NDC 0093-2140
Tri-Lo-Sprintec
Norgestimate And Ethinyl Estradiol
Tri-Lo-Sprintec is a Kit in the Human Prescription Drug category. It is labeled and distributed by Teva Pharmaceuticals Usa, Inc.. The primary component is .
| Product ID | 0093-2140_1bcec3e3-3175-447d-bca6-2513da7af313 |
| NDC | 0093-2140 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Tri-Lo-Sprintec |
| Generic Name | Norgestimate And Ethinyl Estradiol |
| Dosage Form | Kit |
| Marketing Start Date | 2015-12-30 |
| Marketing Category | ANDA / ANDA |
| Application Number | ANDA076784 |
| Labeler Name | Teva Pharmaceuticals USA, Inc. |
| Active Ingredient Strength | 0 |
| NDC Exclude Flag | N |
| Listing Certified Through | 2022-12-31 |
Packaging
NDC 0093-2140-62
3 POUCH in 1 CARTON (0093-2140-62) > 1 BLISTER PACK in 1 POUCH (0093-2140-28) > 1 KIT in 1 BLISTER PACK
| Marketing Start Date | 2015-12-30 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 0093-2140-28 [00093214028]
Tri-Lo-Sprintec KIT
| Marketing Category | ANDA |
| Application Number | ANDA076784 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 2015-12-30 |
NDC 0093-2140-62 [00093214062]
Tri-Lo-Sprintec KIT
| Marketing Category | ANDA |
| Application Number | ANDA076784 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 2015-12-30 |
Drug Details
OpenFDA Data
| SPL SET ID: | ddbeb312-af2b-4d22-ab7c-5ab2f3f8d5cb |
| Manufacturer | |
| RxNorm Concept Unique ID - RxCUI |
NDC Crossover Matching brand name "Tri-Lo-Sprintec" or generic name "Norgestimate And Ethinyl Estradiol"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0093-2140 | Tri-Lo-Sprintec | Norgestimate and Ethinyl Estradiol |
| 50090-2326 | Tri-Lo-Sprintec | Norgestimate and Ethinyl Estradiol |
| 68788-7036 | Tri-Lo-Sprintec | Tri-Lo-Sprintec |
| 71205-287 | Tri-Lo-Sprintec | Tri-Lo-Sprintec |
| 0781-4058 | Estarylla | Norgestimate and ethinyl estradiol |
| 16714-360 | Mono-Linyah | norgestimate and ethinyl estradiol |
| 0378-7286 | NORGESTIMATE AND ETHINYL ESTRADIOL | norgestimate and ethinyl estradiol |
| 0378-7293 | NORGESTIMATE AND ETHINYL ESTRADIOL | norgestimate and ethinyl estradiol |
| 17478-260 | Norgestimate and Ethinyl Estradiol | Norgestimate and Ethinyl Estradiol |
| 17478-261 | Norgestimate and Ethinyl Estradiol | Norgestimate and Ethinyl Estradiol |
| 42291-553 | Norgestimate and Ethinyl Estradiol | Norgestimate and ethinyl estradiol |
| 42291-565 | Norgestimate and Ethinyl Estradiol | norgestimate and ethinyl estradiol |
| 42291-590 | norgestimate and ethinyl estradiol | norgestimate and ethinyl estradiol |
| 50090-2259 | Norgestimate and Ethinyl Estradiol | Norgestimate and Ethinyl Estradiol |
| 0254-2029 | Previfem | Norgestimate and ethinyl estradiol |
| 0603-7642 | Previfem | norgestimate and ethinyl estradiol |
| 0555-9016 | Sprintec | Norgestimate and Ethinyl Estradiol |
| 21695-769 | Sprintec | Norgestimate and Ethinyl Estradiol |
| 0781-4060 | Tri-Estarylla | norgestimate and ethinyl estradiol |
| 16714-363 | Tri-Linyah | Norgestimate and Ethinyl Estradiol |
| 0781-4062 | Tri-Lo- Estarylla | norgestimate and ethinyl estradiol |
| 50090-2429 | TRI-LO-MARZIA | norgestimate and ethinyl estradiol |
| 0254-2030 | Tri-Previfem | norgestimate and ethinyl estradiol |
| 0603-7663 | Tri-Previfem | norgestimate and ethinyl estradiol |
| 0555-9018 | Tri-Sprintec | norgestimate and ethinyl estradiol |
| 21695-770 | Tri-Sprintec | norgestimate and ethinyl estradiol |
| 21695-407 | TriNessa | Norgestimate and Ethinyl Estradiol |
Trademark Results [Tri-Lo-Sprintec]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
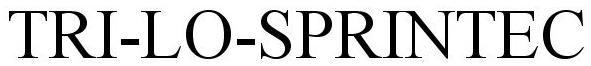 TRI-LO-SPRINTEC 77413014 3680756 Live/Registered |
Barr Laboratories, Inc. 2008-03-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.