medex
GUDID 06971858364290
Diode Laser System
Jiangxi Medex Technology Co., Ltd.
General/multiple surgical diode laser system| Primary Device ID | 06971858364290 |
| NIH Device Record Key | bfb32311-06b5-4c7d-9ce4-62a53bb4165c |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | medex |
| Version Model Number | LaserPro D810 |
| Company DUNS | 544457636 |
| Company Name | Jiangxi Medex Technology Co., Ltd. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | true |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 06971858364290 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Notification: K243141
FDA Product Code
| GEX | Powered Laser Surgical Instrument |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2026-02-11 |
| Device Publish Date | 2026-02-03 |
Devices Manufactured by Jiangxi Medex Technology Co., Ltd.
| 06971858364290 - medex | 2026-02-11Diode Laser System |
| 06971858364290 - medex | 2026-02-11 Diode Laser System |
| 06971858366256 - SLT | 2025-01-01 |
| 06971858360162 - SLT | 2023-11-20 |
| 06971858363927 - LaserPro | 2022-11-25 |
Trademark Results [medex]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 MEDEX 98465437 not registered Live/Pending |
Medex LLC 2024-03-25 |
 MEDEX 97184090 not registered Live/Pending |
Audio Visual Preservation Solutions Inc. 2021-12-22 |
 MEDEX 90095352 not registered Live/Pending |
Medical Explorers LLC 2020-08-05 |
 MEDEX 86861622 5172611 Live/Registered |
INMAR SUPPLY CHAIN SOLUTIONS, LLC 2015-12-30 |
 MEDEX 86094499 not registered Dead/Abandoned |
Medex International, Inc. 2013-10-17 |
 MEDEX 85417170 not registered Dead/Abandoned |
Medex Products Corp.. 2011-09-07 |
 MEDEX 85340768 4087491 Live/Registered |
GOEX Corporation 2011-06-08 |
 MEDEX 80992957 0992957 Dead/Cancelled |
L. S. Reed & Company 0000-00-00 |
 MEDEX 79360752 not registered Live/Pending |
MEDEX, zivilska industrija, d.o.o. 2022-09-22 |
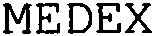 MEDEX 79162176 4920082 Live/Registered |
Pedex GmbH 2014-10-17 |
 MEDEX 78299234 3243493 Dead/Cancelled |
Operation Blessing International Reliefand Development Corporation 2003-09-11 |
 MEDEX 78299233 2879610 Dead/Cancelled |
Operation Blessing International Reliefand Development Corporation 2003-09-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.