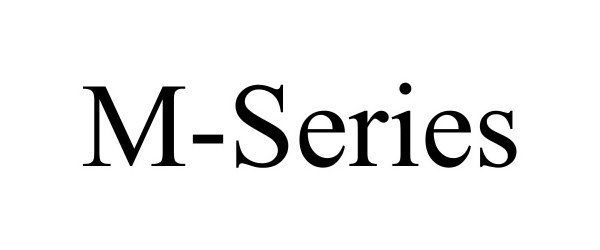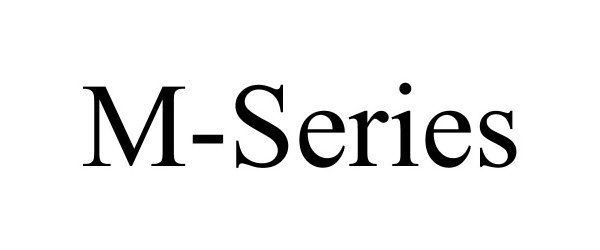M-SERIES
GUDID 10688503000108
Patient lifter
Savaria Concord Lifts Inc
Overhead track patient lifting/transfer system| Primary Device ID | 10688503000108 |
| NIH Device Record Key | ff4629ed-2a76-490f-abbb-bc431a69d253 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | M-SERIES |
| Version Model Number | M2F272-1200-00-NA |
| Company DUNS | 245449095 |
| Company Name | Savaria Concord Lifts Inc |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | true |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | false |
| Serial Number | true |
| Manufacturing Date | true |
| Expiration Date | false |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 10688503000108 [Primary] |
FDA Product Code
| FSA | Lift, Patient, Non-Ac-Powered |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2023-07-28 |
| Device Publish Date | 2023-07-20 |
On-Brand Devices [M-SERIES]
| 10688503000016 | Patient lifter |
| 10688503000009 | Patient lifter |
| 10688503000177 | Patient lifter |
| 10688503000160 | Patient lifter |
| 10688503000153 | Patient lifter |
| 10688503000146 | Patient lifter |
| 10688503000139 | Patient lifter |
| 10688503000122 | Patient lifter |
| 10688503000115 | Patient lifter |
| 10688503000108 | Patient lifter |
| 10688503000092 | Patient lifter |
| 10688503000085 | Patient lifter |
| 10688503000078 | Patient lifter |
| 10688503000061 | Patient lifter |
| 10688503000054 | Patient lifter |
| 10688503000047 | Patient lifter |
| 10688503000030 | Patient lifter |
| 10688503000023 | Patient lifter |
| 10688503000184 | Patient lifter |
| 10688503000191 | Patient lifter |
| 10688503000238 | Patient lifter |
| 10688503000221 | Patient lifter |
| 10688503000214 | Patient lifter |
| 10688503000207 | Patient lifter |
| 10688503000245 | Patient lifter |
| 10688503000252 | Patient lifter |
| 10688503000290 | Patient lifter |
| 10688503000283 | Patient lifter |
| 10688503000276 | Patient lifter |
| 10688503000269 | Patient lifter |
| 10688503000320 | Patient lifter |
| 10688503000313 | Patient lifter |
| 10688503000306 | Patient lifter |
| 10688503000344 | Patient lifter |
| 10688503000337 | Patient lifter |
| 10688503000351 | Patient lifter |
| 10688503000368 | Patient lifter |
| 10688503000375 | Patient lifter |
| 10688503000382 | Patient lifter |
| 10688503000405 | Patient lifter |
| 10688503000399 | Patient lifter |
| 10688503000412 | Patient lifter |
| 10688503000429 | Patient lifter |
| 10688503000450 | Patient lifter |
| 10688503000443 | Patient lifter |
| 10688503000436 | Patient lifter |
| 10688503000467 | Patient lifter |
| 10688503000528 | Patient lifter |
| 10688503000511 | Patient lifter |
| 10688503000504 | Patient lifter |
Trademark Results [M-SERIES]
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.