Triple Twist 10524T
GUDID M68510524T0
Triple Twist 5.5mm PEEK Suture Anchor
PARCUS MEDICAL LLC
Tendon/ligament bone anchor, non-bioabsorbable| Primary Device ID | M68510524T0 |
| NIH Device Record Key | 8c835ab6-df28-421d-ac04-2f4bb7755529 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | Triple Twist |
| Version Model Number | 10524T |
| Catalog Number | 10524T |
| Company DUNS | 859961950 |
| Company Name | PARCUS MEDICAL LLC |
| Device Count | 1 |
| DM Exempt | true |
| Pre-market Exempt | false |
| MRI Safety Status | MR Safe |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | true |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | false |
| Expiration Date | true |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Customer Support Contacts
| Phone | 941-755-7965 |
| CustomerService@ParcusMedical.com |
Device Dimensions
| Outer Diameter | 5.5 Millimeter |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| HIBCC | M68510524T0 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Notification: K120942
FDA Product Code
| MBI | Fastener, Fixation, Nondegradable, Soft Tissue |
Sterilization
| Steralize Prior To Use | false |
| Device Is Sterile | true |
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2019-07-03 |
| Device Publish Date | 2019-06-25 |
On-Brand Devices [Triple Twist]
| M68510525T0 | Triple Twist 6.5mm PEEK Suture Anchor |
| M68510524T0 | Triple Twist 5.5mm PEEK Suture Anchor |
Trademark Results [Triple Twist]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
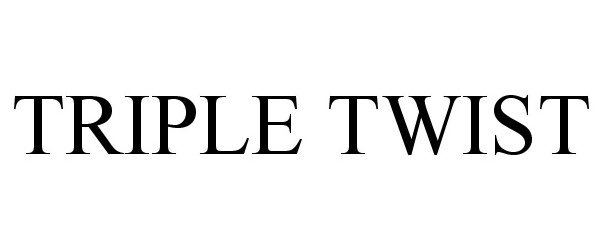 TRIPLE TWIST 78903499 3215884 Live/Registered |
Boyer Candy Company, Inc., The 2006-06-08 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.