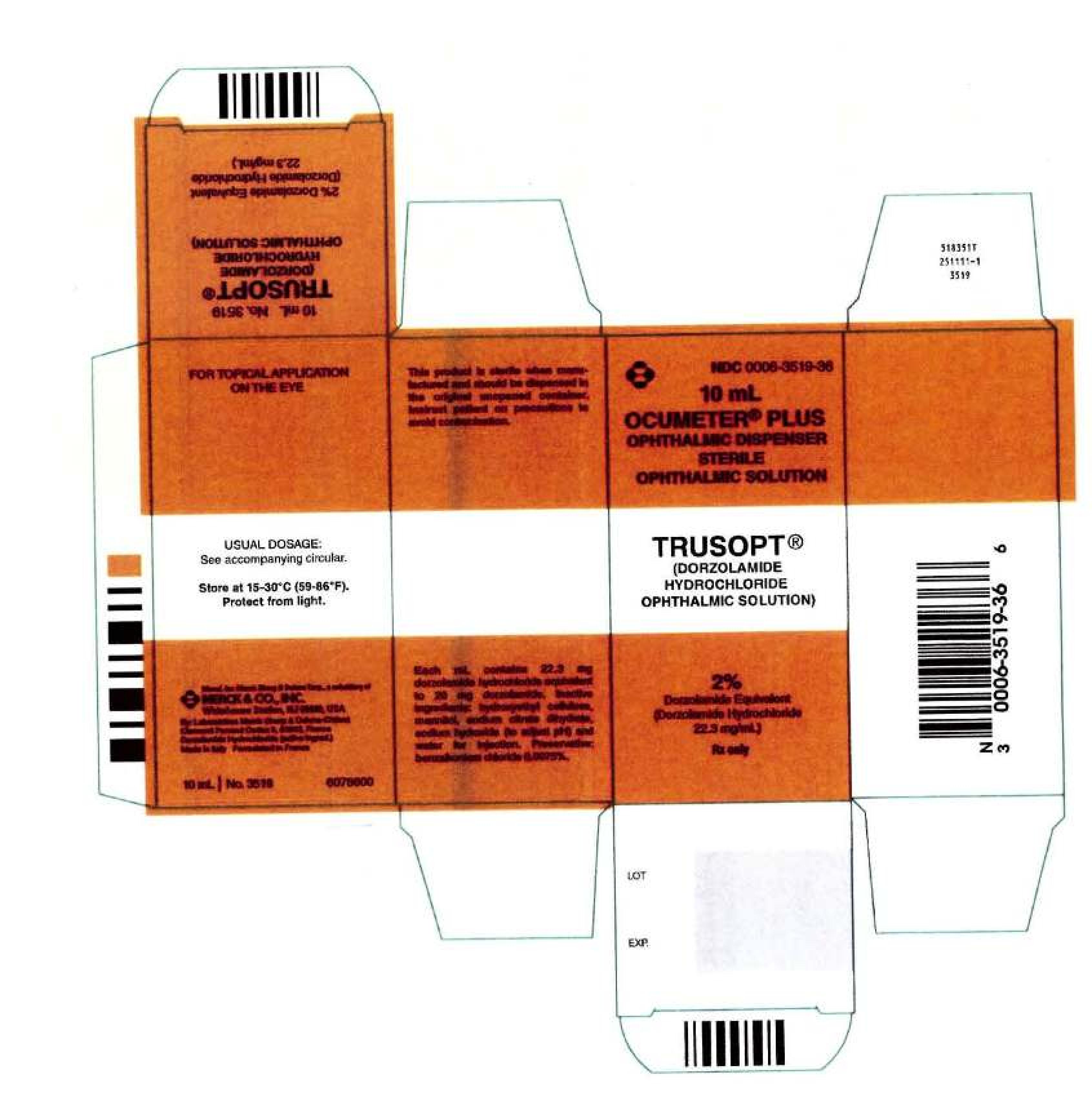
NDC 0006-3519
TRUSOPT
Dorzolamide Hydrochloride
TRUSOPT is a Ophthalmic Solution in the Human Prescription Drug category. It is labeled and distributed by Merck Sharp & Dohme Corp.. The primary component is Dorzolamide Hydrochloride.
| Product ID | 0006-3519_1267564a-94ab-48ef-80a2-15bb583901d0 |
| NDC | 0006-3519 |
| Product Type | Human Prescription Drug |
| Proprietary Name | TRUSOPT |
| Generic Name | Dorzolamide Hydrochloride |
| Dosage Form | Solution |
| Route of Administration | OPHTHALMIC |
| Marketing Start Date | 1994-12-09 |
| Marketing Category | NDA / NDA |
| Application Number | NDA020408 |
| Labeler Name | Merck Sharp & Dohme Corp. |
| Substance Name | DORZOLAMIDE HYDROCHLORIDE |
| Active Ingredient Strength | 20 mg/mL |
| Pharm Classes | Carbonic Anhydrase Inhibitor [EPC],Carbonic Anhydrase Inhibitors [MoA] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2022-12-31 |
Packaging
NDC 0006-3519-36
1 BOTTLE, DROPPER in 1 CARTON (0006-3519-36) > 10 mL in 1 BOTTLE, DROPPER
| Marketing Start Date | 1994-12-09 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 0006-3519-36 [00006351936]
TRUSOPT SOLUTION
| Marketing Category | NDA |
| Application Number | NDA020408 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | ML |
| Marketing Start Date | 1994-12-09 |
| Inactivation Date | 2020-01-31 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| DORZOLAMIDE HYDROCHLORIDE | 20 mg/mL |
OpenFDA Data
| SPL SET ID: | 26d26f80-aebf-441c-6383-327cb95eae4d |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI |
Pharmacological Class
- Carbonic Anhydrase Inhibitor [EPC]
- Carbonic Anhydrase Inhibitors [MoA]
NDC Crossover Matching brand name "TRUSOPT" or generic name "Dorzolamide Hydrochloride"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0006-3519 | TRUSOPT | dorzolamide hydrochloride |
| 50090-2768 | Dorzolamide HCl | Dorzolamide Hydrochloride |
| 0093-7618 | Dorzolamide Hydrochloride | Dorzolamide Hydrochloride |
| 50090-1246 | Dorzolamide Hydrochloride | Dorzolamide Hydrochloride |
| 50090-5280 | DORZOLAMIDE HYDROCHLORIDE | dorzolamide hydrochloride |
| 50383-232 | Dorzolamide Hydrochloride | Dorzolamide Hydrochloride |
| 55545-1008 | Dorzolamide hydrochloride | Dorzolamide hydrochloride |
| 60429-114 | Dorzolamide Hydrochloride | Dorzolamide Hydrochloride |
| 61314-019 | DORZOLAMIDE HYDROCHLORIDE | dorzolamide hydrochloride |
| 62332-519 | Dorzolamide Hydrochloride | Dorzolamide Hydrochloride |
| 68083-489 | Dorzolamide Hydrochloride | Dorzolamide Hydrochloride |
| 69315-304 | Dorzolamide hydrochloride | Dorzolamide hydrochloride |
| 50090-4882 | Dorzolamide Hydrochloride Ophthalmic | Dorzolamide Hydrochloride |
| 42571-141 | Dorzolamide Hydrochloride Ophthalmic Solution | Dorzolamide Hydrochloride |
Trademark Results [TRUSOPT]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 TRUSOPT 74000109 1692342 Live/Registered |
MERCK & CO., INC. 1989-11-16 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.