Mx-Grafter
GUDID 20860002195005
The Mx-Grafter Bone Grafting System is a manual, self-contained, sterile, single use instrument for the removal, harvest, and grafting of autogenous bone in surgical applications. The instrument is comprised of a plastic handle and stainless steel blade. The blade shaves bone from cortical surfaces producing short convoluted ribbons. While being cut, shavings combine with blood and flow into the handle. This graft material, an osseous coagulum, is then delivered with the handle directly to the recipient site, or to a bowl.
MAXILON LABORATORIES INC
Bone collection scraper| Primary Device ID | 20860002195005 |
| NIH Device Record Key | 6b5a43cb-cb6d-4009-9f8a-d91f77c64b01 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | Mx-Grafter |
| Version Model Number | 11302 |
| Company DUNS | 949461941 |
| Company Name | MAXILON LABORATORIES INC |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | true |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | true |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Customer Support Contacts
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com | |
| Phone | (603)-594-9300 |
| maxilon@maxilon.com |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 20860002195005 [Primary] |
FDA Product Code
| GAC | Rasp, Surgical, General & Plastic Surgery |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2020-08-11 |
| Device Publish Date | 2020-08-03 |
On-Brand Devices [Mx-Grafter]
| 00860002195001 | The Mx-Grafter Bone Grafting System is a manual, self-contained, sterile, single use instrument |
| 20860002195005 | The Mx-Grafter Bone Grafting System is a manual, self-contained, sterile, single use instrument |
Trademark Results [Mx-Grafter]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
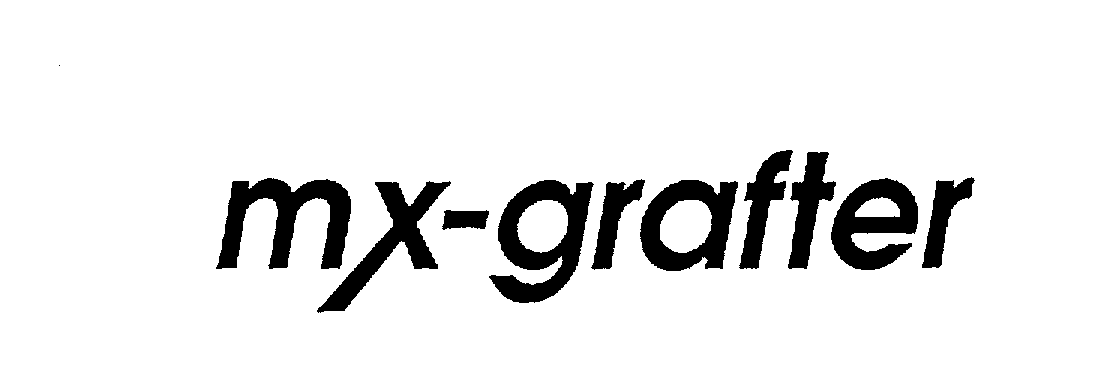 MX-GRAFTER 75168632 2198891 Dead/Expired |
MAXILON LABORATORIES, INC. 1996-09-19 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.