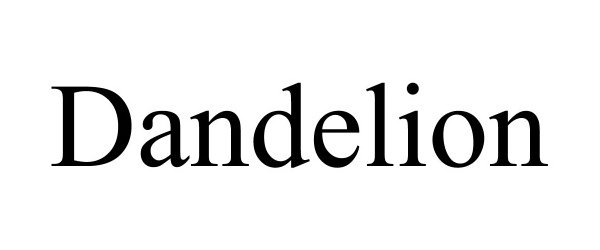NDC 22840-1572
Dandelion
Taraxacum Officinale
Dandelion is a Intradermal; Percutaneous; Subcutaneous Solution in the Non-standardized Allergenic category. It is labeled and distributed by Greer Laboratories, Inc.. The primary component is Taraxacum Officinale Pollen.
| Product ID | 22840-1572_7a051033-10a1-0a21-e053-2a91aa0a18ce |
| NDC | 22840-1572 |
| Product Type | Non-standardized Allergenic |
| Proprietary Name | Dandelion |
| Generic Name | Taraxacum Officinale |
| Dosage Form | Solution |
| Route of Administration | INTRADERMAL; PERCUTANEOUS; SUBCUTANEOUS |
| Marketing Start Date | 1981-09-15 |
| Marketing Category | BLA / BLA |
| Application Number | BLA101833 |
| Labeler Name | Greer Laboratories, Inc. |
| Substance Name | TARAXACUM OFFICINALE POLLEN |
| Active Ingredient Strength | 20000 [PNU]/mL |
| Pharm Classes | Non-Standardized Pollen Allergenic Extract [EPC],Increased Histamine Release [PE],Cell-mediated Immunity [PE],Increased IgG Production [PE],Pollen [CS],Allergens [CS] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2019-12-31 |
Packaging
NDC 22840-1572-2
10 mL in 1 VIAL, MULTI-DOSE (22840-1572-2)
| Marketing Start Date | 1981-09-15 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 22840-1572-2 [22840157202]
Dandelion SOLUTION
| Marketing Category | BLA |
| Application Number | BLA101833 |
| Product Type | NON-STANDARDIZED ALLERGENIC |
| Marketing Start Date | 1981-09-15 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| TARAXACUM OFFICINALE POLLEN | 20000 [PNU]/mL |
Pharmacological Class
- Non-Standardized Pollen Allergenic Extract [EPC]
- Increased Histamine Release [PE]
- Cell-mediated Immunity [PE]
- Increased IgG Production [PE]
- Pollen [CS]
- Allergens [CS]
NDC Crossover Matching brand name "Dandelion" or generic name "Taraxacum Officinale"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0268-7100 | DANDELION | TARAXACUM OFFICINALE POLLEN |
| 22840-1500 | Dandelion | Taraxacum officinale |
| 22840-1501 | Dandelion | Taraxacum officinale |
| 22840-1502 | Dandelion | Taraxacum officinale |
| 22840-1571 | Dandelion | Taraxacum officinale |
| 22840-1572 | Dandelion | Taraxacum officinale |
| 22840-1573 | Dandelion | Taraxacum officinale |
| 22840-5501 | Dandelion | Taraxacum officinale |
| 36987-3125 | Dandelion | Dandelion |
| 36987-3126 | Dandelion | Dandelion |
| 36987-3127 | Dandelion | Dandelion |
| 36987-3128 | Dandelion | Dandelion |
| 36987-3129 | Dandelion | Dandelion |
| 36987-3130 | Dandelion | Dandelion |
| 36987-3131 | Dandelion | Dandelion |
| 36987-3132 | Dandelion | Dandelion |
| 49288-0169 | Dandelion | Dandelion |
| 13838-1430 | Taraxacum (M) | TARAXACUM OFFICINALE |
| 0220-4933 | Taraxacum officinale | TARAXACUM OFFICINALE |
| 0220-4934 | Taraxacum officinale | TARAXACUM OFFICINALE |
| 37662-1293 | Taraxacum Officinale | Taraxacum Officinale |
| 37662-1294 | Taraxacum Officinale | Taraxacum Officinale |
| 37662-1295 | Taraxacum Officinale | Taraxacum Officinale |
Trademark Results [Dandelion]
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.