-434a-be0b-5f0dc4869107 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | Bard-Parker |
| Version Model Number | ST-100 |
| Company DUNS | 027680821 |
| Company Name | ASPEN SURGICAL PRODUCTS, INC. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | true |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | true |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Customer Support Contacts
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com | |
| Phone | 8883647004 |
| customerservice@aspensurgical.com |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00840113231867 [Primary] |
FDA Product Code
| FSM | Tray, Surgical, Instrument |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2026-02-23 |
| Device Publish Date | 2026-02-15 |
Devices Manufactured by ASPEN SURGICAL PRODUCTS, INC.
| 00814707023817 - Protek | 2026-02-24 Protek; Tablet Cover; 8" x 10" (20cm x 25;4cm); PE; w/adhesive seal; flat-fold; sterile; 1/pouch; 24/box; (1028) |
| 00840113231867 - Bard-Parker | 2026-02-23Scalpel Holder Red; Aspen Leaf with Bard-Parker; Serile (100/box) |
| 00840113231867 - Bard-Parker | 2026-02-23 Scalpel Holder Red; Aspen Leaf with Bard-Parker; Serile (100/box) |
| 00887482023389 - Symmetry | 2026-02-23 Symmetry Sharp Kerrison Rongeur Tips; 2 mm Tip; Single Use; (3/bx) |
| 00887482023396 - Symmetry | 2026-02-23 Symmetry Sharp Kerrison Rongeur Tips; 3 mm Tip; Single Use; (3/bx) |
| 00887482023402 - Symmetry | 2026-02-23 Symmetry Sharp Kerrison Rongeur Tips; 4 mm Tip; Single Use; (3/bx) |
| 00887482138373 - Sterion | 2026-02-23 Ultra Container; Mini Loops; Red; Single Use; (2/pk; 10/bx) |
| 00887482156186 - Symmetry | 2026-02-23 Symmetry Sharp Kerrison Rongeur Tips; Thin Footplate; 2 mm Tip; Single Use; (3/bx) |
| 00887482156223 - Symmetry | 2026-02-23 Symmetry Sharp Kerrison Rongeur Tips; Thin Footplate; 4 mm Tip; Single Use; (3/bx) |
Trademark Results [Bard-Parker]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
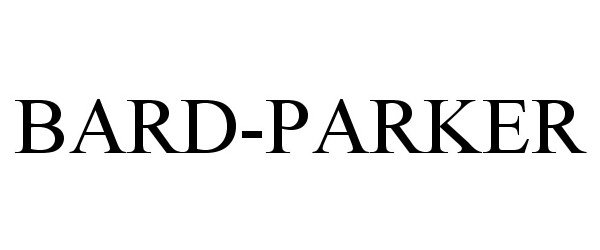 BARD-PARKER 86871555 5219374 Live/Registered |
Aspen Surgical Products, Inc. 2016-01-11 |
 BARD-PARKER 78267573 2884264 Live/Registered |
ASPEN SURGICAL PRODUCTS, INC. 2003-06-26 |
 BARD-PARKER 73237986 1213159 Dead/Cancelled |
BECTON, DICKINSON AND COMPANY 1979-11-05 |
 BARD-PARKER 72078542 0693945 Dead/Cancelled |
BARD-PARKER COMPANY, INC. 1959-07-28 |
 BARD-PARKER 72078541 0703750 Dead/Cancelled |
BARD-PARKER COMPANY, INC. 1959-07-28 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.