NDC 71269-040
Tuxarin
Codeine Phosphate And Chlorpheniramine Maleate
Tuxarin is a Oral Tablet, Extended Release in the Human Prescription Drug category. It is labeled and distributed by Mainpointe Pharmaceuticals. The primary component is Codeine Phosphate; Chlorpheniramine Maleate.
| Product ID | 71269-040_7ecc03e0-b56e-4ac0-9f91-bd245ef56f32 |
| NDC | 71269-040 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Tuxarin |
| Generic Name | Codeine Phosphate And Chlorpheniramine Maleate |
| Dosage Form | Tablet, Extended Release |
| Route of Administration | ORAL |
| Marketing Start Date | 2018-10-15 |
| Marketing Category | NDA / NDA |
| Application Number | NDA206323 |
| Labeler Name | Mainpointe Pharmaceuticals |
| Substance Name | CODEINE PHOSPHATE; CHLORPHENIRAMINE MALEATE |
| Active Ingredient Strength | 54 mg/1; mg/1 |
| Pharm Classes | Full Opioid Agonists [MoA],Opioid Agonist [EPC],Histamine H1 Receptor Antagonists [MoA],Histamine-1 Receptor Antagonist [EPC] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2019-12-31 |
Packaging
NDC 71269-040-10
100 TABLET, EXTENDED RELEASE in 1 BOTTLE, PLASTIC (71269-040-10)
| Marketing Start Date | 2018-10-15 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 71269-040-10 [71269004010]
Tuxarin TABLET, EXTENDED RELEASE
| Marketing Category | NDA |
| Application Number | NDA206323 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 2018-10-15 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| CODEINE PHOSPHATE | 54.3 mg/1 |
OpenFDA Data
| SPL SET ID: | a7cfab67-6087-4ab4-b7cd-4f9295413180 |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI | |
| UPC Code |
Pharmacological Class
- Full Opioid Agonists [MoA]
- Opioid Agonist [EPC]
- Histamine H1 Receptor Antagonists [MoA]
- Histamine-1 Receptor Antagonist [EPC]
Trademark Results [Tuxarin]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
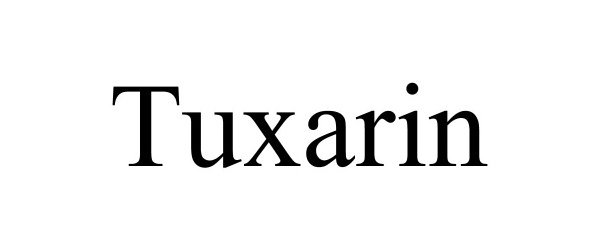 TUXARIN 97027082 not registered Live/Pending |
MainPointe Pharmaceuticals, LLC 2021-09-14 |
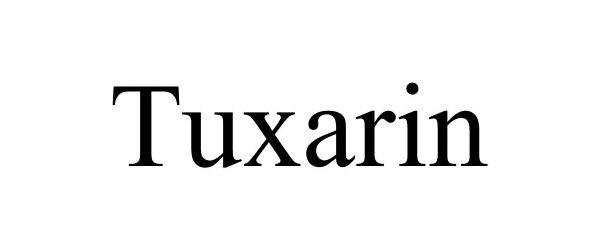 TUXARIN 86659512 not registered Dead/Abandoned |
Spriaso LLC 2015-06-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.