QUIK-CAP
Electrode, Cutaneous
NEUROSCAN
The following data is part of a premarket notification filed by Neuroscan with the FDA for Quik-cap.
Pre-market Notification Details
| Device ID | K000865 |
| 510k Number | K000865 |
| Device Name: | QUIK-CAP |
| Classification | Electrode, Cutaneous |
| Applicant | NEUROSCAN 45150 BUSINESS CT., SUITE 100 Sterling, VA 20166 |
| Contact | David B Jones |
| Correspondent | David B Jones NEUROSCAN 45150 BUSINESS CT., SUITE 100 Sterling, VA 20166 |
| Product Code | GXY |
| CFR Regulation Number | 882.1320 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2000-03-17 |
| Decision Date | 2000-04-18 |
NIH GUDID Devices
Trademark Results [QUIK-CAP]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
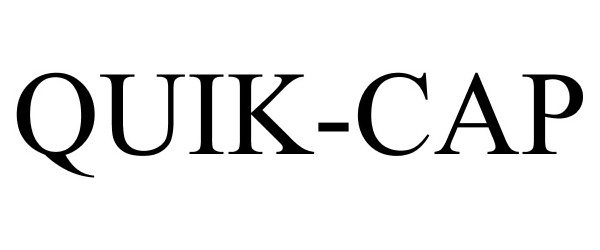 QUIK-CAP 77195815 3535962 Live/Registered |
Compumedics Limited 2007-06-01 |
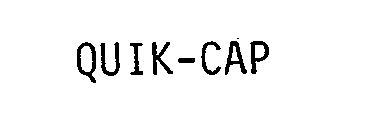 QUIK-CAP 76678090 3516880 Dead/Cancelled |
HAMPTON PRODUCTS INTERNATIONAL CORPORATION 2007-06-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.