PLEXUR M
Filler, Bone Void, Calcium Compound
OSTEOTECH, INC.
The following data is part of a premarket notification filed by Osteotech, Inc. with the FDA for Plexur M.
Pre-market Notification Details
| Device ID | K081227 |
| 510k Number | K081227 |
| Device Name: | PLEXUR M |
| Classification | Filler, Bone Void, Calcium Compound |
| Applicant | OSTEOTECH, INC. 51 JAMES WAY Eatontown, NJ 07724 |
| Contact | Chris Talbot |
| Correspondent | Chris Talbot OSTEOTECH, INC. 51 JAMES WAY Eatontown, NJ 07724 |
| Product Code | MQV |
| CFR Regulation Number | 888.3045 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2008-04-30 |
| Decision Date | 2008-07-22 |
| Summary: | summary |
Trademark Results [PLEXUR M]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
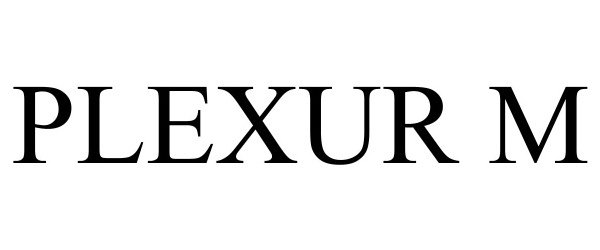 PLEXUR M 98445222 not registered Live/Pending |
McCall International Medical Limited Partnership 2024-03-12 |
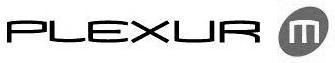 PLEXUR M 77294132 3680435 Dead/Cancelled |
Osteotech, Inc. 2007-10-02 |
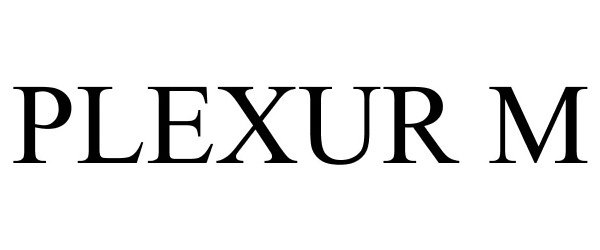 PLEXUR M 77294094 3683687 Dead/Cancelled |
Osteotech, Inc. 2007-10-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.