NEXDRIVE™ MI-2000
GUDID 00873263007723
MI-2000 NEXDRIVE™ Micropositioning Drive
FHC, INC.
Neurological stereotactic surgery system| Primary Device ID | 00873263007723 |
| NIH Device Record Key | f990fe02-b932-442d-8de8-0a3f164e339c |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | NEXDRIVE™ |
| Version Model Number | MI-2000-00772 |
| Catalog Number | MI-2000 |
| Company DUNS | 108179458 |
| Company Name | FHC, INC. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | MR Unsafe |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Customer Support Contacts
| Phone | +1(800)326-2905 |
| fhcinc@fh-co.com | |
| Phone | +1(800)326-2905 |
| fhcinc@fh-co.com | |
| Phone | +1(800)326-2905 |
| fhcinc@fh-co.com | |
| Phone | +1(800)326-2905 |
| fhcinc@fh-co.com |
Operating and Storage Conditions
| Storage Environment Temperature | Between -34 Degrees Celsius and 57 Degrees Celsius |
| Storage Environment Temperature | Between -34 Degrees Celsius and 57 Degrees Celsius |
| Storage Environment Temperature | Between -34 Degrees Celsius and 57 Degrees Celsius |
| Storage Environment Temperature | Between -34 Degrees Celsius and 57 Degrees Celsius |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00873263007723 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Notification: K022100
FDA Product Code
| HAW | Neurological stereotaxic Instrument |
Sterilization
| Steralize Prior To Use | true |
| Device Is Sterile | true |
[00873263007723]
Ethylene Oxide
[00873263007723]
Ethylene Oxide
[00873263007723]
Ethylene Oxide
[00873263007723]
Ethylene Oxide
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2020-11-10 |
| Device Publish Date | 2020-11-02 |
On-Brand Devices [NEXDRIVE™]
| 00873263007723 | MI-2000 NEXDRIVE™ Micropositioning Drive |
| 00873263008713 | MI-2000 NEXDRIVE™ Micropositioning Drive |
Trademark Results [NEXDRIVE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NEXDRIVE 98147972 not registered Live/Pending |
SHENZHEN TECHWINSEMI Technology Co., LTD 2023-08-24 |
 NEXDRIVE 97038013 not registered Live/Pending |
Genuine Parts Company 2021-09-21 |
 NEXDRIVE 79353971 not registered Live/Pending |
Alliance Automotive Group Benelux B.V. 2022-05-27 |
 NEXDRIVE 79353955 not registered Live/Pending |
Alliance Automotive Group Benelux B.V. 2022-05-27 |
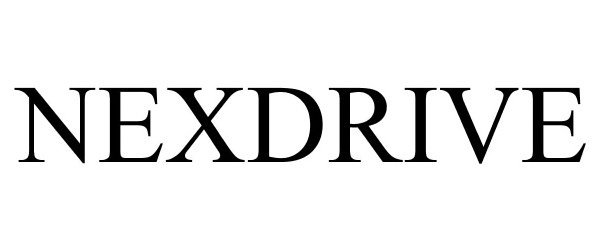 NEXDRIVE 78943173 3812254 Dead/Cancelled |
1384489 Ontario Inc. 2006-08-02 |
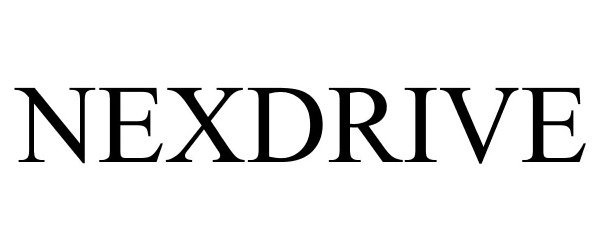 NEXDRIVE 78753134 3235288 Live/Registered |
Medtronic, Inc. 2005-11-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.