NDC 21695-857
Necon
Norethindrone And Ethinyl Estradiol
Necon is a Kit in the Human Prescription Drug category. It is labeled and distributed by Rebel Distributors Corp. The primary component is .
| Product ID | 21695-857_2de68f9e-fa13-4b87-84f9-dbb73883ba40 |
| NDC | 21695-857 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Necon |
| Generic Name | Norethindrone And Ethinyl Estradiol |
| Dosage Form | Kit |
| Marketing Start Date | 1987-01-29 |
| Marketing Category | ANDA / ANDA |
| Application Number | ANDA070687 |
| Labeler Name | Rebel Distributors Corp |
| Active Ingredient Strength | 0 |
| NDC Exclude Flag | E |
| Listing Certified Through | 2017-12-31 |
Packaging
NDC 21695-857-01
6 BLISTER PACK in 1 CARTON (21695-857-01) > 1 KIT in 1 BLISTER PACK
| Marketing Start Date | 1987-01-29 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 21695-857-01 [21695085701]
Necon KIT
| Marketing Category | ANDA |
| Application Number | ANDA070687 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 1987-01-29 |
| Inactivation Date | 2019-09-24 |
Drug Details
OpenFDA Data
| SPL SET ID: | 2de68f9e-fa13-4b87-84f9-dbb73883ba40 |
| Manufacturer | |
| RxNorm Concept Unique ID - RxCUI |
NDC Crossover Matching brand name "Necon" or generic name "Norethindrone And Ethinyl Estradiol"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 21695-857 | Necon | Norethindrone and Ethinyl Estradiol |
| 51862-892 | Necon | Necon |
| 50090-1477 | ALYACEN 1/35 | norethindrone and ethinyl estradiol |
| 50090-1478 | ALYACEN 7/7/7 | norethindrone and ethinyl estradiol |
| 0555-9066 | Aranelle | norethindrone and ethinyl estradiol |
| 0555-9034 | Balziva | Norethindrone and Ethinyl Estradiol |
| 0603-7521 | Cyclafem 1/35 | norethindrone and ethinyl estradiol |
| 0603-7525 | Cyclafem 7/7/7 | norethindrone and ethinyl estradiol |
| 16714-348 | Dasetta 1/35 | Norethindrone and Ethinyl Estradiol |
| 16714-346 | Dasetta 7/7/7 | norethindrone and ethinyl estradiol |
| 0093-3303 | Necon 1/35 | Norethindrone and Ethinyl Estradiol |
| 0555-9008 | Nortrel | Norethindrone and Ethinyl Estradiol |
| 0555-9009 | Nortrel | Norethindrone and Ethinyl Estradiol |
| 0555-9010 | Nortrel | Norethindrone and Ethinyl Estradiol |
| 0555-9012 | Nortrel 7/7/7 | Norethindrone and Ethinyl Estradiol |
| 50090-0159 | Ortho-Novum | norethindrone and ethinyl estradiol |
| 16714-370 | Wera | norethindrone and ethinyl estradiol |
| 50090-1431 | Wera | Norethindrone and Ethinyl Estradiol |
Trademark Results [Necon]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NECON 75009142 2008301 Dead/Cancelled |
Applied Power Inc. 1995-10-23 |
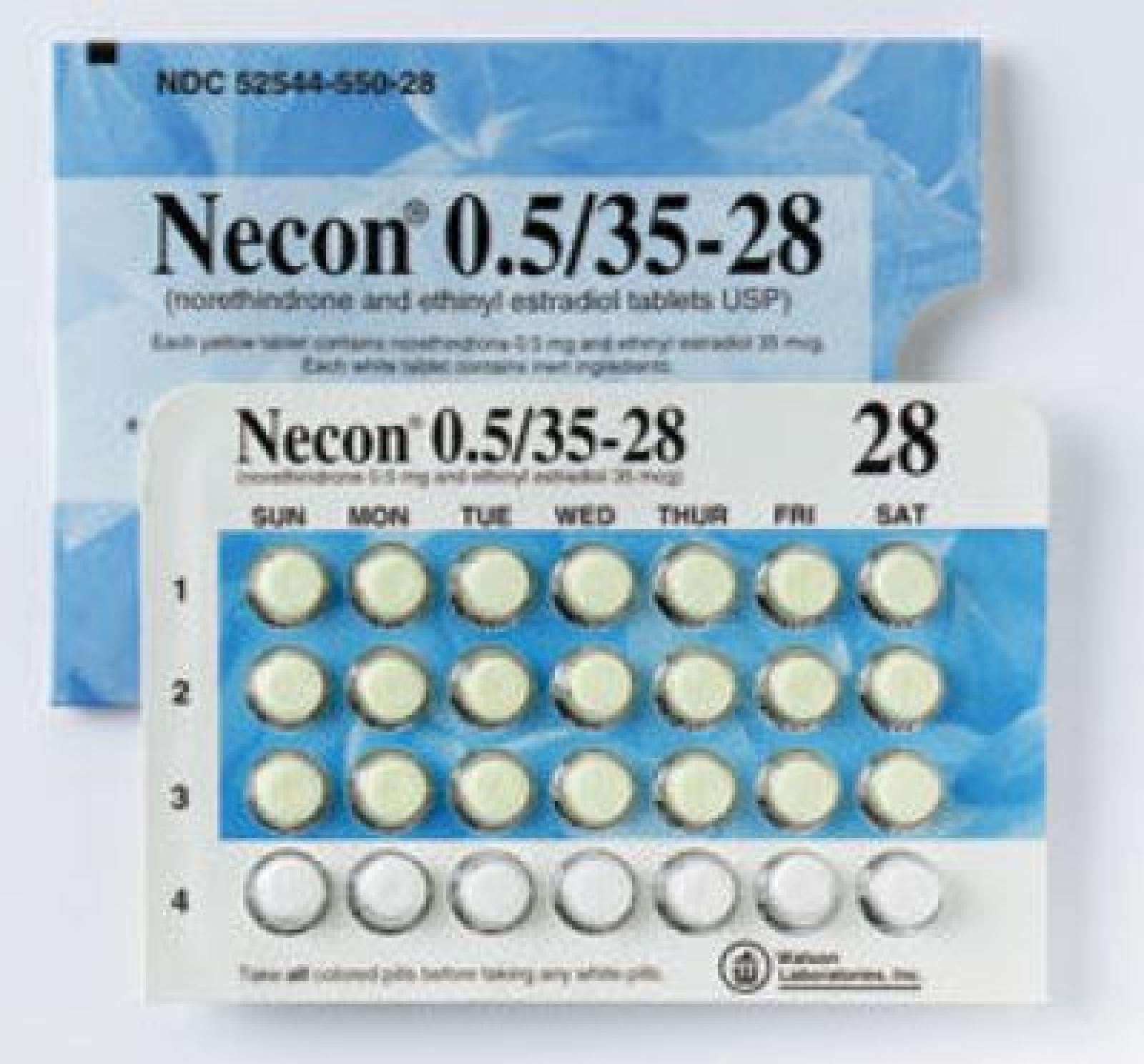 NECON 74536810 1984578 Live/Registered |
MAYNE PHARMA LLC 1994-06-13 |
 NECON 74011081 1709265 Dead/Cancelled |
AUSMEDICS PTY LTD 1989-12-15 |
 NECON 73398161 1284825 Dead/Cancelled |
Alzo Inc. 1982-09-30 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.