FLEXTIME
Material, Impression
HERAEUS KULZER, INC.
The following data is part of a premarket notification filed by Heraeus Kulzer, Inc. with the FDA for Flextime.
Pre-market Notification Details
| Device ID | K000629 |
| 510k Number | K000629 |
| Device Name: | FLEXTIME |
| Classification | Material, Impression |
| Applicant | HERAEUS KULZER, INC. 4315 SOUTH LAFAYETTE BLVD. South Bend, IN 46614 -2517 |
| Contact | Cheryl V Zimmerman |
| Correspondent | Cheryl V Zimmerman HERAEUS KULZER, INC. 4315 SOUTH LAFAYETTE BLVD. South Bend, IN 46614 -2517 |
| Product Code | ELW |
| CFR Regulation Number | 872.3660 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2000-02-24 |
| Decision Date | 2000-04-05 |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 00850007728250 | K000629 | 000 |
| 00850078989338 | K000629 | 000 |
| 00850078989376 | K000629 | 000 |
| 00850078989369 | K000629 | 000 |
Trademark Results [FLEXTIME]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FLEXTIME 88541157 not registered Live/Pending |
Teachmore, LLC 2019-07-26 |
 FLEXTIME 86952373 5068602 Live/Registered |
Flextime Tennis LLC 2016-03-24 |
 FLEXTIME 85384923 4276607 Dead/Cancelled |
WGI Innovations, Ltd. 2011-07-29 |
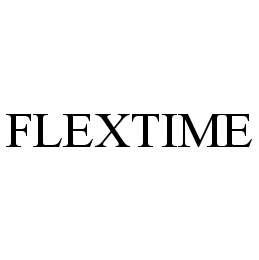 FLEXTIME 78406442 2946175 Dead/Cancelled |
Aerospace Service and Controls, Inc. 2004-04-22 |
 FLEXTIME 78194030 2902309 Live/Registered |
Screenlife, LLC 2002-12-12 |
 FLEXTIME 78124104 not registered Dead/Abandoned |
Aerospace Service & Controls, Inc. 2002-04-25 |
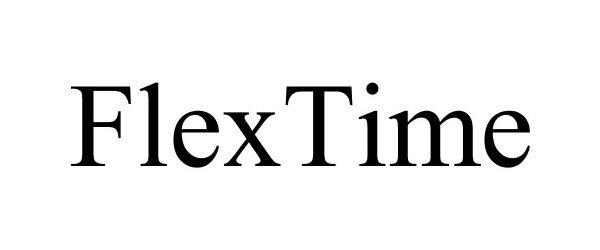 FLEXTIME 77622920 not registered Dead/Abandoned |
Cachet Banq 2008-11-26 |
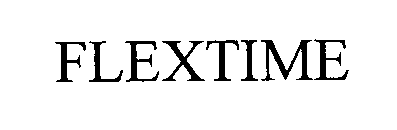 FLEXTIME 76445049 not registered Dead/Abandoned |
Olson, Michelle 2002-08-29 |
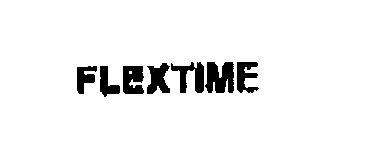 FLEXTIME 76286416 not registered Dead/Abandoned |
FLEXTIME, LLC 2001-07-10 |
 FLEXTIME 76134139 2626176 Live/Registered |
VITAMINS DIRECT (BERMUDA) LIMITED 2000-09-25 |
 FLEXTIME 75558062 2373823 Dead/Cancelled |
Motorola, Inc. 1998-09-24 |
 FLEXTIME 74657299 not registered Dead/Abandoned |
Motorola, Inc. 1995-03-31 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.