NDC 0023-5301
Restasis MultiDose
Cyclosporine
Restasis MultiDose is a Ophthalmic Emulsion in the Human Prescription Drug category. It is labeled and distributed by Allergan, Inc.. The primary component is Cyclosporine.
| Product ID | 0023-5301_e579186c-6dfc-41c3-9cc9-a32d36352507 |
| NDC | 0023-5301 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Restasis MultiDose |
| Generic Name | Cyclosporine |
| Dosage Form | Emulsion |
| Route of Administration | OPHTHALMIC |
| Marketing Start Date | 2016-11-10 |
| Marketing Category | NDA / NDA |
| Application Number | NDA050790 |
| Labeler Name | Allergan, Inc. |
| Substance Name | CYCLOSPORINE |
| Active Ingredient Strength | 1 mg/mL |
| Pharm Classes | Calcineurin Inhibitor Immunosuppressant [EPC],Calcineurin Inhibitors [MoA],Cytochrome P450 3A4 Inhibitors [MoA],P-Glycoprotein Inhibitors [MoA] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2020-12-31 |
Packaging
NDC 0023-5301-01
1 BOTTLE in 1 CARTON (0023-5301-01) > 1.5 mL in 1 BOTTLE
| Marketing Start Date | 2016-11-10 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 0023-5301-05 [00023530105]
Restasis MultiDose EMULSION
| Marketing Category | NDA |
| Application Number | NDA050790 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | ML |
| Marketing Start Date | 2016-11-10 |
NDC 0023-5301-01 [00023530101]
Restasis MultiDose EMULSION
| Marketing Category | NDA |
| Application Number | NDA050790 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | ML |
| Marketing Start Date | 2016-11-10 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| CYCLOSPORINE | .5 mg/mL |
OpenFDA Data
| SPL SET ID: | 7224d810-bb96-4682-a942-3355e6e8061a |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI | |
| UPC Code |
Pharmacological Class
- Calcineurin Inhibitor Immunosuppressant [EPC]
- Calcineurin Inhibitors [MoA]
- Cytochrome P450 3A4 Inhibitors [MoA]
- P-Glycoprotein Inhibitors [MoA]
- Calcineurin Inhibitor Immunosuppressant [EPC]
- Calcineurin Inhibitors [MoA]
- Cytochrome P450 3A4 Inhibitors [MoA]
- P-Glycoprotein Inhibitors [MoA]
NDC Crossover Matching brand name "Restasis MultiDose" or generic name "Cyclosporine"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0023-5301 | Restasis MultiDose | cyclosporine |
| 0074-3108 | Gengraf | Cyclosporine |
| 0074-3109 | Gengraf | Cyclosporine |
| 0074-7269 | Gengraf | Cyclosporine |
| 0078-0246 | Neoral | cyclosporine |
| 0078-0248 | Neoral | cyclosporine |
| 0023-9163 | RESTASIS | cyclosporine |
| 0078-0109 | Sandimmune | cyclosporine |
| 0078-0110 | Sandimmune | cyclosporine |
| 0078-0240 | Sandimmune | cyclosporine |
| 0078-0241 | Sandimmune | cyclosporine |
Trademark Results [Restasis MultiDose]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
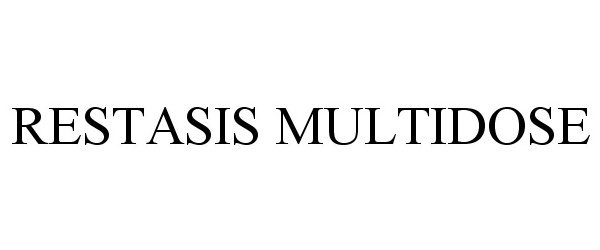 RESTASIS MULTIDOSE 87216956 5243078 Live/Registered |
Allergan, Inc. 2016-10-26 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.