MONOMID 1665B
GUDID 20790986002481
C.P. MEDICAL CORPORATION
Nylon suture, non-bioabsorbable, monofilament| Primary Device ID | 20790986002481 |
| NIH Device Record Key | ec1f28e0-6e09-4a14-9d5f-06bf1c5701d2 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | MONOMID |
| Version Model Number | 1665B |
| Catalog Number | 1665B |
| Company DUNS | 807404611 |
| Company Name | C.P. MEDICAL CORPORATION |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 10790986002484 [Primary] |
| GS1 | 20790986002481 [Package] Contains: 10790986002484 Package: [12 Units] In Commercial Distribution |
FDA Product Code
| GAR | Suture, Nonabsorbable, Synthetic, Polyamide |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | Update |
| Device Record Status | Published |
| Public Version Number | 6 |
| Public Version Date | 2021-02-05 |
| Device Publish Date | 2015-12-21 |
On-Brand Devices [MONOMID]
| 20790986002672 | 1629B |
| 20790986002665 | 919B |
| 20790986002658 | 915B |
| 20790986002634 | 795B |
| 20790986002627 | 738B |
| 20790986002610 | 1893B |
| 20790986002603 | 1865B |
| 20790986002597 | 1864B |
| 20790986002580 | 1856B |
| 20790986002573 | 669B |
| 20790986002566 | 1855B |
| 20790986002559 | 1698B |
| 20790986002542 | 1696B |
| 20790986002535 | 1674B |
| 20790986002528 | 1669B |
| 20790986002511 | 1668B |
| 20790986002504 | 1667B |
| 20790986002498 | 1666B |
| 20790986002481 | 1665B |
| 20790986002474 | 1663B |
| 20790986002467 | 1273B |
| 20790986002450 | 1099B |
| 20790986002443 | 1098B |
| 20790986002436 | 945B |
| 20790986002429 | 944B |
| 20790986002412 | 931B |
| 20790986002405 | 928B |
| 20790986002399 | 913B |
| 20790986002382 | 665B |
| 20790986002375 | 911B |
| 20790986002368 | 699B |
| 20790986002351 | 698B |
| 20790986002344 | 697B |
| 20790986002337 | 691B |
| 20790986002320 | 690B |
| 20790986002313 | 668B |
| 20790986002306 | 667B |
| 20790986002290 | 665M |
| 20790986002283 | 664M |
| 20790986002276 | 664B |
| 20790986002269 | 663M |
| 20790986002252 | 663B |
| 20790986002245 | 662M |
| 20790986002238 | 662B |
| 20790986002221 | 661B |
| 20790986002214 | 660B |
| 20790986002207 | 628B |
| 20790986002191 | 627B |
| 20790986002184 | 593B |
Trademark Results [MONOMID]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 MONOMID 77155202 3368129 Live/Registered |
CP Medical 2007-04-12 |
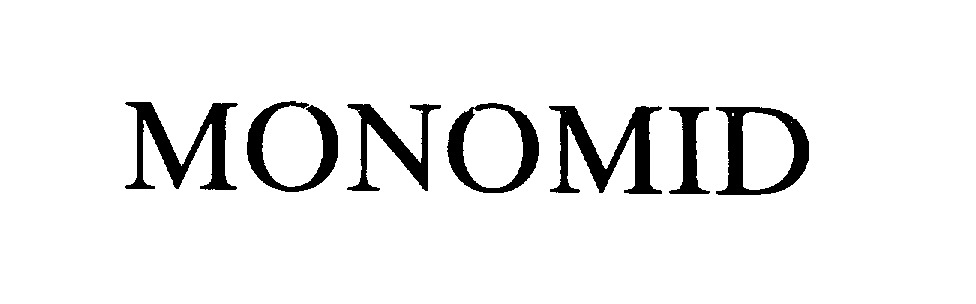 MONOMID 74436661 1875876 Dead/Cancelled |
C.P. MEDICAL 1993-09-17 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.