s-max
GUDID 04054959000280
AAT Alber Antriebstechnik GmbH
Stairclimber| Primary Device ID | 04054959000280 |
| NIH Device Record Key | 5a164f73-8784-48ba-9396-5ee93de7af4a |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | s-max |
| Version Model Number | D1611 |
| Company DUNS | 314612359 |
| Company Name | AAT Alber Antriebstechnik GmbH |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | false |
| Expiration Date | false |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | true |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 04054959000280 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Notification: K130864
FDA Product Code
| ILK | Transport, Patient, Powered |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2025-08-27 |
| Device Publish Date | 2025-08-19 |
Trademark Results [s-max]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
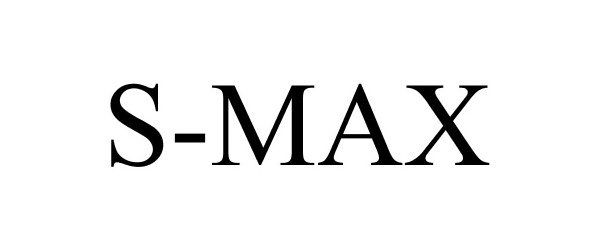 S-MAX 86528677 4938071 Live/Registered |
The ExOne Company 2015-02-09 |
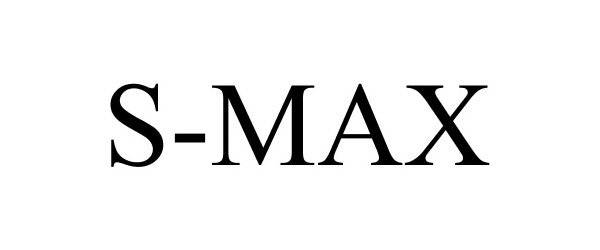 S-MAX 86005120 not registered Dead/Abandoned |
Ford Motor Company 2013-07-09 |
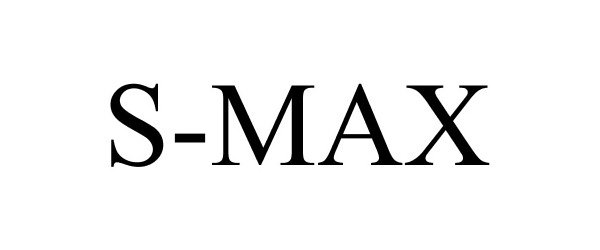 S-MAX 85158380 not registered Dead/Abandoned |
Specialty Fertilizer Products, LLC 2010-10-21 |
 S-MAX 79169853 5139365 Live/Registered |
KSB SE & Co. KGaA 2015-06-03 |
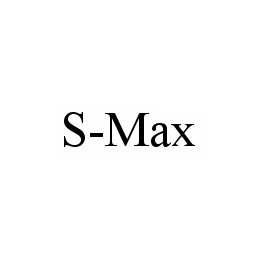 S-MAX 78551855 3319740 Dead/Cancelled |
Yi, Joon Tae 2005-01-21 |
 S-MAX 77925795 not registered Dead/Abandoned |
Ford Motor Company 2010-02-02 |
 S-MAX 77880987 4023369 Dead/Cancelled |
Eaton Corporation 2009-11-25 |
 S-MAX 77819701 not registered Dead/Abandoned |
Ex One Company, LLC, The 2009-09-03 |
 S-MAX 77610054 4061799 Live/Registered |
NAKANISHI INC. 2008-11-07 |
 S-MAX 77061302 not registered Dead/Abandoned |
Ford Motor Company 2006-12-11 |
 S-MAX 74396507 1820116 Dead/Cancelled |
Response Analysis Corporation 1993-06-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.