PULMOLIFE
Spirometer, Diagnostic
MICRO MEDICAL, LTD.
The following data is part of a premarket notification filed by Micro Medical, Ltd. with the FDA for Pulmolife.
Pre-market Notification Details
| Device ID | K061283 |
| 510k Number | K061283 |
| Device Name: | PULMOLIFE |
| Classification | Spirometer, Diagnostic |
| Applicant | MICRO MEDICAL, LTD. QUAYSIDE CHATHAM MARITIME Chatham, Kent, GB Me4 4qy |
| Contact | Glen Hillsley |
| Correspondent | Glen Hillsley MICRO MEDICAL, LTD. QUAYSIDE CHATHAM MARITIME Chatham, Kent, GB Me4 4qy |
| Product Code | BZG |
| CFR Regulation Number | 868.1840 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2006-05-08 |
| Decision Date | 2006-12-14 |
| Summary: | summary |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| 54250892905537 | K061283 | 000 |
| 54250892905520 | K061283 | 000 |
| 54250892905513 | K061283 | 000 |
| 54250892901577 | K061283 | 000 |
| 54250892901560 | K061283 | 000 |
| 54250892901553 | K061283 | 000 |
Trademark Results [PULMOLIFE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
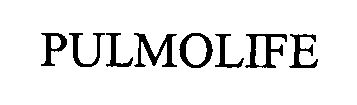 PULMOLIFE 76645664 3232135 Live/Registered |
CAREFUSION U.K. 232 LIMITED 2005-08-26 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.