PRAGER SHELL
Transducer, Ultrasonic, Diagnostic
ESI, INC.
The following data is part of a premarket notification filed by Esi, Inc. with the FDA for Prager Shell.
Pre-market Notification Details
| Device ID | K082893 |
| 510k Number | K082893 |
| Device Name: | PRAGER SHELL |
| Classification | Transducer, Ultrasonic, Diagnostic |
| Applicant | ESI, INC. 377 ROUTE 17 S Hasbrouck Heights, NJ 07604 |
| Contact | George Myers |
| Correspondent | George Myers ESI, INC. 377 ROUTE 17 S Hasbrouck Heights, NJ 07604 |
| Product Code | ITX |
| CFR Regulation Number | 892.1570 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2008-09-30 |
| Decision Date | 2008-12-05 |
| Summary: | summary |
NIH GUDID Devices
| Device Identifier | submissionNumber | Supplement |
|---|---|---|
| B360P110RXP0 | K082893 | 000 |
| B360P1000 | K082893 | 000 |
| B360P100AS0 | K082893 | 000 |
| B360P1010 | K082893 | 000 |
| B360P10120 | K082893 | 000 |
| B360P1020 | K082893 | 000 |
| B360P10220 | K082893 | 000 |
| B360P1030 | K082893 | 000 |
| B360P103GS0 | K082893 | 000 |
| B360P103SV0 | K082893 | 000 |
| B360P1040 | K082893 | 000 |
| B360P104GS0 | K082893 | 000 |
| B360P104SV0 | K082893 | 000 |
| B360P1050 | K082893 | 000 |
| B360P1060 | K082893 | 000 |
| B360P106GS0 | K082893 | 000 |
| B360P106SV0 | K082893 | 000 |
| B360P10720 | K082893 | 000 |
| B360PSVGS0 | K082893 | 000 |
Trademark Results [PRAGER SHELL]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
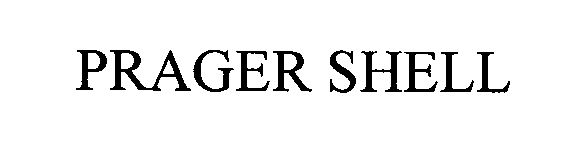 PRAGER SHELL 76484587 2819198 Live/Registered |
ESI, Corporation 2003-01-23 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.