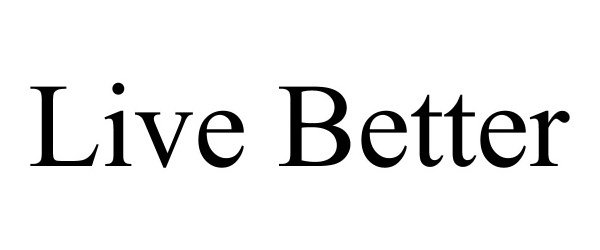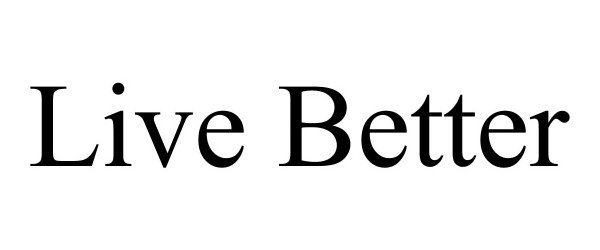| NDC | Brand Name | Generic Name |
|---|
| 0404-0167 | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 41520-016 | CAREONE | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 57243-016 | CVP | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 41163-016 | equaline | bacitracin zinc, neomycin sulfate, and polymyxin B sulfate |
| 51143-016 | Live Better | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 41250-500 | meijer | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 58232-4002 | Neosporin Original | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 41415-049 | Publix | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 63868-356 | Quality Choice Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 37012-016 | Shopko | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 36800-016 | TopCare | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 0924-5610 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 36800-564 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 37012-999 | Triple Antibiotic | BACITRACIN ZINC, NEOMYCIN SULFATE, and POLYMYXIN B SULFATE |
| 46122-414 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 49035-016 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 49035-120 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 50382-023 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 51672-2016 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 51672-2120 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 52426-020 | Triple Antibiotic | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 55315-180 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and POLYMYXIN B SULFATE |
| 57243-120 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 61269-179 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B sulfate |
| 62011-0098 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 62011-0321 | Triple Antibiotic | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 67234-031 | TRIPLE ANTIBIOTIC | BACITRACIN ZINC, NEOMYCIN SULFATE, and POLYMYXIN B SULFATE |
| 54162-080 | Triple Antibiotic GRX | Bacitracin Zinc, Neomycin Sulfate, and Polymyxin B Sulfate |
| 65923-391 | UEC Medical First Aid Antibiotic | Bacitracin zinc, neomycin sulfate, and polymyxin b sulfate |
| 0363-2016 | Walgreens | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 55312-500 | Western Family | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |
| 67091-274 | WinCo Original Strength Triple Antibiotic | Bacitracin zinc, Neomycin sulfate, and Polymyxin B sulfate |