Sparkle™ Prophy Angle
GUDID 50732224002939
Prophy Angle,Tapered Brush
CROSSTEX INTERNATIONAL, INC.
Dental-professional prophylaxis brush, single-use| Primary Device ID | 50732224002939 |
| NIH Device Record Key | 03d50d0e-210a-4588-971c-19a8d54e7fbe |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | Sparkle™ Prophy Angle |
| Version Model Number | TPATBC1 |
| Company DUNS | 057728685 |
| Company Name | CROSSTEX INTERNATIONAL, INC. |
| Device Count | 100 |
| DM Exempt | true |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | false |
| OTC Over-The-Counter | false |
Customer Support Contacts
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00732224002934 [Unit of Use] |
| GS1 | 10732224002931 [Primary] |
| GS1 | 50732224002939 [Package] Contains: 10732224002931 Package: [4 Units] In Commercial Distribution |
FDA Pre-market Approvals/Notifications & deNovo
FDA Product Code
| EFA | Handpiece, belt and/or gear driven, dental |
| EGS | Handpiece, contra- and right-angle attachment, dental |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | false |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2022-10-07 |
| Device Publish Date | 2022-09-29 |
On-Brand Devices [Sparkle™ Prophy Angle]
| 50732224002953 | Prophy Angles, Soft Cup, Purple |
| 50732224002946 | Prophy Angles, Disposable, Firm Cup White |
| 50732224002939 | Prophy Angle,Tapered Brush |
| 50732224002922 | Prophy Angles, Soft Cup, Purple |
| 50732224002915 | Prophy Angle, Firm Cup, White |
Trademark Results [Sparkle]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SPARKLE 98759185 not registered Live/Pending |
FlevoFlora B.V. 2024-09-19 |
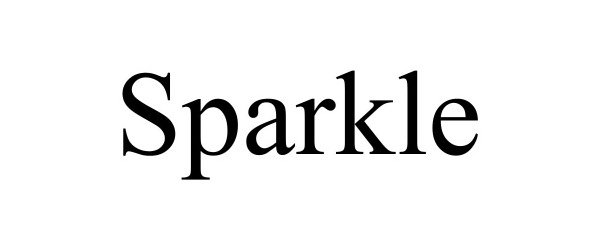 SPARKLE 98643643 not registered Live/Pending |
Feng, Jie 2024-07-11 |
 SPARKLE 98210945 not registered Live/Pending |
GPCP IP Holdings LLC 2023-10-05 |
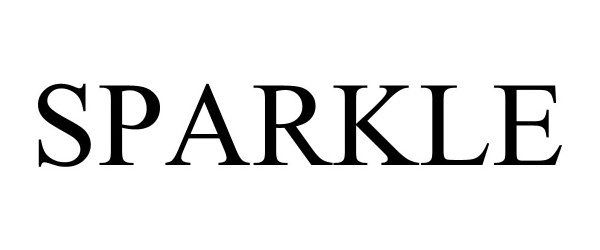 SPARKLE 97763472 not registered Live/Pending |
Mehta, Jinesh 2023-01-21 |
 SPARKLE 97668731 not registered Live/Pending |
Spark*l, a California corporation 2022-11-08 |
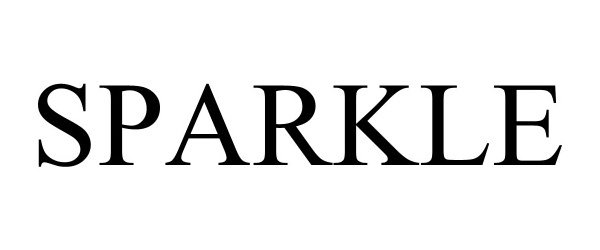 SPARKLE 97610463 not registered Live/Pending |
Pet Services Corp 2022-09-28 |
 SPARKLE 97493266 not registered Live/Pending |
GPCP IP Holdings LLC 2022-07-07 |
 SPARKLE 97114273 not registered Live/Pending |
Sparkle Products LLC 2021-11-08 |
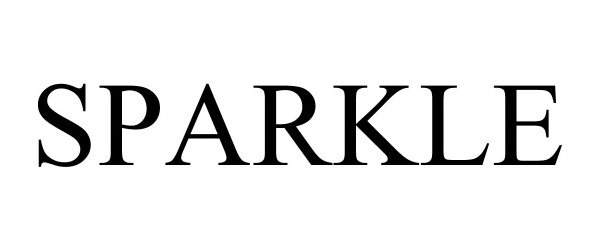 SPARKLE 97114257 not registered Live/Pending |
Sparkle Products LLC 2021-11-08 |
 SPARKLE 97092517 not registered Live/Pending |
Innovision Medical, PA 2021-10-26 |
 SPARKLE 97052162 not registered Live/Pending |
California Exotic Novelties, LLC 2021-09-29 |
 SPARKLE 90980372 not registered Live/Pending |
Sparkle Innovations Inc. 2020-10-13 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.