HI-OX
Valve, Non-rebreathing
SENSORMEDICS CORP.
The following data is part of a premarket notification filed by Sensormedics Corp. with the FDA for Hi-ox.
Pre-market Notification Details
| Device ID | K020665 |
| 510k Number | K020665 |
| Device Name: | HI-OX |
| Classification | Valve, Non-rebreathing |
| Applicant | SENSORMEDICS CORP. 22705 SAVI RANCH PKWY. Yorba Linda, CA 92887 -4645 |
| Contact | Earl W Draper |
| Correspondent | Earl W Draper SENSORMEDICS CORP. 22705 SAVI RANCH PKWY. Yorba Linda, CA 92887 -4645 |
| Product Code | CBP |
| CFR Regulation Number | 868.5870 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2002-03-01 |
| Decision Date | 2002-07-30 |
| Summary: | summary |
Trademark Results [HI-OX]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
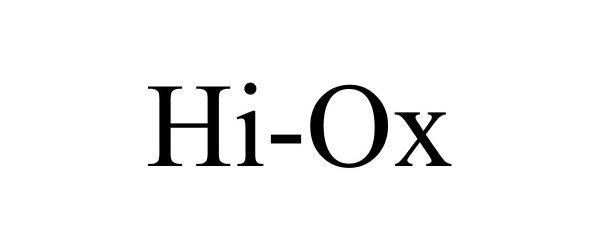 HI-OX 85530896 4273363 Live/Registered |
12TH MAN TECHNOLOGIES, INC. 2012-02-01 |
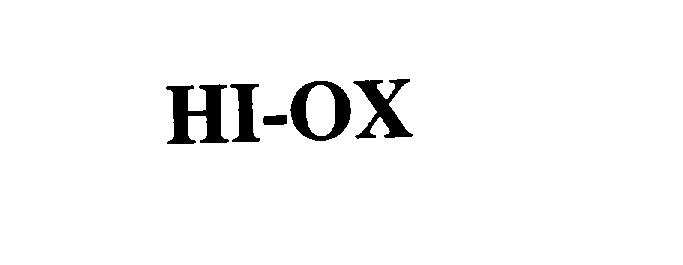 HI-OX 76141796 2879851 Dead/Cancelled |
SENSORMEDICS CORPORATION 2000-10-05 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.