PREGABLE
GUDID 16970429840161
HCG TEST
Co-Innovation Biotech Co., Ltd
Total human chorionic gonadotropin (HCG) IVD, kit, immunochromatographic test (ICT), rapid| Primary Device ID | 16970429840161 |
| NIH Device Record Key | 1c7c0b51-e7c0-4cde-90f2-ac1ab9f8e84d |
| Commercial Distribution Discontinuation | 2021-06-11 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | PREGABLE |
| Version Model Number | 1 |
| Company DUNS | 421302978 |
| Company Name | Co-Innovation Biotech Co., Ltd |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | true |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | true |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 06970429840164 [Primary] |
| GS1 | 16970429840161 [Package] Contains: 06970429840164 Package: [3 Units] Discontinued: 2021-06-11 In Commercial Distribution |
| GS1 | 26970429840168 [Package] Contains: 06970429840164 Package: [20 Units] Discontinued: 2021-06-11 In Commercial Distribution |
| GS1 | 36970429840165 [Package] Contains: 06970429840164 Package: [10 Units] Discontinued: 2021-06-11 In Commercial Distribution |
| GS1 | 46970429840162 [Package] Contains: 06970429840164 Package: [30 Units] Discontinued: 2021-06-11 In Commercial Distribution |
FDA Product Code
| LCX | Kit, Test, Pregnancy, Hcg, Over The Counter |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2021-06-10 |
| Device Publish Date | 2021-06-02 |
Devices Manufactured by Co-Innovation Biotech Co., Ltd
| 16970429843414 - co-innovation | 2025-05-27 |
| 16970429843315 - co-innovation | 2024-08-28 |
| 16970429841342 - DTPM | 2023-12-18 |
| 16970429841632 - DTPM | 2023-12-18 |
| 16970429840154 - HCG TEST STRIP | 2023-07-06 |
| 16970429840178 - co-innovation | 2023-05-08 |
| 16970429841816 - co-innovation | 2022-11-21 Rapid Multi-drug Test Cup |
| 16970429841441 - co-innovation | 2022-01-24 14P Drug Test |
Trademark Results [PREGABLE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
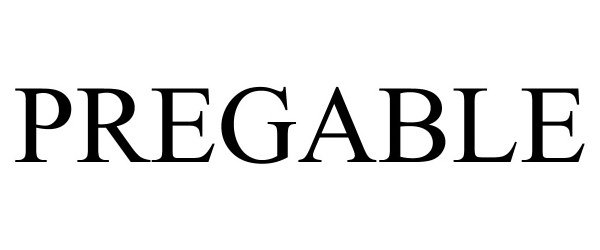 PREGABLE 90693426 not registered Live/Pending |
SMILELAB. CO.,LTD. 2021-05-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.