PROCEED PVPS
GUDID 20705031132419
Polypropylene Nonabsorbable Synthetic Surgical Mesh
Johnson & Johnson International Inc.
Abdominal hernia surgical mesh, synthetic polymer, non-bioabsorbable| Primary Device ID | 20705031132419 |
| NIH Device Record Key | da04ca57-5c65-48b1-9e5a-dd3b0cc6519d |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | PROCEED |
| Version Model Number | PVPS |
| Catalog Number | PVPS |
| Company DUNS | 372587696 |
| Company Name | Johnson & Johnson International Inc. |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Customer Support Contacts
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx | |
| Phone | +1(877)384-4266 |
| xxx@xxx.xxx |
Operating and Storage Conditions
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
| Storage Environment Temperature | Between 0 and 25 Degrees Celsius |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 10705031132412 [Primary] |
| GS1 | 20705031132419 [Package] Contains: 10705031132412 Package: BOX [2 Units] In Commercial Distribution |
FDA Product Code
| FTL | Mesh, surgical, polymeric |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 4 |
| Public Version Date | 2019-07-05 |
| Device Publish Date | 2015-09-01 |
On-Brand Devices [PROCEED]
| 20705031132419 | Polypropylene Nonabsorbable Synthetic Surgical Mesh |
| 20705031132402 | Polypropylene Nonabsorbable Synthetic Surgical Mesh |
Trademark Results [PROCEED]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 PROCEED 90163126 not registered Live/Pending |
Hayes, Michael 2020-09-07 |
 PROCEED 90054319 not registered Live/Pending |
Shedrain Corporation 2020-07-15 |
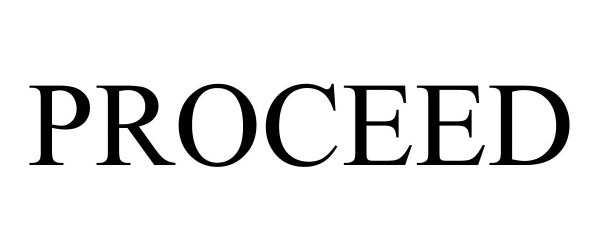 PROCEED 88980517 not registered Live/Pending |
Providence St. Joseph Health 2020-06-11 |
 PROCEED 88960904 not registered Live/Pending |
Providence St. Joseph Health 2020-06-11 |
 PROCEED 88050288 not registered Live/Pending |
Straighterline, Inc. 2018-07-24 |
 PROCEED 87792925 not registered Dead/Abandoned |
K&R Wines, LLC 2018-02-10 |
 PROCEED 87447372 not registered Dead/Abandoned |
Okamura Corporation 2017-05-12 |
 PROCEED 87297494 5421251 Live/Registered |
FCi Federal, Inc. 2017-01-11 |
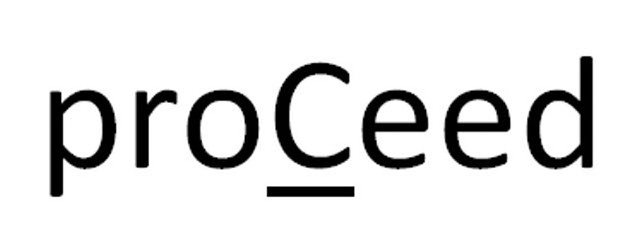 PROCEED 86292467 5186836 Live/Registered |
AbbVie Inc. 2014-05-27 |
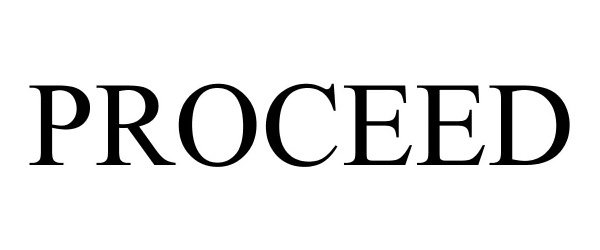 PROCEED 78908961 3360875 Live/Registered |
GOLDEN ARTIST COLORS, INC. 2006-06-15 |
 PROCEED 78613984 not registered Dead/Abandoned |
Bayer Aktiengesellschaft 2005-04-21 |
 PROCEED 78597328 3399496 Dead/Cancelled |
Caterpillar Inc. 2005-03-29 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.