DISSECTOR
GUDID 00888118119636
KLS-Martin L.P.
Paranasal sinus curette| Primary Device ID | 00888118119636 |
| NIH Device Record Key | b508c871-b578-47df-ae0c-221f4b584207 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | DISSECTOR |
| Version Model Number | 99-586-15-12 |
| Company DUNS | 826499238 |
| Company Name | KLS-Martin L.P. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | true |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | false |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Customer Support Contacts
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com | |
| Phone | +1(904)641-7746 |
| KLS-UDI-Info@klsmartin.com |
Device Dimensions
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00888118119636 [Primary] |
FDA Product Code
| GZX | INSTRUMENT, MICROSURGICAL |
Sterilization
| Steralize Prior To Use | true |
| Device Is Sterile | false |
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
[00888118119636]
Moist Heat or Steam Sterilization
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 4 |
| Public Version Date | 2019-12-16 |
| Device Publish Date | 2018-09-26 |
On-Brand Devices [DISSECTOR]
Trademark Results [DISSECTOR]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 DISSECTOR 90477529 not registered Live/Pending |
Beijing Gridsum Technology Co., Ltd. 2021-01-20 |
 DISSECTOR 88318143 not registered Live/Pending |
CHENG SHIN RUBBER IND. CO., LTD 2019-02-27 |
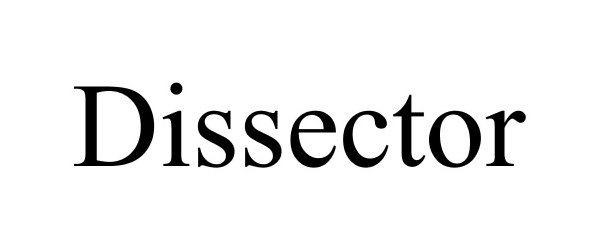 DISSECTOR 85641900 not registered Dead/Abandoned |
BEIJING GRIDSUM TECHNOLOGY CO., LTD. 2012-06-04 |
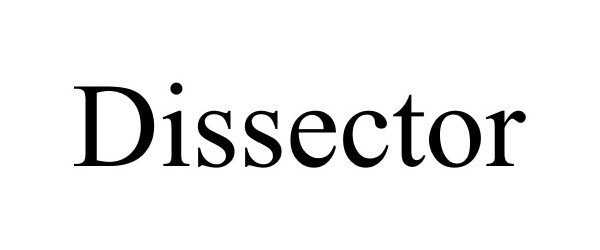 DISSECTOR 85641897 4401247 Live/Registered |
BEIJING GRIDSUM TECHNOLOGY CO., LTD. 2012-06-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.