EAR MUFFINS
Device, Biofeedback
BIO-LOGIC SYSTEMS CORP.
The following data is part of a premarket notification filed by Bio-logic Systems Corp. with the FDA for Ear Muffins.
Pre-market Notification Details
| Device ID | K012384 |
| 510k Number | K012384 |
| Device Name: | EAR MUFFINS |
| Classification | Device, Biofeedback |
| Applicant | BIO-LOGIC SYSTEMS CORP. ONE BIO-LOGIC PLAZA Mundelein, IL 60060 -3700 |
| Contact | Norman E Brunner |
| Correspondent | Norman E Brunner BIO-LOGIC SYSTEMS CORP. ONE BIO-LOGIC PLAZA Mundelein, IL 60060 -3700 |
| Product Code | HCC |
| CFR Regulation Number | 882.5050 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2001-07-27 |
| Decision Date | 2001-10-15 |
| Summary: | summary |
Trademark Results [EAR MUFFINS]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
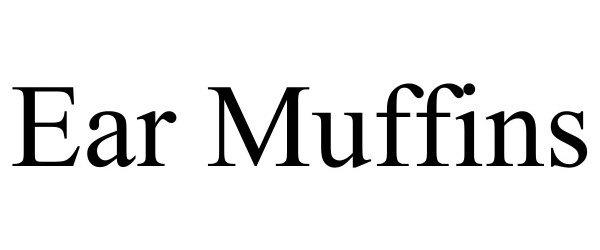 EAR MUFFINS 98146685 not registered Live/Pending |
Wormser, Brett A 2023-08-23 |
 EAR MUFFINS 85716157 not registered Dead/Abandoned |
Wormser, Brett 2012-08-29 |
 EAR MUFFINS 76260627 not registered Dead/Abandoned |
Bio-logic Systems Corp 2001-05-21 |
 EAR MUFFINS 75691558 not registered Dead/Abandoned |
Paris Accessories, Inc. 1999-04-26 |
 EAR MUFFINS 74243482 not registered Dead/Abandoned |
Linea Pelle, Inc. 1992-02-05 |
 EAR MUFFINS 73304348 1222669 Dead/Cancelled |
Rayman; William 1981-04-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.