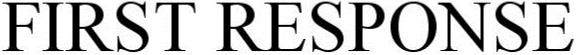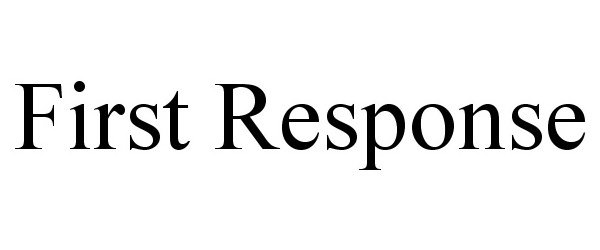Mark Image
Registration | Serial | Company Trademark
Application Date |
|---|
 FIRST RESPONSE 98324007 not registered Live/Pending |
PokeBean, LLC 2023-12-20 |
 FIRST RESPONSE 98066264 not registered Live/Pending |
Church & Dwight Co., Inc. 2023-06-30 |
 FIRST RESPONSE 98064725 not registered Live/Pending |
Church & Dwight Co., Inc. 2023-06-29 |
 FIRST RESPONSE 97036611 not registered Live/Pending |
Safe Life Defense, LLC 2021-09-20 |
 FIRST RESPONSE 87612048 5458328 Live/Registered |
Turbo-Chem International, Inc. 2017-09-18 |
 FIRST RESPONSE 86945123 not registered Dead/Abandoned |
CHURCH & DWIGHT CO., INC. 2016-03-18 |
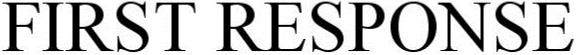 FIRST RESPONSE 86881378 not registered Dead/Abandoned |
RGR Enterprises, LLC. 2016-01-20 |
 FIRST RESPONSE 86637428 4970396 Live/Registered |
CHURCH & DWIGHT CO., INC. 2015-05-21 |
 FIRST RESPONSE 85975189 4748541 Live/Registered |
CHURCH & DWIGHT CO., INC. 2013-02-21 |
 FIRST RESPONSE 85906991 4946542 Live/Registered |
CHURCH & DWIGHT CO., INC. 2013-04-17 |
 FIRST RESPONSE 85856459 not registered Dead/Abandoned |
CHURCH & DWIGHT CO., INC. 2013-02-21 |
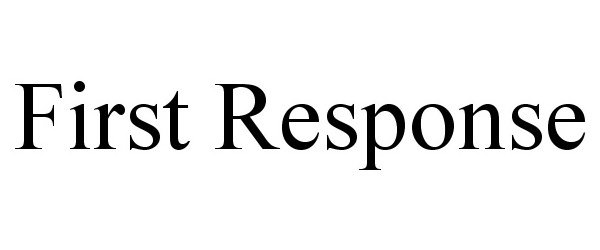 FIRST RESPONSE 85158620 not registered Dead/Abandoned |
Universal Storage Containers LLC 2010-10-21 |