TACAHE
Wheelchair, Powered
DALTON MEDICAL CORP.
The following data is part of a premarket notification filed by Dalton Medical Corp. with the FDA for Tacahe.
Pre-market Notification Details
| Device ID | K052534 |
| 510k Number | K052534 |
| Device Name: | TACAHE |
| Classification | Wheelchair, Powered |
| Applicant | DALTON MEDICAL CORP. 1103 VENTURE CT. Carrollton, TX 75006 |
| Contact | Will Ridgway |
| Correspondent | Will Ridgway DALTON MEDICAL CORP. 1103 VENTURE CT. Carrollton, TX 75006 |
| Product Code | ITI |
| CFR Regulation Number | 890.3860 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2005-09-15 |
| Decision Date | 2006-01-27 |
| Summary: | summary |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 00815027011768 | K052534 | 000 |
| 00815027011751 | K052534 | 000 |
| 00815027011706 | K052534 | 000 |
| 00815027011690 | K052534 | 000 |
| 00815027011409 | K052534 | 000 |
| 00815027011393 | K052534 | 000 |
| 00815027010686 | K052534 | 000 |
| 00815027010679 | K052534 | 000 |
Trademark Results [TACAHE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 TACAHE 97096768 not registered Live/Pending |
XIAMEN XINGFEIXIANG E-COMMERCE CO.,LTD. 2021-10-28 |
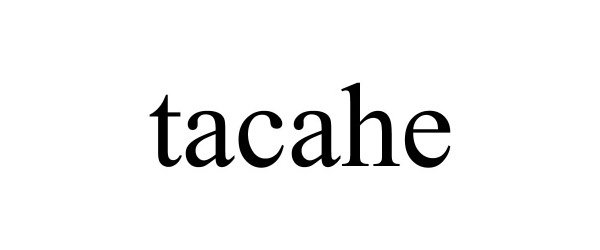 TACAHE 97096760 not registered Live/Pending |
XIAMEN XINGFEIXIANG E-COMMERCE CO.,LTD. 2021-10-28 |
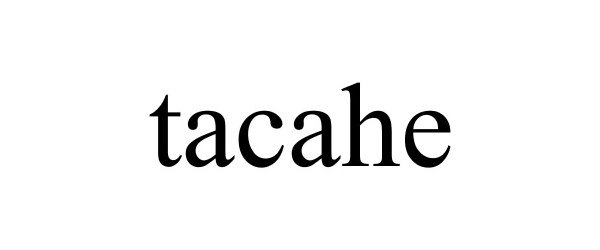 TACAHE 88333416 5862018 Live/Registered |
Xiamen Henghuixiang Technology Co.,Ltd. 2019-03-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.