pde-neo
GUDID 04582389010093
HAMAMATSU PHOTONICS K.K.
Surgical microscope fluorescent angiography system| Primary Device ID | 04582389010093 |
| NIH Device Record Key | e6c77787-8cbc-4f72-8671-04b591956eb7 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | pde-neo |
| Version Model Number | C10935-40 |
| Company DUNS | 704524537 |
| Company Name | HAMAMATSU PHOTONICS K.K. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | true |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 04582389010093 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Notification: K133719
FDA Product Code
| OWN | Confocal Optical Imaging |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 3 |
| Public Version Date | 2018-07-06 |
| Device Publish Date | 2016-09-23 |
Devices Manufactured by HAMAMATSU PHOTONICS K.K.
| 04582389010871 - PDE-GEN3 | 2023-06-09 |
| 04582389010659 - NanoZoomer S360MD Slide scanner system | 2022-10-10 |
| 04582389010031 - PDE | 2018-07-06 |
| 04582389010062 - NIRO-200NX | 2018-07-06 |
| 04582389010093 - pde-neo | 2018-07-06 |
| 04582389010093 - pde-neo | 2018-07-06 |
| 04582389010543 - pde-neoII | 2018-07-06 |
Trademark Results [pde-neo]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
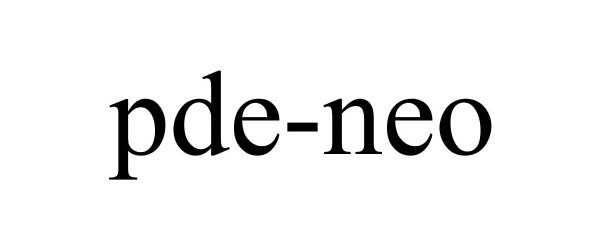 PDE-NEO 85711737 4641678 Live/Registered |
Hamamatsu Photonics K.K. 2012-08-23 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.