NDC 17856-8237
SENNA
Senna
SENNA is a Oral Syrup in the Human Otc Drug category. It is labeled and distributed by Atlantic Biologicals Corp.. The primary component is Sennosides A And B.
| Product ID | 17856-8237_30e99a1b-cecf-4b8d-ac99-e59ed79d0ab4 |
| NDC | 17856-8237 |
| Product Type | Human Otc Drug |
| Proprietary Name | SENNA |
| Generic Name | Senna |
| Dosage Form | Syrup |
| Route of Administration | ORAL |
| Marketing Start Date | 2019-10-25 |
| Marketing Category | OTC MONOGRAPH NOT FINAL / OTC MONOGRAPH NOT FINAL |
| Application Number | part334 |
| Labeler Name | ATLANTIC BIOLOGICALS CORP. |
| Substance Name | SENNOSIDES A AND B |
| Active Ingredient Strength | 417 mg/237mL |
| NDC Exclude Flag | N |
| Listing Certified Through | 2021-12-31 |
Packaging
NDC 17856-8237-1
72 CUP, UNIT-DOSE in 1 BOX, UNIT-DOSE (17856-8237-1) > 5 mL in 1 CUP, UNIT-DOSE
| Marketing Start Date | 2020-05-04 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 17856-8237-3 [17856823703]
SENNA SYRUP
| Marketing Category | OTC monograph not final |
| Application Number | part334 |
| Product Type | HUMAN OTC DRUG |
| Marketing Start Date | 2020-05-04 |
NDC 17856-8237-4 [17856823704]
SENNA SYRUP
| Marketing Category | OTC monograph not final |
| Application Number | part334 |
| Product Type | HUMAN OTC DRUG |
| Marketing Start Date | 2020-05-04 |
NDC 17856-8237-2 [17856823702]
SENNA SYRUP
| Marketing Category | OTC monograph not final |
| Application Number | part334 |
| Product Type | HUMAN OTC DRUG |
| Marketing Start Date | 2020-05-04 |
NDC 17856-8237-1 [17856823701]
SENNA SYRUP
| Marketing Category | OTC monograph not final |
| Application Number | part334 |
| Product Type | HUMAN OTC DRUG |
| Marketing Start Date | 2020-05-04 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| SENNOSIDES A AND B | 417.12 mg/237mL |
NDC Crossover Matching brand name "SENNA" or generic name "Senna"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0220-4642 | Senna | SENNA LEAF |
| 0363-0180 | Senna | SENNOSIDES |
| 0363-8750 | Senna | Sennosides |
| 0363-8820 | Senna | Sennosides |
| 0536-1138 | SENNA | SENNA |
| 0615-7923 | SENNA | SENNA |
| 0904-6289 | Senna | Sennosides A and B |
| 0904-6434 | SENNA | SENNA |
| 11673-886 | Senna | Sennosides |
| 11822-1567 | SENNA | Sennosides |
| 11822-1571 | SENNA | Sennosides |
| 11822-3311 | SENNA | SENNOSIDES |
| 11822-6523 | SENNA | SENNOSIDES |
| 11822-7851 | SENNA | SENNOSIDES |
| 15631-0401 | SENNA | SENNA |
| 17714-122 | Senna | Sennosides |
| 17856-1107 | SENNA | SENNOSIDES A AND B |
| 17856-6289 | Senna | Sennosides A and B |
| 17856-8237 | SENNA | SENNA |
| 24451-289 | Senna | SENNOSIDES A AND B |
| 36800-135 | SENNA | SENNOSIDES |
| 37808-675 | Senna | Sennosides |
| 37808-782 | Senna | Sennosides |
| 68016-117 | Senna | Senna |
| 68016-745 | SENNA | SENNA |
| 68151-0648 | Senna | Senna |
| 68428-634 | Senna | Senna |
| 69618-048 | Senna | Senna |
| 69842-320 | SENNA | SENNA |
| 70000-0441 | SENNA | SENNA |
| 70166-636 | SENNA | SENNA |
| 71610-054 | SENNA | SENNA |
| 71919-625 | Senna | Senna |
| 45865-624 | SENNA | SENNA |
| 48433-301 | SENNA | SENNA |
| 53807-556 | Senna | Senna |
| 54162-007 | SENNA | SENNA |
| 57896-452 | Senna | Senna |
| 59779-586 | Senna | Senna |
| 61919-425 | SENNA | SENNA |
| 62011-0162 | Senna | Senna |
| 62011-0363 | SENNA | SENNA |
| 62011-0387 | SENNA | SENNA |
| 62011-0289 | SENNA | SENNA |
| 64418-1122 | Senna | Senna |
| 65437-087 | SENNA | SENNA |
| 67046-677 | SENNA | SENNA |
| 71406-105 | Senna | Senna |
| 71399-8237 | SENNA | SENNA |
| 45865-971 | Senna | Senna |
Trademark Results [SENNA]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SENNA 97868919 not registered Live/Pending |
Natty Collection LLC 2023-04-02 |
 SENNA 90583748 not registered Live/Pending |
Jeremey Decena 2021-03-17 |
 SENNA 90399285 not registered Live/Pending |
AYRTON SENNA EMPREENDIMENTOS LTDA. 2020-12-21 |
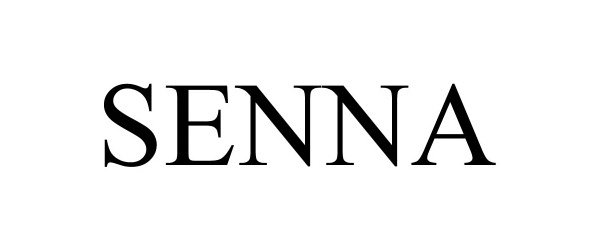 SENNA 90022160 not registered Live/Pending |
OPWEST DEVELOPMENT LLC 2020-06-26 |
 SENNA 88624114 not registered Live/Pending |
Ceritas Wines LLC 2019-09-19 |
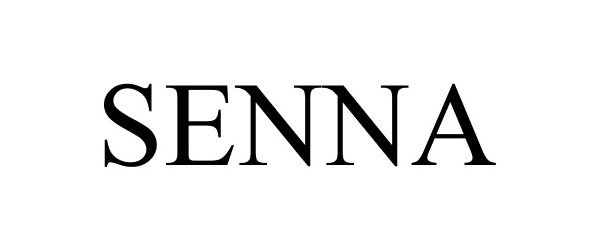 SENNA 87683504 5564030 Live/Registered |
OMM Imports Inc. 2017-11-14 |
 SENNA 76601884 3268781 Dead/Cancelled |
Studio RTA 2004-07-12 |
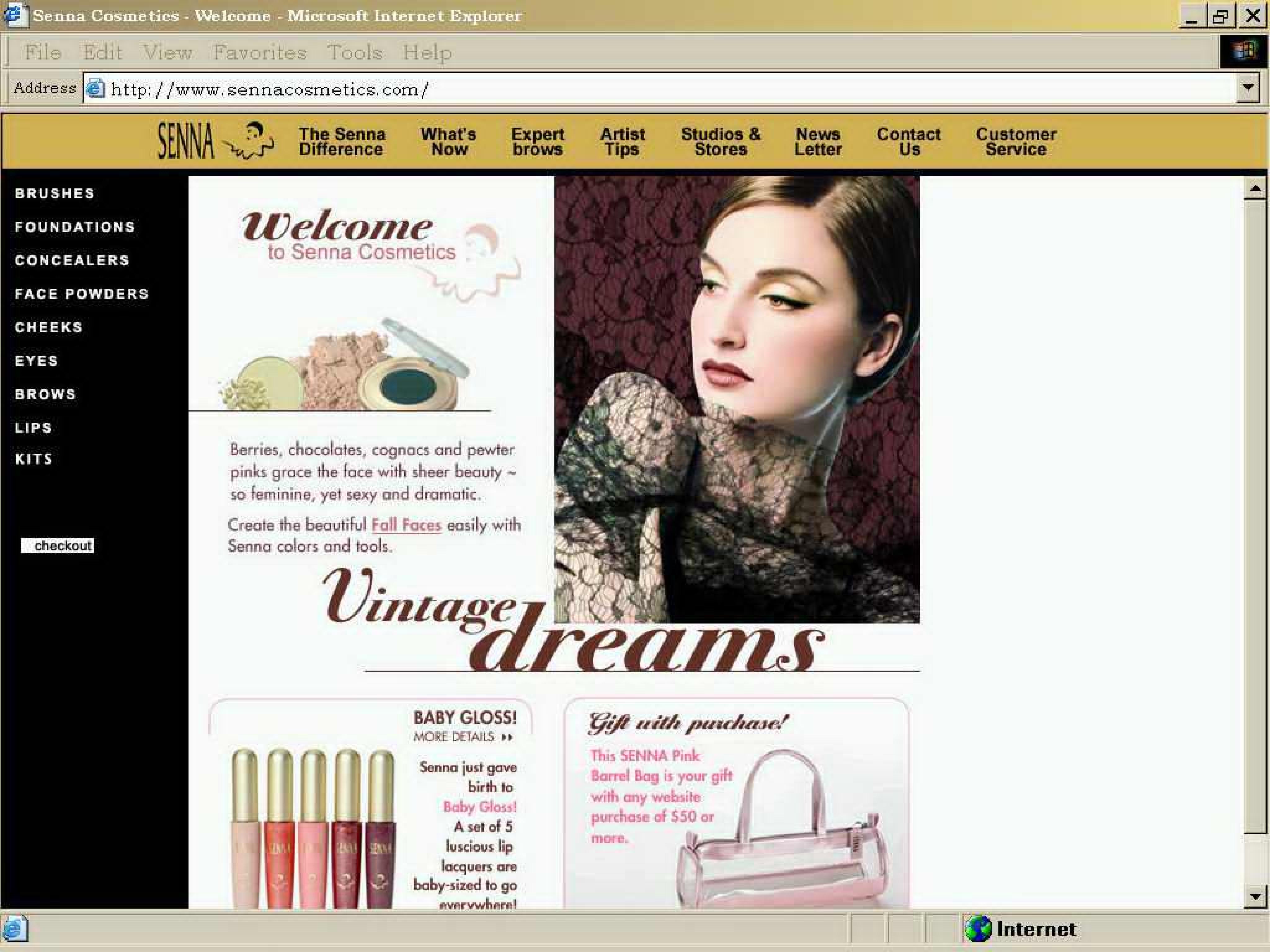 SENNA 75170094 2188775 Live/Registered |
Senna Cosmetics, Inc. 1996-09-23 |
 SENNA 74561186 not registered Dead/Abandoned |
Senna Cosmetics, Inc. 1994-08-15 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.