NDC 72839-265
Formula 2 Skin Care
Lidocaine 5%
Formula 2 Skin Care is a Topical Cream in the Human Otc Drug category. It is labeled and distributed by Derma Care Research Labs. The primary component is Lidocaine.
| Product ID | 72839-265_a05d5e07-ae4a-38cc-e053-2a95a90abced |
| NDC | 72839-265 |
| Product Type | Human Otc Drug |
| Proprietary Name | Formula 2 Skin Care |
| Generic Name | Lidocaine 5% |
| Dosage Form | Cream |
| Route of Administration | TOPICAL |
| Marketing Start Date | 2020-03-09 |
| Marketing Category | OTC MONOGRAPH FINAL / OTC MONOGRAPH FINAL |
| Application Number | part346 |
| Labeler Name | Derma Care Research Labs |
| Substance Name | LIDOCAINE |
| Active Ingredient Strength | 5 g/100g |
| NDC Exclude Flag | N |
| Listing Certified Through | 2021-12-31 |
Packaging
NDC 72839-265-00
28 g in 1 TUBE (72839-265-00)
| Marketing Start Date | 2020-03-09 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 72839-265-00 [72839026500]
Formula 2 Skin Care CREAM
| Marketing Category | OTC monograph final |
| Application Number | part346 |
| Product Type | HUMAN OTC DRUG |
| Marketing Start Date | 2020-03-09 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| LIDOCAINE | 5 g/100g |
OpenFDA Data
| SPL SET ID: | a05d5e07-ae4b-38cc-e053-2a95a90abced |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI | |
| UPC Code |
NDC Crossover Matching brand name "Formula 2 Skin Care" or generic name "Lidocaine 5%"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 72839-265 | Formula 2 Skin Care | Lidocaine 5% |
| 24357-702 | Anecream 5 | Lidocaine 5% |
| 70942-183 | Doctor Butlers Numb It All Cream | Lidocaine 5% |
| 42291-400 | Lidocaine | Lidocaine 5% |
| 0363-4530 | Lidocaine 5% | Lidocaine 5% |
| 42858-118 | Lidocaine 5% | Lidocaine 5% |
| 63629-2472 | Lidocaine 5% | Lidocaine 5% |
| 68752-015 | Lidocaine 5% | Lidocaine 5% |
| 70518-3522 | Lidocaine 5% | Lidocaine 5% |
| 72275-716 | Lidovix L | Lidocaine 5% |
| 63135-811 | LIPOCAINE 5 | LIDOCAINE 5% |
| 63135-924 | LUBRICAINE 5 | LIDOCAINE 5% |
| 82429-203 | NUMB | Lidocaine 5% |
| 72839-141 | Rectal Care Cream | Lidocaine 5% |
| 69315-301 | RectaSmoothe | Lidocaine 5% |
| 11822-0265 | Rite Aid Rectal Care | Lidocaine 5% |
| 63135-581 | TOPICAINE 5 | LIDOCAINE 5% |
| 57483-108 | Xyralid | Lidocaine 5% |
Trademark Results [Formula 2 Skin Care]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
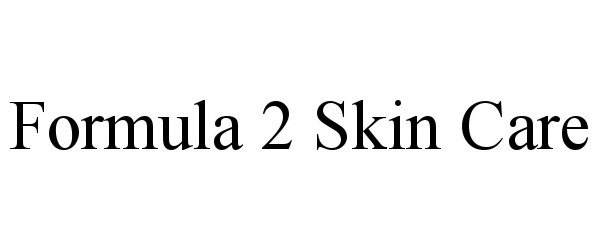 FORMULA 2 SKIN CARE 90775614 not registered Live/Pending |
MSI Enterprises, Inc. 2021-06-15 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.