NDC 49281-420
FLUZONE QUADRIVALENT
Influenza A Virus A/guangdong-maonan/swl1536/2019 Cnic-1909 (h1n1) Antigen (formaldehyde Inactivated), Influenza A Virus A/hong Kong/2671/2019 (h3n2) Antigen (formaldehyde Inactivated), Influenza B Virus B/phuket/3073/2013 Antigen (formaldehyde Inactivated), And Influenza B Virus B/washington/02/2019 Antigen (formaldehyde Inactivated)
FLUZONE QUADRIVALENT is a Intramuscular Injection, Suspension in the Vaccine category. It is labeled and distributed by Sanofi Pasteur Inc.. The primary component is Influenza A Virus A/guangdong-maonan/swl1536/2019 Cnic-1909 (h1n1) Antigen (formaldehyde Inactivated); Influenza A Virus A/hong Kong/2671/2019 Ivr-208 (h3n2) Antigen (formaldehyde Inactivated); Influenza B Virus B/phuket/3073/2013 Antigen (formaldehyde Inactivated); Influenza B Virus B/washington/02/2019 Antigen (formaldehyde Inactivated).
| Product ID | 49281-420_b0c45c5e-1402-4009-a82f-032165ccfa23 |
| NDC | 49281-420 |
| Product Type | Vaccine |
| Proprietary Name | FLUZONE QUADRIVALENT |
| Generic Name | Influenza A Virus A/guangdong-maonan/swl1536/2019 Cnic-1909 (h1n1) Antigen (formaldehyde Inactivated), Influenza A Virus A/hong Kong/2671/2019 (h3n2) Antigen (formaldehyde Inactivated), Influenza B Virus B/phuket/3073/2013 Antigen (formaldehyde Inactivated), And Influenza B Virus B/washington/02/2019 Antigen (formaldehyde Inactivated) |
| Dosage Form | Injection, Suspension |
| Route of Administration | INTRAMUSCULAR |
| Marketing Start Date | 2020-07-01 |
| Marketing Category | BLA / BLA |
| Application Number | BLA103914 |
| Labeler Name | Sanofi Pasteur Inc. |
| Substance Name | INFLUENZA A VIRUS A/GUANGDONG-MAONAN/SWL1536/2019 CNIC-1909 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED); INFLUENZA A VIRUS A/HONG KONG/2671/2019 IVR-208 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED); INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED); INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| Active Ingredient Strength | 15 ug/.5mL; ug/.5mL; ug/.5mL; ug/.5mL |
| NDC Exclude Flag | N |
| Listing Certified Through | 2021-12-31 |
Packaging
NDC 49281-420-10
10 VIAL, SINGLE-DOSE in 1 PACKAGE (49281-420-10) > .5 mL in 1 VIAL, SINGLE-DOSE (49281-420-58)
| Marketing Start Date | 2020-07-02 |
| Marketing End Date | 2021-06-30 |
| NDC Exclude Flag | N |
| Sample Package? | N |
Drug Details
NDC Crossover Matching brand name "FLUZONE QUADRIVALENT" or generic name "Influenza A Virus A/guangdong-maonan/swl1536/2019 Cnic-1909 (h1n1) Antigen (formaldehyde Inactivated), Influenza A Virus A/hong Kong/2671/2019 (h3n2) Antigen (formaldehyde Inactivated), Influenza B Virus B/phuket/3073/2013 Antigen (formaldehyde Inactivated), And Influenza B Virus B/washington/02/2019 Antigen (formaldehyde Inactivated)"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 49281-418 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/Michigan/45/2015 X-275 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/SINGAPORE/INFIMH-16-0019/2016 IVR-186 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/MARYLAND/15/2016 BX-69A ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-419 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/BRISBANE/02/2018 IVR-190 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/KANSAS/14/2017 X-327 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/MARYLAND/15/2016 BX-69A ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-420 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/GUANGDONG-MAONAN/SWL1536/2019 CNIC-1909 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/HONG KONG/2671/2019 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-421 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/Victoria/2570/2019 IVR-215 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/TASMANIA/503/2020 IVR-221 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-518 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/Michigan/45/2015 X-275 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/SINGAPORE/INFIMH-16-0019/2016 IVR-186 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/MARYLAND/15/2016 BX-69A ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-519 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/BRISBANE/02/2018 IVR-190 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/KANSAS/14/2017 X-327 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/MARYLAND/15/2016 BX-69A ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-520 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/GUANGDONG-MAONAN/SWL1536/2019 CNIC-1909 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/HONG KONG/2671/2019 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-521 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/Victoria/2570/2019 IVR-215 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/TASMANIA/503/2020 IVR-221 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-629 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/Michigan/45/2015 X-275 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/SINGAPORE/INFIMH-16-0019/2016 IVR-186 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/MARYLAND/15/2016 BX-69A ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-631 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/BRISBANE/02/2018 IVR-190 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/KANSAS/14/2017 X-327 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/MARYLAND/15/2016 BX-69A ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-633 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/GUANGDONG-MAONAN/SWL1536/2019 CNIC-1909 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/HONG KONG/2671/2019 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-635 | FLUZONE QUADRIVALENT | INFLUENZA A VIRUS A/Victoria/2570/2019 IVR-215 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/TASMANIA/503/2020 IVR-221 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
| 49281-120 | FLUZONE High-Dose Quadrivalent Northern Hemisphere | INFLUENZA A VIRUS A/GUANGDONG-MAONAN/SWL1536/2019 CNIC-1909 (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/HONG KONG/2671/2019 (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/PHUKET/3073/2013 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/WASHINGTON/02/2019 ANTIGEN (FORMALDEHYDE INACTIVATED) |
Trademark Results [FLUZONE QUADRIVALENT]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
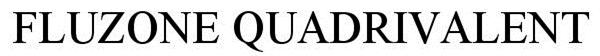 FLUZONE QUADRIVALENT 85786237 4492252 Live/Registered |
Sanofi Pasteur Limited / Sanofi PasteurLimitée 2012-11-23 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.