MAXCHECK CDTC121
GUDID 10852359007066
12 Panel Rapid Multi-Drug Test Cup
MAXPERT MEDICAL
Cannabinoid IVD, kit, immunochromatographic test (ICT), rapid| Primary Device ID | 10852359007066 |
| NIH Device Record Key | bb67b1e0-bfe3-4e46-a0dc-c02478b56d60 |
| Commercial Distribution Discontinuation | 2018-12-12 |
| Commercial Distribution Status | Not in Commercial Distribution |
| Brand Name | MAXCHECK |
| Version Model Number | CDTC121 |
| Catalog Number | CDTC121 |
| Company DUNS | 066886996 |
| Company Name | MAXPERT MEDICAL |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | true |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00852359007069 [Primary] |
| GS1 | 10852359007066 [Package] Contains: 00852359007069 Package: [25 Units] Discontinued: 2018-12-12 Not in Commercial Distribution |
| GS1 | 20852359007063 [Package] Package: [4 Units] Discontinued: 2018-12-12 Not in Commercial Distribution |
FDA Product Code
| LDJ | Enzyme Immunoassay, Cannabinoids |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 4 |
| Public Version Date | 2018-12-14 |
| Device Publish Date | 2017-12-19 |
On-Brand Devices [MAXCHECK]
| 10852359007066 | 12 Panel Rapid Multi-Drug Test Cup |
| 20852359007049 | One Step Pregnancy (HCG) Urine Test Strip |
Trademark Results [MAXCHECK]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
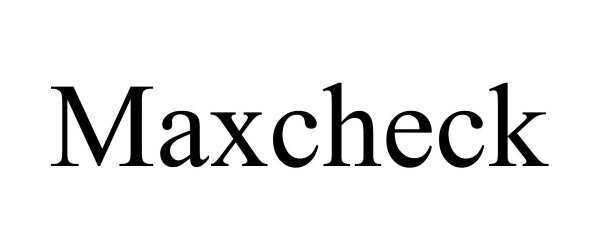 MAXCHECK 97073439 not registered Live/Pending |
Hefei Danneng Trading Co., Ltd 2021-10-13 |
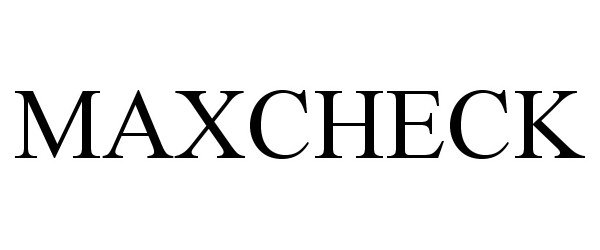 MAXCHECK 87402631 5306663 Live/Registered |
Maxpert Medical, LLC 2017-04-07 |
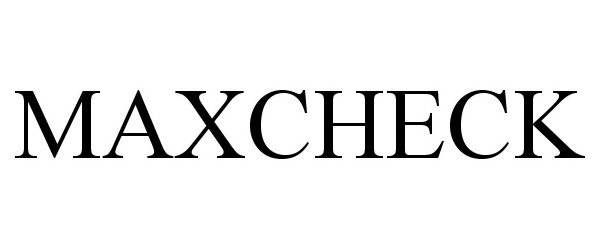 MAXCHECK 86211304 not registered Dead/Abandoned |
Max2 Inc. 2014-03-05 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.