NDC 29860-203
Virx
Instant Hand Sanitizer With Security Clip
Virx is a Topical Gel in the Human Otc Drug category. It is labeled and distributed by Ningbo United Group Co. Ltd.. The primary component is Alcohol.
| Product ID | 29860-203_57c78e9b-2171-4627-80b1-e9644c022d83 |
| NDC | 29860-203 |
| Product Type | Human Otc Drug |
| Proprietary Name | Virx |
| Generic Name | Instant Hand Sanitizer With Security Clip |
| Dosage Form | Gel |
| Route of Administration | TOPICAL |
| Marketing Start Date | 2010-01-01 |
| Marketing Category | OTC MONOGRAPH FINAL / OTC MONOGRAPH FINAL |
| Application Number | part333 |
| Labeler Name | Ningbo United Group Co. Ltd. |
| Substance Name | ALCOHOL |
| Active Ingredient Strength | 74 mL/120mL |
| NDC Exclude Flag | E |
| Listing Certified Through | 2017-12-31 |
Packaging
NDC 29860-203-14
120 mL in 1 BOTTLE, PLASTIC (29860-203-14)
| Marketing Start Date | 2010-01-01 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 29860-203-14 [29860020314]
Virx GEL
| Marketing Category | OTC monograph final |
| Application Number | part333 |
| Product Type | HUMAN OTC DRUG |
| Marketing Start Date | 2010-01-01 |
| Inactivation Date | 2019-10-21 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| ALCOHOL | 74.4 mL/120mL |
OpenFDA Data
| SPL SET ID: | 63f36498-f2da-4ce8-9d79-ba10155b6cc2 |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI | |
| UPC Code |
NDC Crossover Matching brand name "Virx" or generic name "Instant Hand Sanitizer With Security Clip"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 29860-200 | Virx | Instant Hand Sanitizer |
| 29860-203 | Virx | Instant Hand Sanitizer with Security Clip |
| 29860-205 | Virx | Instant Hand Sanitizer |
| 40104-556 | Virx | Instant Hand Sanitizing Spray with Aloe Vera |
| 40104-703 | Virx | Instant Hand Sanitizer |
| 40104-811 | Virx | Hand Sanitizer and Moisturizer with Shea Butter |
| 50438-807 | Virx | Instant Hand Sanitizer |
Trademark Results [Virx]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
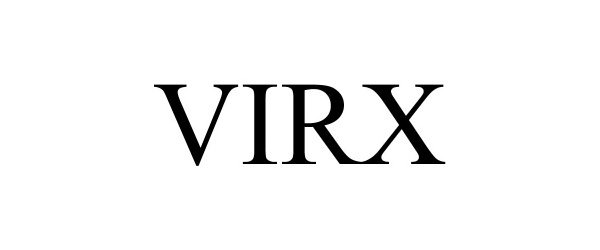 VIRX 97053535 not registered Live/Pending |
Oven Industries, Inc. 2021-09-30 |
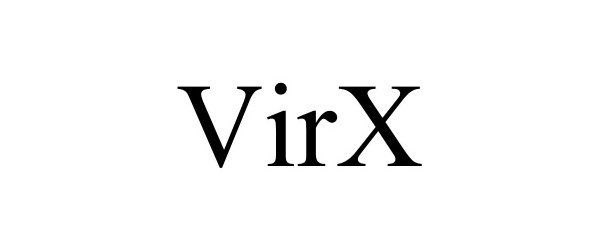 VIRX 90840548 not registered Live/Pending |
SaNOtize Research and Development Corp. 2021-07-21 |
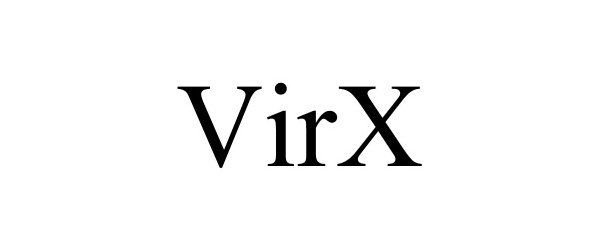 VIRX 90494079 not registered Live/Pending |
HMI Industries, Inc. 2021-01-28 |
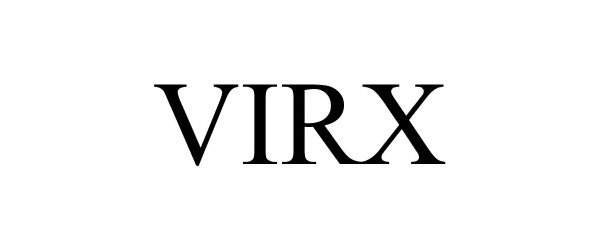 VIRX 88503688 not registered Live/Pending |
FILO AMERICA 2019-07-08 |
 VIRX 86526109 not registered Dead/Abandoned |
MedForce Solutions, Inc. 2015-02-05 |
 VIRX 77230009 3397984 Dead/Cancelled |
Filo America 2007-07-15 |
 VIRX 75561284 not registered Dead/Abandoned |
Gel Tech Industries, Inc. 1998-09-29 |
 VIRX 73741458 1563908 Dead/Cancelled |
VIRX, INC. 1988-07-21 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.