NDC 65584-200
SaniGel
Ethyl Alcohol
SaniGel is a Topical Gel in the Human Otc Drug category. It is labeled and distributed by Higley Industries, Inc.. The primary component is Alcohol.
| Product ID | 65584-200_4ef039ad-50b5-48e4-a7b9-c24b3f738b4f |
| NDC | 65584-200 |
| Product Type | Human Otc Drug |
| Proprietary Name | SaniGel |
| Generic Name | Ethyl Alcohol |
| Dosage Form | Gel |
| Route of Administration | TOPICAL |
| Marketing Start Date | 2020-09-16 |
| Marketing Category | OTC MONOGRAPH NOT FINAL / OTC MONOGRAPH NOT FINAL |
| Application Number | part333A |
| Labeler Name | Higley Industries, Inc. |
| Substance Name | ALCOHOL |
| Active Ingredient Strength | 1 mL/mL |
| NDC Exclude Flag | N |
| Listing Certified Through | 2021-12-31 |
Packaging
NDC 65584-200-10
3790 mL in 1 BOTTLE (65584-200-10)
| Marketing Start Date | 2020-09-16 |
| NDC Exclude Flag | N |
| Sample Package? | N |
Drug Details
NDC Crossover Matching brand name "SaniGel" or generic name "Ethyl Alcohol"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 65584-200 | SaniGel | SaniGel |
| 0363-1050 | A Little Something Eucalyptus Mist Hand Sanitier | Ethyl Alcohol |
| 0363-1051 | A Little Something Prosecco Bubbles Hand Sanitizer | Ethyl Alcohol |
| 0363-0952 | Advanced Hand Sanitizer | Ethyl Alcohol |
| 0363-0663 | Crisp Apple Hand Sani | Ethyl Alcohol |
| 0363-0876 | Ethyl Rubbing Alcohol | Ethyl Alcohol |
| 0113-7077 | hand sanitizer | ethyl alcohol |
| 0363-0469 | Hand Sanitizer | Ethyl Alcohol |
| 0363-0596 | Hand Sanitizer | Ethyl Alcohol |
| 0363-0643 | Hand Sanitizer | Ethyl Alcohol |
| 0363-0826 | Hand Sanitizer | Ethyl Alcohol |
| 0363-0890 | Hand Sanitizer | Ethyl Alcohol |
| 0363-0898 | Hand Sanitizer | Ethyl Alcohol |
| 0363-0984 | Hand Sanitizer | Ethyl Alcohol |
| 0064-1080 | SURGICEPT | ethyl alcohol |
| 0363-5721 | Walgreens | Ethyl Alcohol |
Trademark Results [SaniGel]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
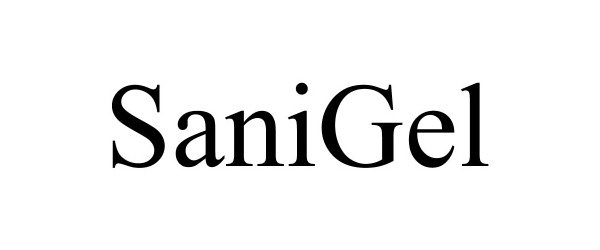 SANIGEL 88837503 not registered Live/Pending |
Valisa Mfg., LLC 2020-03-17 |
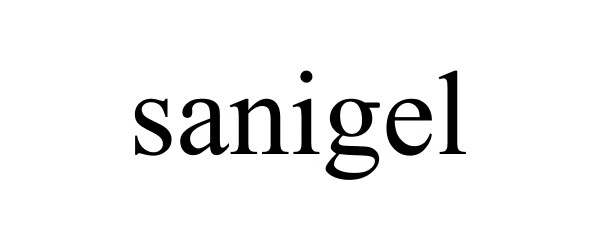 SANIGEL 78636520 not registered Dead/Abandoned |
John T. Novak 2005-05-24 |
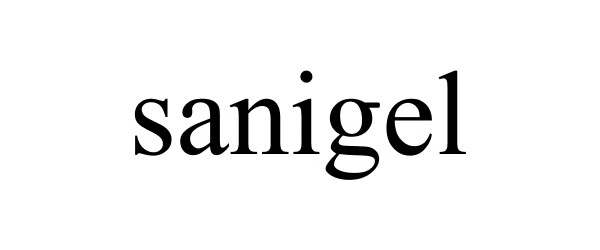 SANIGEL 78636513 not registered Dead/Abandoned |
John T. Novak 2005-05-24 |
 SANIGEL 76261263 not registered Dead/Abandoned |
Apogee, Inc. 2001-05-23 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.